Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly those incorporating trifluoromethyl groups which enhance metabolic stability and lipophilicity. A groundbreaking preparation method disclosed in patent CN113105402B offers a transformative approach to synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds. This technology addresses critical bottlenecks in traditional heterocycle synthesis by utilizing a metal-free, iodine-promoted cascade reaction that operates under relatively mild conditions without the need for stringent anhydrous or oxygen-free environments. The significance of this scaffold cannot be overstated, as evidenced by its presence in major therapeutic agents such as Maraviroc, Sitagliptin, and Deferasirox, where the triazole core plays a pivotal role in biological activity.
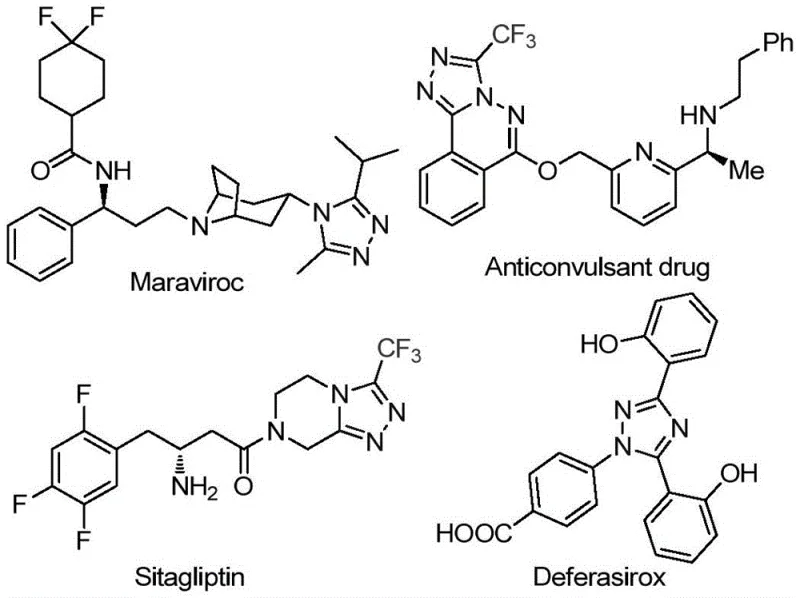
For R&D directors and process chemists, the ability to access these complex molecular architectures efficiently is paramount. The patented methodology leverages readily available aryl ethyl ketones and trifluoroethylimide hydrazides as starting materials, streamlining the supply chain for high-purity pharmaceutical intermediates. By avoiding toxic heavy metal catalysts, this route not only reduces environmental impact but also significantly lowers the burden on quality control laboratories regarding residual metal testing, a common pain point in API manufacturing. The reaction proceeds through a tandem sequence involving Kornblum oxidation followed by intramolecular cyclization, demonstrating exceptional functional group tolerance and operational simplicity that is highly attractive for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings has relied heavily on transition metal catalysis or harsh reaction conditions that pose significant challenges for industrial application. Traditional pathways often necessitate the use of expensive copper, palladium, or rhodium catalysts, which not only inflate raw material costs but also introduce severe complications in downstream processing due to the difficulty of removing trace metal residues to meet stringent regulatory limits. Furthermore, many existing protocols require strictly anhydrous and oxygen-free conditions, demanding specialized equipment and inert atmosphere handling that drastically increases operational expenditures and limits the feasibility of large-scale production. The reliance on pre-functionalized starting materials, such as acid chlorides or hydrazonoyl halides, further complicates the supply chain, introducing additional synthetic steps and safety hazards associated with hazardous reagents.
The Novel Approach
In stark contrast, the innovation presented in patent CN113105402B utilizes a simple yet powerful iodine-promoted system in dimethyl sulfoxide (DMSO) to achieve the desired transformation with remarkable efficiency. This novel approach eliminates the need for transition metals entirely, replacing them with inexpensive elemental iodine which acts as both an oxidant and a promoter for the cyclization event. The reaction tolerates ambient moisture and oxygen, removing the necessity for glovebox techniques or rigorous solvent drying, thereby simplifying the engineering requirements for reactor setup. By employing a one-pot strategy that combines oxidation and cyclization, the method reduces the number of isolation steps, minimizes solvent waste, and improves the overall atom economy of the process. This represents a paradigm shift towards greener, more cost-effective manufacturing of high-value heterocyclic intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation chemistry that ensures high selectivity for the 3,4,5-trisubstituted product. The reaction initiates with the iodine-mediated Kornblum oxidation of the aryl ethyl ketone in DMSO, generating an reactive alpha-dicarbonyl or alpha-iodo ketone intermediate in situ. This activated species then undergoes nucleophilic attack by the trifluoroethylimide hydrazide, leading to the formation of a hydrazone intermediate through a dehydration condensation step. The presence of sodium dihydrogen phosphate and pyridine serves a dual purpose: buffering the acidic byproducts generated during oxidation and facilitating the deprotonation steps required for the subsequent ring closure.
