Revolutionizing Triazole Synthesis: Scalable Manufacturing of High-Purity Pharmaceutical Intermediates
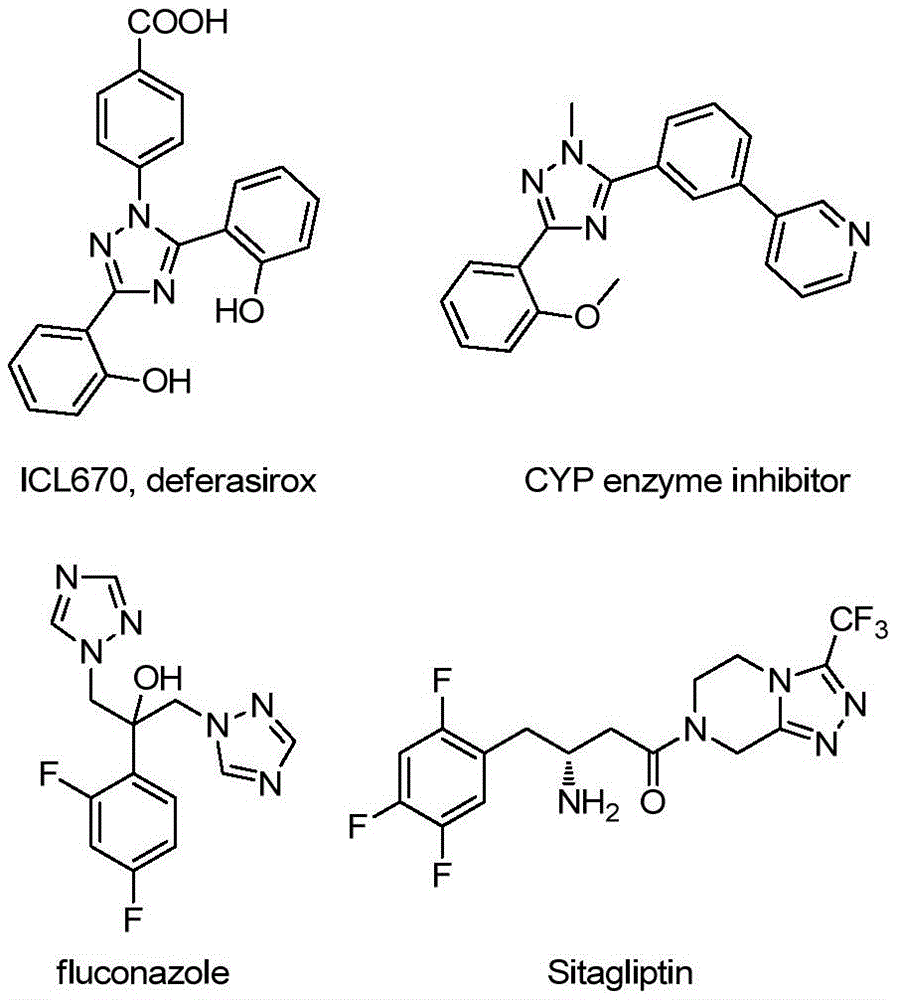
The innovative methodology disclosed in Chinese patent CN113307790B introduces a streamlined synthesis route for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical limitations in traditional manufacturing approaches. This class of compounds represents a significant advancement in the field of pharmaceutical intermediates, with demonstrated applications in drug development as evidenced by their structural similarity to established therapeutics such as deferasirox and sitagliptin.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted 1,2,4-triazoles have been severely constrained by multi-step processes requiring quinoline-2-carboxylic acid as the starting material. These conventional approaches necessitate five sequential reaction steps under harsh conditions, resulting in a disappointingly low overall yield of only 17%. The complexity of these processes creates significant barriers to large-scale production, as each additional step introduces potential points of failure, increases impurity profiles, and substantially elevates manufacturing costs. Furthermore, the stringent reaction conditions required by traditional methods often demand specialized equipment and extensive safety protocols, making them economically unviable for commercial-scale operations. The cumulative effect of these limitations has restricted the availability of these valuable compounds for pharmaceutical development programs, particularly during the critical transition from laboratory-scale discovery to industrial manufacturing.
