Advanced Organocatalytic Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
Advanced Organocatalytic Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for complex chiral scaffolds in modern drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN114524701B, which discloses a novel and efficient methodology for constructing N-N axis chiral pyrrole derivatives. These compounds are not merely structural curiosities; they represent a critical class of intermediates with demonstrated potent cytotoxic activity against QGP-1 pancreatic tumor cells. The patent outlines a robust organocatalytic strategy that bypasses the limitations of traditional transition metal catalysis, offering a pathway to high-purity pharmaceutical intermediates with exceptional stereocontrol. This technological advancement positions manufacturers as a reliable pharmaceutical intermediate supplier capable of delivering complex chiral building blocks that were previously difficult to access.
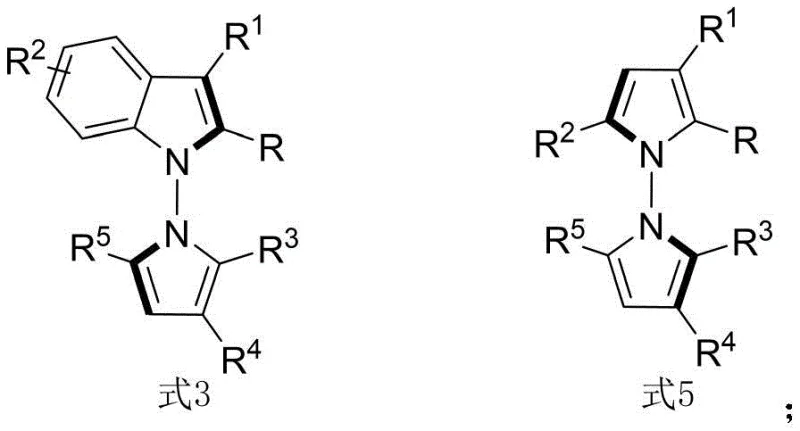
The core innovation lies in the utilization of chiral phosphoric acids to catalyze the condensation of indoleamines or pyrrole amines with 1,4-diketone derivatives. This approach achieves remarkable enantiomeric excess (ee) values, often exceeding 90%, under remarkably mild conditions. The structural diversity accessible through this method is vast, allowing for the modulation of substituents on both the pyrrole and indole rings to fine-tune biological activity. As the industry seeks cost reduction in API manufacturing, such atom-economical and selective processes become invaluable assets for supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral compounds has relied heavily on dynamic kinetic resolution or desymmetrization strategies, which often suffer from inherent theoretical yield limits of 50% or require expensive, specialized starting materials. Furthermore, many conventional routes depend on transition metal catalysts containing palladium, rhodium, or iridium. While effective, these metals pose significant challenges in a GMP environment due to strict regulatory limits on residual metal content in final drug substances. The removal of trace metals often necessitates additional purification steps, such as scavenging or recrystallization, which drastically increases processing time and operational costs. Additionally, traditional methods frequently require harsh reaction conditions, including high temperatures or cryogenic cooling, which complicate scale-up and increase energy consumption.
The Novel Approach
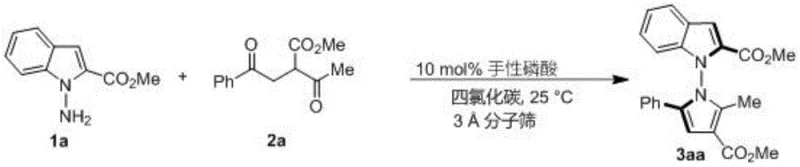
The methodology described in CN114524701B represents a paradigm shift by employing an organocatalytic in-situ ring formation strategy. This novel approach utilizes chiral phosphoric acids, specifically binaphthyl or spiro-cycle derived variants, to induce asymmetry without the use of metals. The reaction proceeds efficiently in carbon tetrachloride at ambient temperature (25°C), utilizing 3A molecular sieves to drive the equilibrium forward by removing water. This mild protocol not only preserves sensitive functional groups but also simplifies the downstream processing significantly. By achieving high yields and stereoselectivity in a single step from readily available 1,4-diketones and amines, this method offers a streamlined solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthesis hinges on the precise dual-activation mode of the chiral phosphoric acid catalyst. The catalyst acts as a Brønsted acid, protonating the carbonyl oxygen of the 1,4-diketone derivative to enhance its electrophilicity, while simultaneously forming hydrogen bonds with the nucleophilic amine substrate. This bifunctional activation organizes the transition state within a well-defined chiral pocket created by the bulky substituents on the catalyst backbone, such as the 2,4,6-trimethylphenyl groups found in the optimal catalyst 6j. This rigid environment forces the reaction to proceed through a specific trajectory, effectively discriminating between the pro-chiral faces of the intermediate and ensuring the formation of a single atropisomer with high fidelity.
