Advanced Organocatalytic Synthesis of Chiral Indoxazinones for High-Purity Pharmaceutical Intermediates
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for High-Purity Pharmaceutical Intermediates
The landscape of modern pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more efficient, selective, and scalable synthetic methodologies. A groundbreaking advancement in this field is detailed in patent CN112209947A, which discloses a novel class of chiral indoxazinone compounds and a highly efficient synthesis method thereof. These chiral indole-fused ring systems are of paramount importance in medicinal chemistry, serving as critical scaffolds for various bioactive molecules, including S1P1 receptor antagonists and antimalarial agents. The disclosed technology addresses a significant gap in the prior art, where existing chiral indole compounds often exhibited weak cytotoxic activity against critical cell lines like PC-3 human prostate cancer cells. By leveraging a sophisticated organocatalytic approach, this innovation not only delivers compounds with enhanced biological potency but also establishes a robust platform for the cost reduction in pharmaceutical intermediate manufacturing through streamlined processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex chiral indole-fused ring compounds has been plagued by significant technical and operational challenges that hinder their widespread adoption in commercial drug development. Traditional synthetic routes often necessitate the use of harsh reaction conditions, including extreme temperatures and aggressive reagents, which pose substantial safety risks and increase the likelihood of operational errors leading to accidents. Furthermore, these conventional methods frequently suffer from poor stereocontrol, resulting in low enantioselectivity and the formation of racemic mixtures that require expensive and time-consuming chiral separation processes. The resulting products from these older methodologies have also demonstrated insensitivity to PC-3 human prostate cancer cells, limiting their utility in oncology research. Additionally, the low yields associated with these violent reaction conditions indirectly drive up production costs and generate excessive chemical waste, creating a bottleneck for supply chain reliability and environmental compliance in the fine chemical sector.
The Novel Approach
In stark contrast to these legacy issues, the novel approach described in the patent utilizes a mild, organocatalytic strategy that fundamentally transforms the synthesis landscape for these valuable heterocycles. The method employs 2-indolylmethanol and nitrone as readily available starting materials, reacting them in toluene solvent under the synergistic catalysis of a chiral phosphoric acid and hexafluoroisopropanol. This dual-catalyst system operates effectively at ambient temperatures ranging from 20-30°C, eliminating the need for energy-intensive heating or cryogenic cooling. The reaction proceeds with exceptional efficiency, achieving high yields and extremely high enantioselectivity, which directly translates to superior product purity without the need for complex downstream resolution. As illustrated in the general reaction scheme below, this methodology allows for a broad substrate scope, accommodating various substituents to generate structurally diverse products with high complexity.
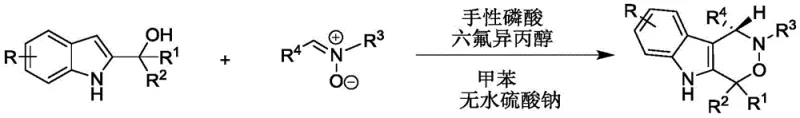
This versatility ensures that the method is not limited to a single compound but serves as a versatile platform for generating a library of potential drug candidates. The simplicity of the work-up procedure, involving basic filtration and concentration, further underscores the practical advantages of this new route over traditional multi-step syntheses.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthesis hinges on the precise mechanistic interplay between the chiral phosphoric acid catalyst and the hexafluoroisopropanol (HFIP) co-catalyst, which creates a highly organized chiral environment for the reaction. The chiral phosphoric acid, typically derived from binaphthyl or spiro skeletons, acts as a Brønsted acid activator, engaging the nitrone substrate through hydrogen bonding interactions to lower the activation energy of the cycloaddition process. Simultaneously, the hexafluoroisopropanol enhances the acidity of the phosphoric acid and stabilizes the transition state through a network of hydrogen bonds, thereby enforcing strict stereocontrol over the formation of the new chiral centers. The specific structure of the catalyst, such as the binaphthyl derivative shown below with bulky 9-phenanthryl groups, plays a critical role in shielding one face of the reactive intermediate, ensuring that the nucleophilic attack occurs exclusively from the desired trajectory.
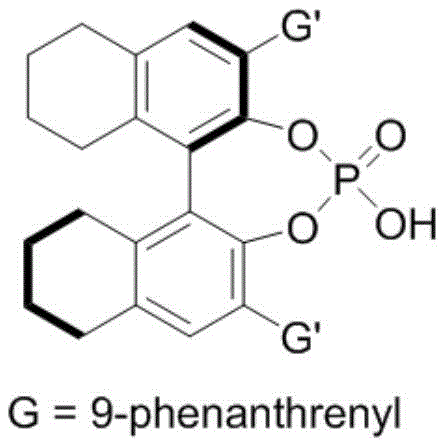
This rigorous control over the reaction pathway is the key to achieving the reported enantiomeric excess (ee) values, which often exceed 90% across a wide range of substrates. From an impurity control perspective, this high stereoselectivity is invaluable; it minimizes the formation of diastereomers and enantiomeric impurities that are notoriously difficult to separate. By preventing the formation of these unwanted isomers at the source, the process significantly reduces the burden on purification units, leading to a cleaner final product profile that meets the stringent purity specifications required for pharmaceutical intermediates. The mechanism also tolerates a variety of electronic and steric environments on the indole and nitrone rings, allowing for the incorporation of halogens, alkyl groups, and heteroaryl moieties without compromising the reaction efficiency or selectivity.