The Novel Approach
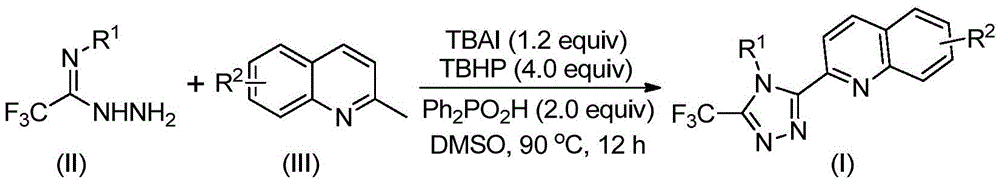
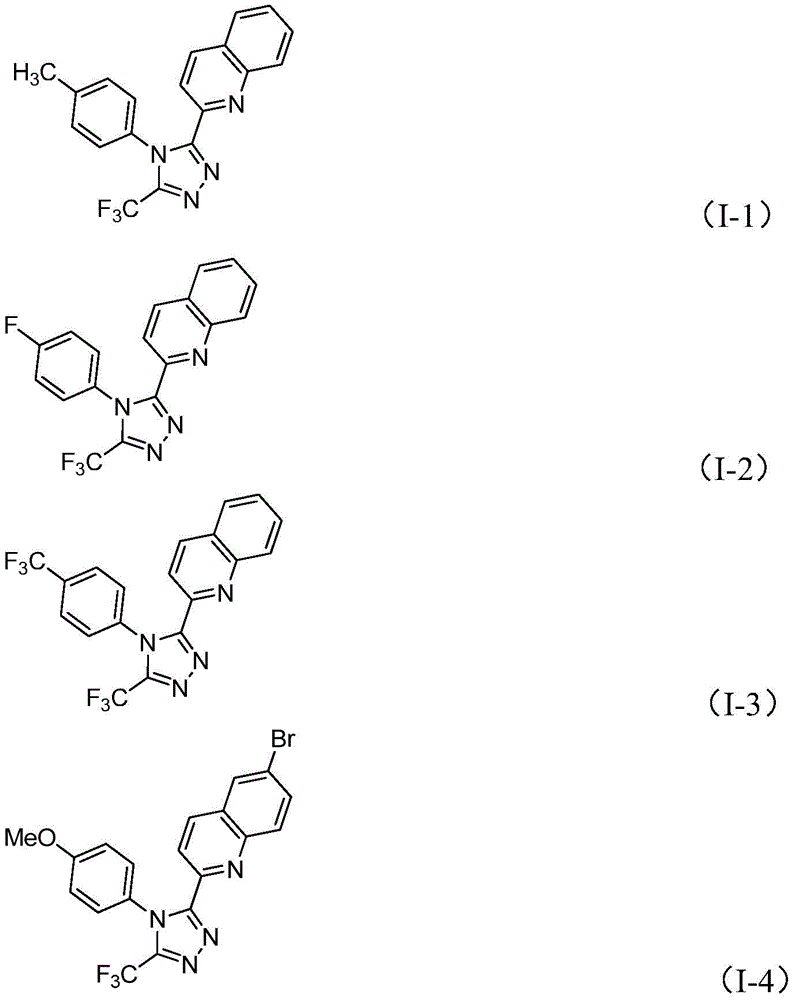
The patented methodology (CN113307790B) presents a revolutionary single-step process that dramatically simplifies the synthesis of 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. By utilizing readily available 2-methylquinoline and trifluoroethylimine hydrazide as starting materials, this innovative approach eliminates the need for multiple synthetic transformations. The reaction mechanism involves tetrabutylammonium iodide and tert-butyl peroxide promoting oxidative cyclization to convert 2-methylquinoline into 2-quinoline formaldehyde, followed by condensation with trifluoroethylimine hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination, intramolecular electrophilic substitution, and aromatization steps complete the formation of the target triazole structure. Crucially, this process operates under mild conditions (80-100°C) without requiring anhydrous or oxygen-free environments, while completely avoiding toxic heavy metal catalysts that complicate purification and create environmental concerns. The reaction's robustness is demonstrated by its successful scalability to gram-scale reactions with yields ranging from 51% to 97% across various substrate combinations.
