Advanced Palladium-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
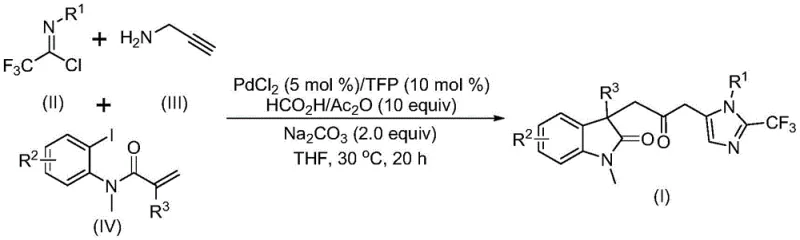
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting indolinone-imidazole derivatives which are prevalent in bioactive molecules. This technology represents a significant leap forward by replacing hazardous gaseous carbon monoxide with a safe, liquid-phase CO surrogate system. For R&D directors and process chemists, this offers a pathway to access diverse chemical space with improved safety profiles and operational simplicity. The core innovation lies in the seamless integration of a palladium-catalyzed carbonylation cascade that constructs multiple bonds in a single pot, drastically reducing step counts compared to linear synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of carbonyl-bridged biheterocycles has relied heavily on direct coupling reactions or oxidative cyclizations that often suffer from poor atom economy and harsh reaction conditions. A major bottleneck in conventional carbonylation strategies is the absolute requirement for exogenous carbon monoxide gas, which poses severe safety hazards due to its toxicity and flammability. Handling high-pressure CO gas necessitates specialized equipment, rigorous safety protocols, and expensive containment infrastructure, all of which drive up the capital expenditure for manufacturing facilities. Furthermore, traditional methods often exhibit limited substrate scope, struggling to tolerate sensitive functional groups like halogens or nitro groups without side reactions, thereby restricting the diversity of accessible analogs for drug discovery programs.
The Novel Approach
The methodology disclosed in CN115353511A circumvents these critical limitations by employing a transition metal palladium-catalyzed cascade reaction that operates under remarkably mild conditions. Instead of toxic CO gas, the process utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in situ, effectively acting as a safe and controllable CO source. This shift allows the reaction to proceed at a low temperature of 30°C in common organic solvents like tetrahydrofuran, eliminating the need for high-pressure reactors. The multi-component nature of the reaction, involving trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, enables the simultaneous formation of C-C, C-N, and C-O bonds, streamlining the synthesis of complex architectures from cheap and readily available starting materials.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing yield and purity in a commercial setting. The reaction likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction that forms the indolinone core, creating a divalent alkyl-palladium species. Subsequently, the in situ generated carbon monoxide inserts into the palladium-carbon bond to form an acyl-palladium intermediate, a key step that installs the crucial carbonyl bridge. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate, which undergoes isomerization. The final cyclization is driven by the activation of this amidine by the acyl-palladium species, closing the imidazole ring and releasing the final carbonyl-bridged biheterocyclic product while regenerating the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages. The use of mild temperatures (30°C) minimizes thermal degradation pathways and polymerization side reactions that are common in high-temperature Heck or carbonylation processes. The specific ligand system, utilizing trifurylphosphine (TFP) alongside palladium chloride, stabilizes the active catalytic species and prevents the formation of palladium black, ensuring consistent turnover numbers. Moreover, the orthogonal reactivity of the three components ensures that intermolecular oligomerization is suppressed in favor of the desired intramolecular cyclization. This high selectivity translates directly to a cleaner crude reaction profile, simplifying downstream purification and reducing the burden on quality control laboratories to identify and quantify trace impurities.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction parameters is essential. The patent outlines a standardized protocol where palladium chloride and trifurylphosphine are combined with sodium carbonate and the CO-generating mixture in an organic solvent. The substrates—trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative—are then introduced to initiate the cascade. The detailed standardized synthesis steps are provided in the guide below.
- Mix palladium chloride, trifurylphosphine, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel under stirring.
- React the mixture at 30°C for 12 to 20 hours, then filter, purify via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational efficiencies and risk mitigation. The elimination of gaseous carbon monoxide removes a significant logistical hurdle, as there is no longer a need to manage the delivery, storage, and disposal of hazardous gas cylinders. This simplification of the raw material portfolio enhances supply chain resilience and reduces the regulatory compliance costs associated with handling toxic gases. Furthermore, the use of commercially available and inexpensive starting materials like propargylamine and acryloyl chloride derivatives ensures a stable and cost-effective supply base, shielding production schedules from volatility in specialty reagent markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the reduction in energy consumption. By operating at 30°C rather than requiring elevated temperatures or high pressures, the process significantly lowers utility costs associated with heating and pressurization. Additionally, the high atom economy of the multi-component approach means less waste is generated per kilogram of product, reducing the costs associated with waste treatment and disposal. The avoidance of expensive protecting group strategies, which are often needed in stepwise syntheses, further contributes to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against various functional groups ensures that supply disruptions due to batch failures are minimized. The broad substrate compatibility means that if a specific substituted acrylamide is unavailable, alternative analogs can often be substituted without re-optimizing the entire process, providing flexibility in sourcing. The scalability demonstrated in the patent, extending from milligram to gram scales without loss of efficiency, indicates a smooth path to ton-scale production, ensuring that commercial demands can be met consistently without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: From an environmental standpoint, replacing toxic CO gas with formic acid derivatives aligns with green chemistry principles, facilitating easier permitting and environmental compliance. The reaction produces fewer hazardous byproducts, and the use of common solvents like THF allows for established recycling protocols. This environmental friendliness not only reduces the ecological footprint of the manufacturing process but also enhances the marketability of the final intermediates to downstream pharmaceutical clients who are increasingly prioritizing sustainable supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific application needs.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly reducing safety risks and regulatory burdens.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds efficiently under mild conditions, specifically at 30°C in tetrahydrofuran (THF) solvent, using a PdCl2/TFP catalyst system over a period of 12 to 20 hours.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with high substrate compatibility and simple post-treatment procedures, making it viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN115353511A can be effectively realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbonyl-bridged biheterocyclic intermediates meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that your drug development pipeline remains uninterrupted by supply inconsistencies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall time to market.
