Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in patent CN111423381A, the development of efficient routes to 2-trifluoromethyl substituted imidazole compounds addresses a critical bottleneck in the supply chain for high-value pharmaceutical intermediates. These structural motifs are pervasive in bioactive molecules, ranging from antihistamines like Alcaftadine to complex natural products such as Naamidine A, underscoring their versatility in drug design. 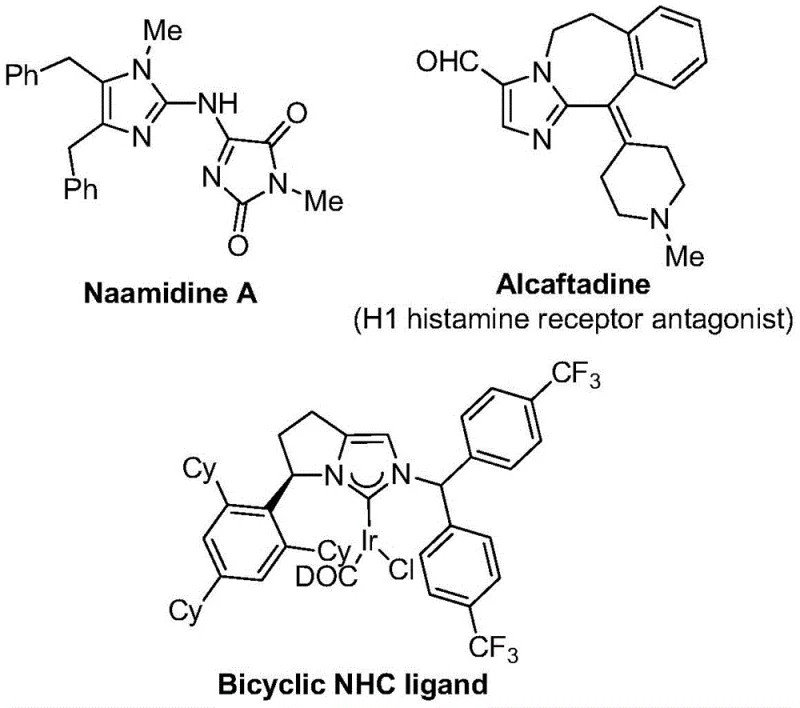 . The disclosed methodology leverages a transition metal palladium-catalyzed carbonylation cascade, offering a robust alternative to conventional synthetic strategies that often suffer from harsh conditions or limited substrate scope. For R&D directors and procurement managers alike, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex fluorinated building blocks.
. The disclosed methodology leverages a transition metal palladium-catalyzed carbonylation cascade, offering a robust alternative to conventional synthetic strategies that often suffer from harsh conditions or limited substrate scope. For R&D directors and procurement managers alike, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functionalities has relied heavily on the direct reaction of trifluoromethyl synthons with suitable substrates. While effective in specific contexts, traditional approaches frequently encounter significant hurdles regarding operational safety and economic feasibility. Many established protocols require the handling of hazardous reagents such as trifluorodiazoethane, which poses substantial explosion risks and necessitates specialized containment infrastructure. Furthermore, conventional methods often demand elevated temperatures and prolonged reaction times, leading to excessive energy consumption and potential degradation of sensitive functional groups. The reliance on pre-functionalized starting materials can also drive up raw material costs, creating a barrier to entry for cost-sensitive manufacturing projects. Additionally, the narrow substrate compatibility of older techniques often restricts the chemical diversity accessible to medicinal chemists, limiting the ability to rapidly iterate on lead optimization campaigns.