How to Synthesize Chiral Indoxazinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction parameters defined in the patent examples to ensure optimal results. The process begins with the preparation of the reaction mixture, where 2-indolylmethanol and nitrone are combined in toluene, typically with a molar ratio favoring the indole component (1:1 to 2:1) to drive the reaction to completion. Anhydrous sodium sulfate is added as a crucial additive, likely serving to sequester trace water that could deactivate the sensitive organocatalysts or hydrolyze the intermediates. The reaction is then initiated by the addition of the chiral phosphoric acid and HFIP, followed by stirring at a controlled temperature of 30°C. Monitoring is performed via Thin Layer Chromatography (TLC) to determine the endpoint, after which the mixture is filtered to remove the solid additive and concentrated. The final purification is achieved through silica gel column chromatography using a petroleum ether and dichloromethane eluent system. For a detailed, step-by-step standard operating procedure based on the optimized examples, please refer to the guide below.
- Prepare the reaction mixture by combining 2-indolylmethanol and nitrone in toluene solvent with a molar ratio between 1: 1 and 2:1, adding anhydrous sodium sulfate as an additive.
- Introduce the chiral phosphoric acid catalyst and hexafluoroisopropanol co-catalyst to the mixture and stir at a controlled temperature of 20-30°C.
- Monitor reaction progress via TLC until completion, then filter to remove solids, concentrate the filtrate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology offers compelling strategic benefits that extend far beyond simple chemical transformation. The shift from traditional metal-catalyzed or harsh condition syntheses to this mild organocatalytic protocol introduces significant opportunities for cost reduction in pharmaceutical intermediate manufacturing by simplifying the entire production lifecycle. The elimination of expensive transition metal catalysts removes the necessity for costly heavy metal scavenging and removal steps, which are often regulatory bottlenecks in API production. Furthermore, the use of common, commodity-grade solvents like toluene and the ability to run reactions at near-ambient temperatures drastically reduce energy consumption and solvent recovery costs. This efficiency translates directly into a more competitive pricing structure for the final intermediates, allowing downstream partners to optimize their own bill of materials.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the high atom economy and the avoidance of precious metal catalysts. By utilizing organocatalysts that are effective at low loadings, the raw material costs are significantly minimized compared to palladium or rhodium-based alternatives. Additionally, the high yields and selectivity mean that less starting material is wasted, and the downstream purification burden is lighter, reducing the consumption of silica gel and eluents. These factors combine to create a leaner manufacturing process that inherently lowers the cost of goods sold without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commercially available starting materials such as substituted indoles and nitrones, which are less susceptible to geopolitical supply shocks than specialized organometallic reagents. The mild reaction conditions (20-30°C) also imply that the process can be run in standard glass-lined or stainless steel reactors without the need for specialized cryogenic equipment or high-pressure vessels. This compatibility with existing infrastructure reduces the lead time for high-purity pharmaceutical intermediates by facilitating faster technology transfer and scale-up from lab to pilot to commercial production.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process aligns perfectly with green chemistry principles. The absence of toxic heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The straightforward work-up procedure, involving simple filtration and concentration, is easily adaptable to continuous flow chemistry or large batch processing, supporting the commercial scale-up of complex pharmaceutical intermediates. This scalability ensures a consistent and reliable supply of high-quality material, mitigating the risk of production delays that can impact downstream drug development timelines.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common questions regarding the synthesis and application of these chiral indoxazinones. The following insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, addressing key concerns about catalyst selection, substrate tolerance, and biological efficacy. Understanding these details is crucial for R&D directors planning new synthetic routes and for quality assurance teams establishing specification limits.
Q: What represents the key advantage of this synthesis method over traditional routes?
A: The primary advantage lies in the use of a chiral phosphoric acid and hexafluoroisopropanol dual catalytic system, which enables extremely high enantioselectivity under mild conditions (20-30°C), avoiding the harsh conditions and safety risks associated with conventional methods.
Q: What is the biological activity profile of the synthesized compounds?
A: The synthesized chiral indoxazinone compounds demonstrate significant cytotoxic activity against PC-3 human prostate cancer cells, with specific derivatives showing potent inhibitory effects, making them valuable candidates for novel antitumor drug research.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for industrial scale-up due to its mild reaction temperatures, use of common solvents like toluene, simple post-treatment procedures involving filtration and chromatography, and high atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral indoxazinone synthesis technology for the development of next-generation antitumor agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements via chiral HPLC, guaranteeing that every batch meets the highest standards required for pharmaceutical applications. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug discovery programs with reliable, high-quality building blocks.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your specific pipeline needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and purity requirements. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your timeline to market with our superior manufacturing capabilities and deep technical expertise.
