Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks efficient pathways to access privileged heterocyclic scaffolds, particularly quinolin-2(1H)-one derivatives, which serve as critical cores in numerous bioactive molecules. As disclosed in patent CN113045489A, a novel preparation method has been developed that leverages palladium-catalyzed aminocarbonylation to construct these valuable structures with high precision. This technology represents a significant leap forward in synthetic methodology, utilizing benzisoxazole not merely as a reactant but as a dual-purpose reagent providing both nitrogen and formyl equivalents. The versatility of this approach is underscored by its ability to generate a diverse library of derivatives, including those found in MAP Kinase inhibitors and HBV inhibitors, as illustrated in the structural diversity of known bioactive compounds. 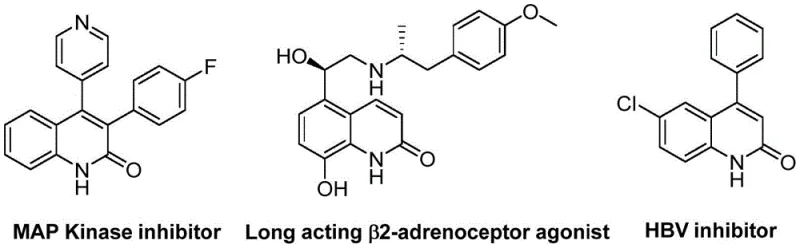 For procurement managers and R&D directors alike, this patent offers a compelling solution for the reliable production of high-purity pharmaceutical intermediates, addressing both cost concerns and supply chain stability through a streamlined catalytic cycle.
For procurement managers and R&D directors alike, this patent offers a compelling solution for the reliable production of high-purity pharmaceutical intermediates, addressing both cost concerns and supply chain stability through a streamlined catalytic cycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these traditional routes have served the industry for decades, they often suffer from significant drawbacks that hinder modern manufacturing efficiency. These conventional methods frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to poor functional group tolerance and the formation of complex impurity profiles. Furthermore, many transition-metal-catalyzed modifications previously employed often necessitate the use of hazardous carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized, expensive equipment for containment. The reliance on such dangerous reagents complicates the regulatory approval process and increases the operational overhead for chemical manufacturers, ultimately driving up the cost of goods sold for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN113045489A introduces a groundbreaking palladium-catalyzed aminocarbonylation strategy that fundamentally reshapes the synthetic landscape. This novel approach utilizes benzisoxazole and benzyl chloride compounds as readily available starting materials, reacting them in the presence of a palladium catalyst system and molybdenum carbonyl as a solid carbon monoxide source. 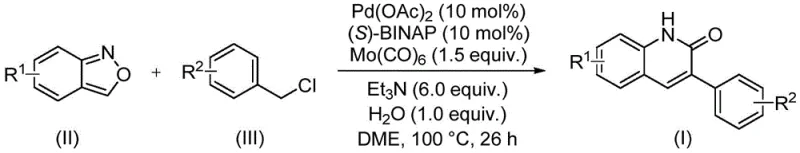 By employing molybdenum carbonyl, the process eliminates the need for handling gaseous CO, thereby drastically improving operational safety and simplifying the reactor setup. The reaction proceeds smoothly at 100°C in DME solvent with triethylamine and water as additives, demonstrating exceptional compatibility with a wide range of functional groups including halogens, alkoxy groups, and cyano groups. This method not only simplifies the operation but also widens the practicability of the synthesis, allowing for the rapid generation of diverse chemical libraries essential for drug discovery programs.
By employing molybdenum carbonyl, the process eliminates the need for handling gaseous CO, thereby drastically improving operational safety and simplifying the reactor setup. The reaction proceeds smoothly at 100°C in DME solvent with triethylamine and water as additives, demonstrating exceptional compatibility with a wide range of functional groups including halogens, alkoxy groups, and cyano groups. This method not only simplifies the operation but also widens the practicability of the synthesis, allowing for the rapid generation of diverse chemical libraries essential for drug discovery programs.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate catalytic cycle driven by the palladium/(S)-BINAP complex. The mechanism initiates with the oxidative addition of the benzyl chloride compound to the active Pd(0) species, generating an organopalladium intermediate. Subsequently, carbon monoxide, released in situ from the decomposition of molybdenum carbonyl, inserts into the palladium-carbon bond to form an acyl-palladium complex. A critical and unique feature of this reaction is the role of benzisoxazole; under the basic conditions provided by triethylamine and the influence of the metal center, the benzisoxazole ring undergoes cleavage. This cleavage releases the necessary nitrogen nucleophile and effectively provides the formyl component required to close the quinolinone ring. The presence of water in the reaction mixture is also pivotal, likely facilitating the hydrolysis steps or stabilizing intermediates during the ring-closing event. The use of the chiral ligand (S)-BINAP, although the final product is achiral in many cases, ensures high catalytic turnover and stability of the palladium species, preventing premature catalyst deactivation which is a common pitfall in carbonylation reactions.
