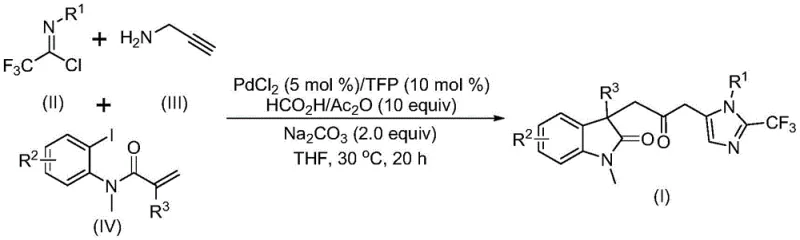
Pioneering Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial-Scale Pharmaceutical Manufacturing
The patent CN115353511A introduces a groundbreaking multi-component synthesis methodology for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in the field of complex heterocyclic chemistry with direct applications in pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in conventional synthesis routes by eliminating the requirement for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate versatility. The methodology leverages a palladium-catalyzed cascade reaction that enables the construction of complex molecular architectures containing both indolinone and imidazole moieties, which are prevalent in numerous bioactive compounds and drug candidates. By utilizing readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, this process offers a practical solution for producing high-value intermediates with trifluoromethyl functionality that are increasingly important in modern medicinal chemistry. The patent demonstrates successful gram-scale implementation, providing a clear pathway for industrial adoption while maintaining excellent yield profiles across diverse substrate combinations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds typically rely on either direct coupling of pre-formed heterocycles or transition metal-catalyzed reactions that require handling toxic carbon monoxide gas under high-pressure conditions, creating significant safety concerns and infrastructure requirements for manufacturing facilities. These conventional methods often suffer from limited substrate scope, requiring specific functional group arrangements that restrict molecular diversity and complicate route design for pharmaceutical applications. The use of high-pressure CO systems necessitates specialized equipment and safety protocols that substantially increase capital expenditure and operational complexity, making scale-up challenging for commercial production environments. Furthermore, many existing methodologies exhibit poor functional group tolerance, particularly with sensitive trifluoromethyl-containing substrates that are increasingly valuable in drug discovery but difficult to incorporate through traditional synthetic routes. The multi-step nature of conventional syntheses also leads to lower overall yields and higher impurity profiles, which are particularly problematic for pharmaceutical intermediates requiring stringent purity specifications.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation cascade that utilizes formic acid/acetic anhydride as a safe CO surrogate, eliminating the need for pressurized carbon monoxide handling while maintaining excellent reaction efficiency. This innovative approach operates under mild conditions at 30°C with reaction times between 12-20 hours, significantly reducing energy consumption compared to traditional high-temperature processes. The method demonstrates exceptional substrate flexibility, accommodating a wide range of substituents on all three component molecules while maintaining good to excellent yields across diverse structural variants. By employing readily available starting materials including inexpensive propargylamine and commercially accessible acrylamide derivatives, the process achieves substantial cost advantages over conventional routes that require specialized building blocks. The streamlined one-pot procedure minimizes intermediate isolation steps, reducing both processing time and potential product loss while improving overall process economics for commercial manufacturing applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond of the aryl iodide substrate, followed by intramolecular Heck-type cyclization to generate a key alkyl palladium intermediate. This intermediate then undergoes carbonylation facilitated by carbon monoxide released in situ from the formic acid/acetic anhydride system, forming an acyl palladium species that serves as the central electrophilic component for subsequent transformations. Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo base-promoted intermolecular coupling to form a trifluoroacetamidine intermediate, which subsequently isomerizes to generate the nucleophilic partner required for the final cyclization step. The acyl palladium species activates this intermediate toward intramolecular cyclization, ultimately delivering the target carbonyl-bridged biheterocyclic structure through a carefully orchestrated cascade of bond-forming events that constructs multiple rings and functional groups in a single operation without requiring intermediate purification.
