Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective synthetic routes for complex heterocyclic scaffolds that serve as the backbone of modern therapeutics. A recent technological breakthrough documented in patent CN113105402B introduces a highly efficient preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value active pharmaceutical ingredients (APIs). This innovation addresses critical bottlenecks in traditional heterocycle synthesis by eliminating the reliance on toxic heavy metal catalysts and严苛 reaction conditions. The 1,2,4-triazole ring system is a privileged structure found in numerous blockbuster drugs, including antiviral agents like Maraviroc and antidiabetic medications such as Sitagliptin, as illustrated in the structural diversity of current pharmaceuticals. Furthermore, the strategic incorporation of a trifluoromethyl group into these heterocycles is known to drastically enhance metabolic stability, lipophilicity, and bioavailability, making this specific synthetic methodology a valuable asset for medicinal chemists aiming to optimize lead compounds.
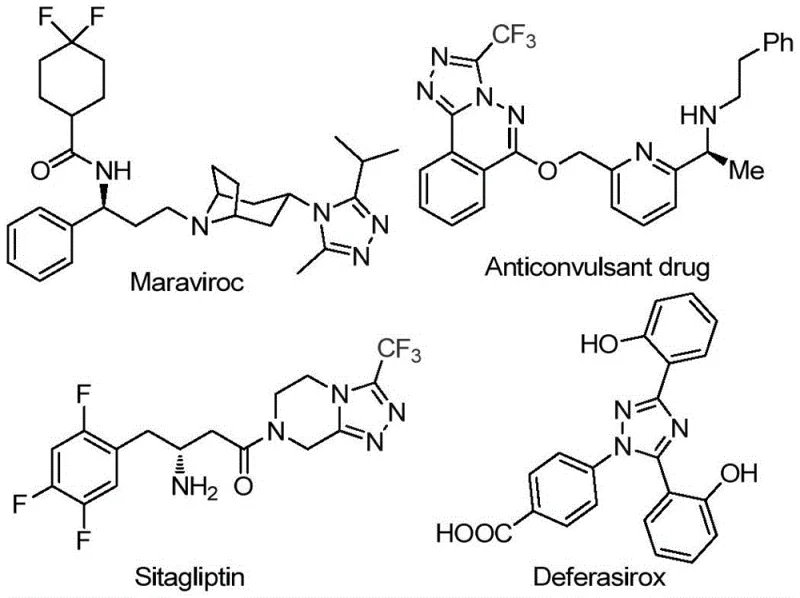
For procurement managers and supply chain directors, the implications of this patent extend far beyond the laboratory bench. The disclosed method utilizes inexpensive, commodity-grade starting materials such as aryl ethyl ketones and elemental iodine, which are widely available in the global chemical market. By shifting away from precious metal catalysis (such as palladium or copper systems) towards a non-metallic iodine-promoted system, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing. This transition not only lowers the raw material expenditure but also simplifies the downstream processing required to remove trace metal contaminants, a rigorous requirement for FDA and EMA compliance. Consequently, this technology represents a paradigm shift towards greener, more economical, and supply-chain-resilient production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, particularly those bearing both electron-withdrawing trifluoromethyl groups and acyl functionalities, has been fraught with synthetic challenges. Conventional methodologies often rely on multi-step sequences involving harsh reagents, strict anhydrous and oxygen-free conditions, and the use of expensive transition metal catalysts. These traditional routes frequently suffer from poor atom economy and generate significant amounts of hazardous waste, complicating environmental compliance and increasing the overall carbon footprint of the manufacturing process. Moreover, the presence of heavy metal residues in the final product necessitates complex and costly purification steps, such as scavenging treatments or repeated recrystallizations, which inevitably reduce the overall yield and extend the production lead time. For large-scale operations, the sensitivity of these reactions to moisture and oxygen requires specialized equipment and inert atmosphere handling, further inflating capital expenditure and operational complexity.