From an impurity control perspective, the mildness of the reaction conditions is paramount. High temperatures often promote racemization of axially chiral bonds or lead to polymerization side reactions. By maintaining the reaction at 25°C, the thermal energy is insufficient to overcome the rotational barrier of the formed N-N axis once established, effectively locking in the chirality. Furthermore, the use of molecular sieves prevents the hydrolysis of the product or the starting materials, ensuring a clean reaction profile. This mechanistic understanding allows for the rational design of substrate scopes, confirming that electron-withdrawing or electron-donating groups on the aromatic rings do not significantly disrupt the catalyst-substrate interaction, thus broadening the applicability of this synthetic platform.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
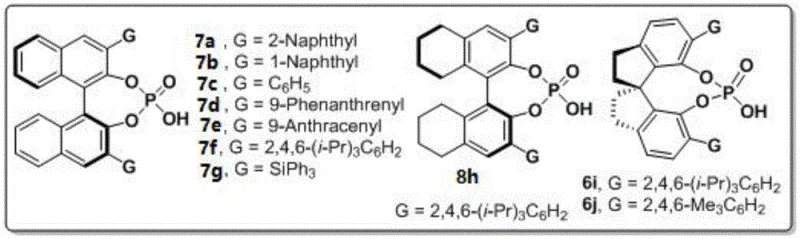
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and environmental controls to maximize yield and enantioselectivity. The process is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes or specialized high-pressure equipment. The key to success lies in the quality of the chiral catalyst and the efficiency of water removal during the reaction. Detailed standard operating procedures regarding reagent addition rates and stirring speeds are critical for reproducibility. For a comprehensive guide on the exact experimental parameters, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining indoleamine or pyrrole amine with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of chiral phosphoric acid catalyst (preferably compound 6j).
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology translates into tangible strategic benefits beyond mere chemical elegance. The elimination of precious metal catalysts removes a major cost driver and supply risk associated with fluctuating prices of metals like palladium or rhodium. Moreover, the simplified purification process reduces the consumption of solvents and silica gel, directly lowering the cost of goods sold (COGS). The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term supply contracts with pharmaceutical clients.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with organocatalysts significantly lowers raw material costs. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, and the simplified workup minimizes waste disposal costs associated with heavy metal contamination. This holistic reduction in operational overhead contributes to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted indoleamines and 1,4-diketones, are commercially available or easily synthesized from bulk chemicals. This abundance ensures a stable supply chain不受 geopolitical constraints often associated with rare earth metals. The high yield and selectivity of the process also mean less raw material is required to produce the same amount of active intermediate, further securing supply continuity.
- Scalability and Environmental Compliance: The reaction operates at ambient temperature and pressure, making it inherently safer and easier to scale from gram to ton quantities without complex engineering controls. The absence of toxic heavy metals simplifies environmental compliance and wastewater treatment, aligning with green chemistry principles and reducing the regulatory burden on manufacturing facilities.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for their specific drug pipelines. The following questions address common inquiries regarding the scope, safety, and scalability of the technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this synthesis method over traditional transition metal catalysis?
A: This method utilizes chiral phosphoric acid organocatalysis, which eliminates the need for toxic heavy metals, thereby simplifying purification processes and reducing environmental impact while maintaining high enantioselectivity.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological assays indicate that these derivatives possess strong cytotoxic activity against QGP-1 pancreatic tumor cells, suggesting significant potential for oncology drug development.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (25°C) with readily available substrates and simple workup procedures, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN114524701B for developing next-generation anticancer agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle organocatalytic processes with the precision required to meet stringent purity specifications. Our rigorous QC labs ensure that every batch of N-N axis chiral pyrrole derivatives delivered meets the highest standards of enantiomeric purity and chemical integrity, ready for immediate integration into your drug discovery or development programs.
We invite you to leverage our technical expertise to accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive your innovative therapies to the market faster.