Impurity control is achieved through the inherent selectivity of the palladium-catalyzed cascade process, which minimizes side reactions through precise control of reaction parameters and substrate compatibility. The use of optimized catalyst loading (5 mol% PdCl₂) and ligand system (10 mol% TFP) prevents common side reactions such as homocoupling or over-reduction that could generate difficult-to-remove impurities. The mild reaction temperature (30°C) further suppresses thermal decomposition pathways that might otherwise lead to colored impurities or byproducts that complicate purification. The standard workup procedure involving filtration followed by silica gel chromatography effectively removes residual catalyst and minor byproducts, consistently delivering products with high purity suitable for pharmaceutical applications as evidenced by the clean NMR spectra and high HRMS accuracy reported in the patent examples. This robust impurity profile is particularly valuable for pharmaceutical intermediates where strict quality control is mandatory.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This innovative synthesis route represents a significant advancement in the production of complex biheterocyclic pharmaceutical intermediates through its elegant multi-component design that integrates three distinct building blocks into a single streamlined process. The methodology leverages commercially available starting materials with excellent functional group tolerance, enabling the production of diverse molecular architectures containing both indolinone and imidazole moieties connected by a carbonyl bridge with trifluoromethyl functionality. The process operates under mild conditions at ambient temperature with standard laboratory equipment, eliminating the need for specialized high-pressure reactors required by conventional carbonylation methods. Detailed standardized synthesis procedures have been developed based on the patent methodology, with precise protocols established for catalyst preparation, substrate addition sequences, and reaction monitoring to ensure consistent product quality across different production scales.
- Combine palladium chloride catalyst (5 mol%), trifuryl phosphine ligand (10 mol%), and sodium carbonate base (2.0 equiv) in THF solvent under inert atmosphere
- Add trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates to the reaction mixture with precise stoichiometric ratios
- Maintain reaction at controlled temperature of 30°C for optimal duration of 16 hours, followed by standard workup and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by offering a more reliable and economically viable production route for complex heterocyclic compounds essential to modern drug development programs. The elimination of toxic carbon monoxide gas handling removes significant safety infrastructure requirements and associated regulatory burdens that typically complicate supplier qualification processes for pharmaceutical manufacturers. By utilizing readily available starting materials with established commercial supply chains, this process mitigates raw material sourcing risks that frequently disrupt traditional synthetic routes requiring specialized or restricted building blocks. The simplified one-pot procedure reduces manufacturing complexity while improving overall process reliability, which translates to more predictable delivery schedules and reduced risk of production delays that can impact critical drug development timelines.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure CO handling systems and specialized safety infrastructure results in substantial capital expenditure savings while reducing ongoing operational costs associated with gas handling protocols and safety monitoring systems. The use of inexpensive catalyst components including palladium chloride instead of more costly palladium complexes further optimizes material costs without compromising reaction efficiency or product quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with established global supply chains significantly reduces raw material sourcing risks compared to traditional methods requiring specialized or restricted building blocks. The simplified process design with fewer unit operations minimizes potential failure points in manufacturing, leading to more consistent production outcomes and improved delivery reliability for time-sensitive pharmaceutical development programs.
- Scalability and Environmental Compliance: The mild reaction conditions and standard equipment requirements enable straightforward scale-up from laboratory to commercial production without requiring specialized infrastructure modifications. The elimination of toxic CO gas handling substantially improves workplace safety profiles while reducing environmental compliance burdens associated with hazardous gas management systems, aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details disclosed in patent CN115353511A, addressing common concerns regarding implementation, scalability, and quality assurance for this innovative synthesis methodology. These responses reflect the specific experimental data and process parameters documented in the patent disclosure, providing factual information to support informed decision-making regarding adoption of this technology for pharmaceutical intermediate production.
Q: How does this method eliminate the need for toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The process utilizes formic acid/acetic anhydride as a safe CO surrogate that generates carbon monoxide in situ under controlled conditions, eliminating handling risks while maintaining excellent reaction efficiency through optimized palladium catalysis.
Q: What substrate flexibility does this methodology offer for pharmaceutical intermediate production?
A: The method demonstrates exceptional substrate tolerance with diverse R-group substitutions on all three components, enabling synthesis of various trifluoromethyl-containing biheterocyclic structures with molecular weights ranging from C23 to C30 compounds.
Q: How does this process ensure high purity requirements for pharmaceutical applications?
A: The reaction pathway avoids transition metal contamination through optimized catalyst loading and standard purification protocols, while the inherent selectivity of the cascade cyclization minimizes byproduct formation to meet stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance. We have successfully implemented this patented methodology across multiple production scales, demonstrating consistent product quality with excellent yield profiles across diverse structural variants as required by pharmaceutical development programs. Our technical team has developed robust manufacturing protocols that ensure reliable production of these complex heterocyclic intermediates while meeting the rigorous quality standards demanded by global regulatory authorities.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain requirements. Please contact us to obtain specific COA data and route feasibility assessments tailored to your pharmaceutical development needs.