The Novel Approach
In stark contrast, the innovative process detailed in the patent utilizes a palladium-catalyzed carbonylation tandem reaction that operates under remarkably mild conditions, specifically at 30°C. This approach employs readily available and inexpensive starting materials, including trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts, effectively bypassing the need for dangerous diazo compounds. A key feature of this novel route is the in situ generation of carbon monoxide from a mixture of formic acid and acetic anhydride, eliminating the requirement for high-pressure CO gas cylinders and enhancing overall process safety. The reaction exhibits exceptional functional group tolerance, accommodating a wide array of substituents such as methyl, tert-butyl, halogens, and nitro groups on the aromatic rings. 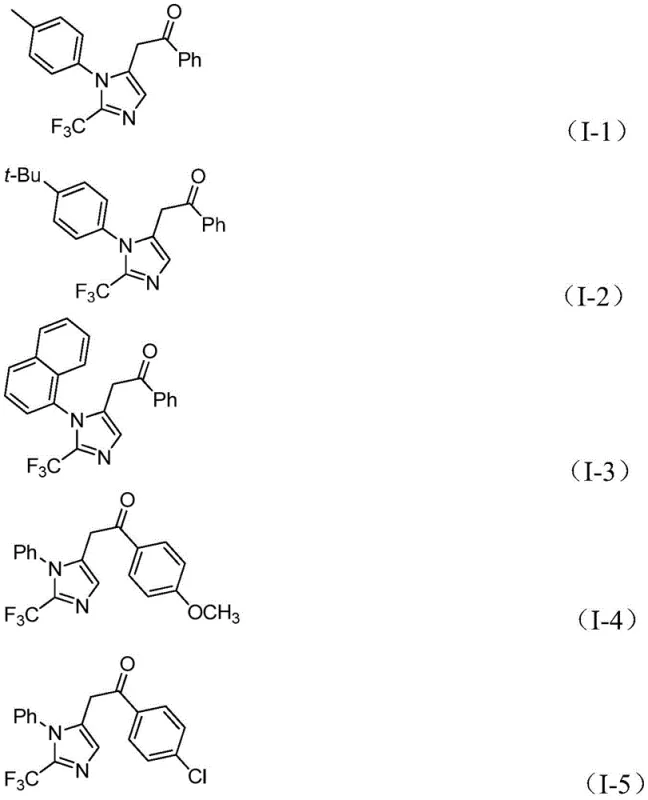 . This versatility allows for the rapid synthesis of diverse libraries of 1,5-disubstituted imidazoles, providing a powerful tool for structure-activity relationship studies while maintaining high reaction efficiency and yields.
. This versatility allows for the rapid synthesis of diverse libraries of 1,5-disubstituted imidazoles, providing a powerful tool for structure-activity relationship studies while maintaining high reaction efficiency and yields.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps orchestrated by the palladium catalyst. Initially, the reaction likely proceeds through a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to facilitate the palladium-catalyzed aminopalladation of the alkyne moiety. The resulting alkenyl palladium intermediate further isomerizes to a more stable alkyl palladium species, setting the stage for the critical carbonylation event. Crucially, the carbon monoxide required for this step is released in situ from the decomposition of the formic acid and acetic anhydride mixture, which then inserts into the palladium-carbon bond to form an acyl palladium intermediate. 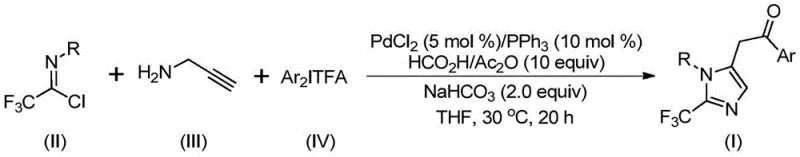 . The cycle concludes with the oxidative addition of the diaryliodonium salt to generate a tetravalent palladium species, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
. The cycle concludes with the oxidative addition of the diaryliodonium salt to generate a tetravalent palladium species, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in minimizing side reactions such as polymerization or decomposition of sensitive intermediates. The use of sodium bicarbonate as a base ensures a buffered environment that promotes the desired cyclization without inducing harsh hydrolysis of the imidoyl chloride or other electrophilic centers. Furthermore, the choice of tetrahydrofuran (THF) as the preferred solvent optimizes the solubility of all reactants, ensuring homogeneous reaction conditions that favor high conversion rates. The post-processing workflow is equally streamlined, involving simple filtration and silica gel treatment followed by column chromatography, which effectively removes palladium residues and inorganic salts. This rigorous control over reaction parameters and purification steps ensures that the final product meets stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of scale-up, making it highly attractive for process chemistry teams. The protocol involves charging a reaction vessel with palladium chloride, triphenylphosphine, sodium bicarbonate, and the formic acid/acetic anhydride mixture in an organic solvent. To this mixture, the trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt are added, and the system is stirred at 30°C for a duration of 16 to 24 hours. Upon completion, the reaction mixture is filtered to remove insoluble salts, and the filtrate is concentrated and purified via standard chromatographic techniques. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical novelty. The shift towards a safer, more efficient synthetic route directly translates into reduced operational risks and lower total cost of ownership for manufacturing facilities. By eliminating the need for specialized high-pressure equipment to handle carbon monoxide gas and avoiding explosive diazo reagents, companies can significantly lower their capital expenditure on safety infrastructure and insurance premiums. Moreover, the use of commodity chemicals like formic acid and acetic anhydride as CO surrogates ensures a stable and predictable supply chain, mitigating the risks associated with sourcing exotic or regulated reagents.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive catalysts and ligands, specifically palladium chloride and triphenylphosphine, which are widely available in the global market. The mild reaction conditions of 30°C drastically reduce energy consumption compared to traditional high-temperature protocols, leading to substantial utility savings over long production runs. Additionally, the high reaction efficiency and broad substrate compatibility minimize the formation of byproducts, thereby reducing the burden on waste treatment systems and lowering the cost of goods sold. The simplified post-processing workflow further contributes to cost optimization by shortening the overall production cycle time and reducing labor requirements.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as aromatic amines and propargylamine ensures a robust supply chain that is less susceptible to disruptions. Since the key building blocks are derived from abundant feedstocks, manufacturers can secure long-term contracts with multiple suppliers, guaranteeing continuity of supply even during market fluctuations. The scalability of the method, demonstrated from milligram to gram levels in the patent, suggests a clear pathway for commercial scale-up without the need for extensive process re-engineering. This reliability is crucial for meeting the just-in-time delivery demands of major pharmaceutical clients who require consistent quality and volume.
- Scalability and Environmental Compliance: From an environmental standpoint, this green chemistry approach aligns with increasingly stringent regulatory standards regarding waste generation and solvent usage. The in situ generation of carbon monoxide prevents the release of toxic gases into the atmosphere, enhancing workplace safety and environmental compliance. The use of THF as a solvent, while requiring proper recovery systems, is well-established in the industry, facilitating easy integration into existing manufacturing suites. The ability to produce diverse derivatives through simple substrate modification allows for a flexible manufacturing platform that can quickly adapt to changing market needs without significant retooling investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method operates at a mild temperature of 30°C, utilizes inexpensive starting materials like trifluoroethylimidoyl chloride, and avoids the use of toxic carbon monoxide gas by generating it in situ from formic acid and acetic anhydride.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and potentially larger scales due to its simple operation, high reaction efficiency, and excellent substrate compatibility.
Q: What types of substituents are tolerated in this reaction?
A: The reaction demonstrates broad functional group tolerance, accommodating various substituents on the aryl rings including methyl, tert-butyl, halogens (chloro, bromo), trifluoromethyl, and nitro groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our expertise in fluorine chemistry and transition metal catalysis positions us as a strategic partner capable of solving complex synthetic challenges.
We invite you to contact our technical procurement team to discuss how this advanced palladium-catalyzed technology can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic advantages of switching to this more efficient synthetic route. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your target molecules, allowing us to demonstrate our commitment to quality and innovation in the fine chemical sector.