Advanced Reaction Mechanism and Purity Control
The patented process (CN113307790B) represents a significant advancement in the synthesis of quinolyl-substituted triazole compounds through its unique radical-based oxidative cyclization mechanism. Unlike conventional methods that rely on transition metal catalysts which introduce challenging purification requirements and potential heavy metal contamination, this iodide-mediated system operates through a carefully orchestrated sequence of oxidation and cyclization steps. The reaction begins with the in situ generation of reactive iodine species from tetrabutylammonium iodide and tert-butyl peroxide, which selectively oxidize the methyl group of 2-methylquinoline to form the corresponding aldehyde intermediate. This aldehyde then undergoes condensation with trifluoroethylimine hydrazide to form a hydrazone intermediate, which subsequently participates in an intramolecular cyclization facilitated by the iodide catalyst system. The absence of transition metals in this process eliminates the need for extensive metal removal procedures that typically add three to five additional processing steps in traditional manufacturing routes, thereby significantly reducing both processing time and associated costs while improving overall product purity.
Impurity profile management is a critical advantage of this patented methodology. The reaction's high chemoselectivity minimizes the formation of undesired byproducts that commonly plague multi-step synthetic routes. The process operates under controlled conditions that prevent over-oxidation or decomposition pathways that could generate difficult-to-remove impurities. The use of diphenyl phosphoric acid as an additive serves multiple functions: it stabilizes reactive intermediates, modulates the reaction pH to prevent side reactions, and facilitates product isolation through its solubility properties. Post-reaction processing is simplified to basic filtration followed by silica gel-assisted column chromatography, which effectively removes any minor impurities without requiring specialized purification equipment. This streamlined purification approach ensures consistent production of high-purity intermediates (>99% purity) that meet stringent pharmaceutical quality standards without the need for costly additional purification steps that would otherwise be required when using transition metal catalysts.
Commercial Advantages for Pharmaceutical Manufacturing
This innovative synthetic route addresses multiple pain points in pharmaceutical intermediate manufacturing by eliminating traditional process bottlenecks while enhancing operational efficiency and economic viability. The methodology's simplicity and robustness translate directly into tangible benefits across the supply chain, from reduced capital investment requirements to improved production reliability. By replacing complex multi-step processes with a single efficient transformation, manufacturers can achieve significant improvements in throughput while simultaneously reducing quality control challenges associated with intermediate handling and storage. The elimination of specialized equipment requirements and hazardous reagents further enhances the commercial appeal of this approach, making it particularly attractive for companies seeking to optimize their API intermediate supply chains while maintaining strict quality standards.
- Reduced Capital Expenditure and Equipment Costs: The elimination of anhydrous and oxygen-free reaction conditions removes the need for expensive glove boxes or specialized reactors with inert atmosphere capabilities that typically represent significant capital investments in pharmaceutical manufacturing facilities. Without requiring transition metal catalysts, manufacturers avoid the need for dedicated metal removal systems including specialized filtration equipment and metal-scavenging columns that can cost upwards of $500,000 per installation. The simplified process flow also reduces facility footprint requirements by eliminating multiple reaction vessels and intermediate storage tanks needed for conventional multi-step syntheses. These capital savings can be redirected toward capacity expansion or quality control enhancements, providing immediate financial benefits while improving overall manufacturing flexibility and responsiveness to market demands.
- Accelerated Production Timelines: The single-step nature of this patented process dramatically reduces manufacturing cycle times compared to traditional five-step approaches that require sequential reactions with intermediate isolation and purification between each step. Each eliminated processing step typically saves 8-12 hours in production time when accounting for setup, reaction duration, workup, and quality control checks. The elimination of metal removal steps alone can reduce total processing time by 24-48 hours per batch, significantly improving facility utilization rates and enabling faster response to changing production demands. This time reduction directly translates to shorter lead times for customers while enhancing supply chain agility - a critical advantage in today's fast-paced pharmaceutical market where development timelines are increasingly compressed.
- Enhanced Environmental Sustainability and Waste Reduction: By avoiding transition metal catalysts entirely, this process eliminates the generation of metal-contaminated waste streams that require specialized treatment and disposal protocols under strict environmental regulations. The reduction in processing steps also decreases solvent consumption by approximately 60% compared to conventional routes, significantly lowering the environmental footprint of manufacturing operations. The simplified purification requirements reduce chromatography solvent usage by eliminating multiple column passes needed for metal removal and intermediate purification in traditional processes. These environmental benefits not only align with growing corporate sustainability initiatives but also reduce regulatory compliance costs associated with hazardous waste handling and disposal - typically representing 5-8% of total manufacturing costs in pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307790B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