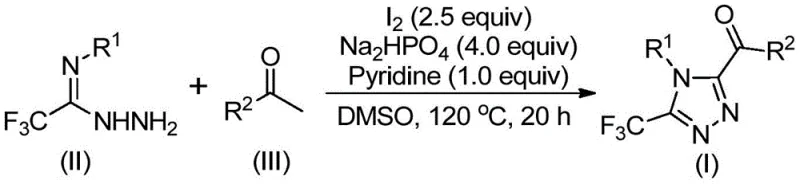
Following hydrazone formation, the system undergoes an intramolecular oxidative cyclization driven by the remaining iodine species and the thermal energy provided at elevated temperatures (110-130°C). This final step constructs the 1,2,4-triazole ring while simultaneously installing the trifluoromethyl group at the 3-position and the acyl group at the 5-position. The use of DMSO is critical not only as a solvent but as an oxygen donor in the initial oxidation phase. The careful optimization of reagent stoichiometry, specifically the molar ratio of hydrazide to ketone to base to iodine (1:2:4:1:2.5), ensures that side reactions such as over-oxidation or polymerization are suppressed, resulting in a clean impurity profile that facilitates easier purification via standard column chromatography or crystallization techniques.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis requires precise control over temperature gradients and reagent addition sequences to maximize yield and purity. The process begins by dissolving the aryl ethyl ketone and a portion of elemental iodine in DMSO, followed by heating to initiate the oxidation phase. Once the intermediate is formed, the second stage involves the introduction of the hydrazide component along with the phosphate buffer and pyridine base. Maintaining the reaction temperature between 110°C and 130°C for an extended period of 12 to 20 hours is crucial for driving the cyclization to completion. Detailed standardized operating procedures for this specific transformation are outlined below to ensure reproducibility across different manufacturing sites.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial strategic advantages by decoupling production from volatile precious metal markets and complex logistical constraints. The shift away from heavy metal catalysts directly translates to a more resilient supply chain, as elemental iodine and commodity solvents like DMSO are globally available and subject to far less regulatory scrutiny than specialized organometallic reagents. This accessibility ensures consistent raw material availability, reducing the risk of production delays caused by supplier shortages or geopolitical instability affecting rare earth or precious metal exports. Additionally, the simplified workup procedure reduces the consumption of silica gel and eluents typically required for metal scavenging, leading to significant operational cost savings.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper removes a major cost driver from the bill of materials, while the avoidance of rigorous anhydrous conditions lowers energy consumption associated with solvent drying and inert gas purging. The use of cheap, bulk chemicals like aryl ethyl ketones and iodine further drives down the unit cost of the final intermediate, making it economically viable for high-volume API production. Furthermore, the reduced need for specialized metal-removal resins or extensive aqueous washes simplifies the waste treatment process, lowering environmental compliance costs.
- Enhanced Supply Chain Reliability: Because the starting materials are commodity chemicals with established global supply chains, manufacturers can secure long-term contracts with multiple vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process can be transferred between different manufacturing facilities with minimal re-validation, providing flexibility in production scheduling and capacity allocation. This reliability is critical for meeting the just-in-time delivery demands of downstream pharmaceutical clients who require uninterrupted supply of key intermediates for their own formulation lines.
- Scalability and Environmental Compliance: The method has been demonstrated to scale effectively from gram-level laboratory experiments to multi-kilogram pilot batches without loss of efficiency, indicating strong potential for ton-scale commercial production. The absence of toxic heavy metals simplifies the disposal of reaction waste, aligning with increasingly stringent environmental regulations regarding heavy metal discharge in chemical manufacturing zones. The use of DMSO, while requiring proper recovery systems, is a well-understood solvent in the industry with established recycling protocols, further enhancing the sustainability profile of the overall manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and scope analysis provided in the patent documentation. Understanding these nuances is essential for process engineers aiming to adapt this chemistry for specific target molecules within their pipeline. The answers reflect the practical realities of running this reaction in a GMP-compliant environment.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, eliminating the need for costly heavy metal catalysts and simplifying downstream purification.
Q: What are the optimal reaction conditions for scaling this process?
A: The process operates effectively in DMSO solvent with a two-stage heating profile: initial oxidation at 90-110°C followed by cyclization at 110-130°C, ensuring high conversion rates suitable for industrial scale-up.
Q: Is the method tolerant to various functional groups on the aryl rings?
A: Yes, the protocol demonstrates broad substrate scope, successfully accommodating substituents such as methyl, methoxy, chloro, and trifluoromethyl groups on both the hydrazide and ketone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole meets the exacting standards required by global regulatory authorities. Our commitment to quality assurance extends beyond mere compliance, focusing on delivering consistent performance that supports your clinical and commercial timelines.
We invite you to collaborate with us to leverage this innovative metal-free synthesis for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your overall manufacturing budget. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your path to market with reliable, cost-effective chemical solutions.