The Novel Approach
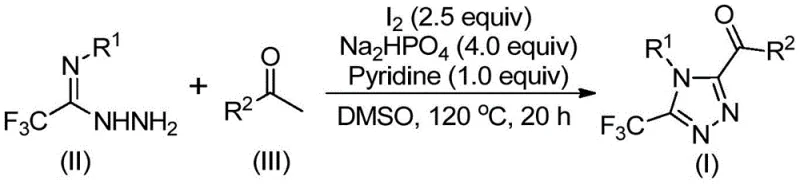
In stark contrast, the novel approach detailed in patent CN113105402B offers a streamlined, one-pot strategy that leverages the oxidative power of elemental iodine in dimethyl sulfoxide (DMSO). This method ingeniously combines an iodination and Kornblum oxidation sequence with a subsequent tandem cyclization, allowing for the direct assembly of the target triazole scaffold from simple precursors. The reaction proceeds efficiently under air-tolerant conditions, completely removing the need for inert gas protection or rigorous drying of solvents, which dramatically simplifies the operational protocol. By utilizing cheap and readily available aryl ethyl ketones and trifluoroethylimide hydrazides, the process ensures a stable and cost-effective supply of raw materials. The absence of heavy metals not only aligns with green chemistry principles but also facilitates a much cleaner workup procedure, typically involving simple filtration and standard column chromatography, thereby enhancing the throughput and scalability of the synthesis for industrial applications.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this innovative synthesis lies in a sophisticated cascade reaction mechanism initiated by the interaction between elemental iodine and DMSO. Initially, the aryl ethyl ketone undergoes an iodination followed by a Kornblum oxidation to generate an reactive alpha-dicarbonyl or alpha-iodo ketone intermediate in situ. This activated species then undergoes a condensation reaction with the trifluoroethylimide hydrazide, forming a hydrazone intermediate through a dehydration process. The presence of sodium dihydrogen phosphate and pyridine acts as a crucial buffer and base system, facilitating the subsequent intramolecular cyclization. Under the thermal conditions of 110-130°C, the hydrazone intermediate cyclizes to form the 1,2,4-triazole ring, with the iodine species potentially acting as a Lewis acid promoter to accelerate the ring closure. This mechanistic pathway elegantly bypasses the need for pre-functionalized building blocks, allowing for the direct installation of the trifluoromethyl and acyl groups in a single operational sequence.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high product purity. The use of DMSO as both solvent and oxidant ensures high conversion rates of the starting ketone, minimizing the accumulation of unreacted starting materials that are often difficult to separate. Furthermore, the mild basic conditions provided by the phosphate/pyridine system help to suppress side reactions such as over-oxidation or polymerization of the sensitive hydrazide species. The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the aromatic rings, including halogens, alkoxy groups, and alkyl chains, without significant degradation. This robustness translates to a cleaner crude reaction profile, reducing the burden on purification teams and ensuring that the final API intermediate meets stringent quality specifications with minimal effort. The ability to tune the electronic properties of the substrate without compromising the reaction efficiency makes this method highly versatile for generating diverse libraries of triazole derivatives.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
The practical implementation of this synthesis route is designed for ease of execution, making it accessible for both laboratory discovery and pilot plant operations. The process begins by combining the aryl ethyl ketone and a stoichiometric amount of elemental iodine in DMSO, followed by heating to initiate the oxidation phase. Once this initial transformation is complete, the remaining reagents, including the hydrazide source, base, and additional iodine, are introduced to drive the cyclization to completion. The detailed standardized synthesis steps, including precise molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducible results and optimal yields.
- Mix aryl ethyl ketone and elemental iodine in DMSO solvent and heat to 90-110°C for 4-6 hours to initiate oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this patented methodology presents a compelling value proposition that addresses several persistent pain points in fine chemical manufacturing. The shift from precious metal catalysis to a base-metal-free iodine system fundamentally alters the cost structure of producing these complex heterocycles. By eliminating the need for expensive catalysts like palladium or rhodium, and removing the associated ligand costs, the direct material cost of the synthesis is significantly reduced. Additionally, the simplified purification process reduces solvent consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing operation. The use of commodity chemicals ensures that supply chain disruptions related to specialized reagents are minimized, providing a stable foundation for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of toxic heavy metal catalysts removes the necessity for expensive metal scavenging resins and complex purification protocols, leading to substantial operational savings. The reliance on inexpensive iodine and DMSO, coupled with high atom economy, ensures that the cost per kilogram of the final intermediate is optimized for commercial viability. Furthermore, the energy efficiency of the process, which operates at moderate temperatures without cryogenic cooling or high-pressure equipment, contributes to lower utility costs across the production lifecycle.
- Enhanced Supply Chain Reliability: The starting materials, specifically aryl ethyl ketones and trifluoroethylimide hydrazides, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to delays caused by environmental factors or equipment limitations associated with inert atmosphere processing. This reliability allows for more accurate forecasting and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The method has been demonstrated to scale effectively from gram-level experiments to larger batches without loss of efficiency, indicating strong potential for multi-ton annual production. The absence of heavy metals simplifies regulatory compliance regarding residual metal limits in drug substances, accelerating the approval process for new drug applications. Moreover, the reduced generation of hazardous waste aligns with increasingly strict environmental regulations, positioning manufacturers as responsible corporate citizens while avoiding potential fines or remediation costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's capabilities and limitations for potential adopters.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying purification and reducing heavy metal residue risks in the final API.
Q: What are the key advantages of this route for large-scale manufacturing?
A: The process operates under air-tolerant conditions without the need for strict anhydrous or oxygen-free environments, uses cheap and readily available starting materials like aryl ethyl ketones, and allows for easy scale-up from gram to industrial levels.
Q: Can this method introduce both trifluoromethyl and acyl groups simultaneously?
A: Yes, this specific methodology is designed to efficiently construct the 1,2,4-triazole core while simultaneously incorporating both a trifluoromethyl group at the 3-position and an acyl group at the 5-position, which is often challenging with conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging innovations such as the metal-free triazole synthesis described in CN113105402B, we can offer our partners superior quality products with improved cost structures and faster turnaround times.
We invite you to collaborate with us to explore how this cutting-edge methodology can be integrated into your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and accelerate your drug development timeline.