From an impurity control perspective, this mechanism offers distinct advantages over traditional cyclization methods. The specificity of the palladium-catalyzed coupling minimizes side reactions such as homocoupling of the benzyl chloride or polymerization of the reactive intermediates. The mild conditions prevent the degradation of sensitive functional groups on the aromatic rings, such as the cyano or trifluoromethyl groups seen in substrates like those leading to compound I-3 or I-9. This high chemoselectivity results in a cleaner crude reaction mixture, which significantly reduces the burden on downstream purification processes. For R&D teams focused on impurity profiling, this means a more predictable and manageable impurity spectrum, facilitating faster regulatory filings. The ability to tolerate substituents at both the meta and para positions on the benzyl chloride ring, as well as various substitutions on the benzisoxazole moiety, confirms the robustness of the catalytic system against steric and electronic variations.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The patented procedure outlines a straightforward protocol for accessing these valuable heterocycles, emphasizing ease of execution and reproducibility. The process involves charging a sealed tube with the requisite amounts of palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative. The reaction is then heated to 100°C for approximately 26 hours, ensuring complete conversion of the starting materials into the desired 3-arylquinolin-2(1H)-one skeleton. Following the reaction, the workup is remarkably simple, involving filtration to remove insoluble metal residues and salts, followed by standard silica gel chromatography to isolate the pure product.
- Combine palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and stir for approximately 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards this palladium-catalyzed method addresses several critical pain points associated with the sourcing and manufacturing of complex heterocyclic intermediates. By utilizing benzisoxazole and benzyl chlorides, which are commodity chemicals available from multiple global suppliers, the risk of raw material shortage is significantly mitigated. This diversification of the supply base ensures continuity of supply even in volatile market conditions. Furthermore, the elimination of high-pressure carbon monoxide gas removes a major safety bottleneck, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized autoclaves designed for toxic gases. This flexibility translates directly into reduced capital expenditure and lower operational costs for manufacturing partners.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and widely available starting materials. Benzisoxazole acts as a dual-source reagent, effectively combining two synthetic steps into one, which reduces the overall number of unit operations and solvent consumption. Additionally, the use of solid molybdenum carbonyl as a CO surrogate avoids the logistical costs and safety premiums associated with purchasing and storing pressurized carbon monoxide cylinders. The high yields reported, reaching up to 97% for certain substrates, mean that less raw material is wasted, directly improving the mass balance and lowering the cost per kilogram of the final API intermediate. The simplified workup procedure further contributes to cost savings by reducing labor hours and chromatography media usage compared to more tedious traditional purifications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, a critical factor for maintaining a reliable supply chain. The tolerance for various functional groups means that the same platform technology can be applied to synthesize a wide array of derivatives without needing to re-optimize the entire process for each new analog. This modularity allows manufacturers to respond quickly to changing demands from R&D departments or clinical trial requirements. Since the reagents are stable and the reaction does not require cryogenic conditions or inert atmosphere gloveboxes for setup, the barrier to entry for contract manufacturing organizations is low, increasing the number of potential qualified suppliers and reducing dependency on single-source vendors.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous gases. The waste profile is relatively benign, primarily consisting of organic solvents and metal residues that can be managed through standard waste treatment protocols. The high atom economy of using benzisoxazole reduces the generation of stoichiometric byproducts, aligning with green chemistry principles. This environmental friendliness simplifies the permitting process for new manufacturing lines and supports corporate sustainability goals. The ability to run the reaction at 100°C in common solvents like DME ensures that energy consumption remains moderate, further enhancing the overall sustainability and cost-efficiency of the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived directly from the experimental data and claims of the patent. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing pipelines.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (C1) source, eliminating the need for separate toxic carbonylating agents like carbon monoxide gas and simplifying the atom economy of the reaction.
Q: What is the typical yield range for this palladium-catalyzed process?
A: The patented method demonstrates high reaction efficiency, with isolated yields ranging significantly from 68% to 97% across various substituted substrates, indicating robust functional group tolerance.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials and standard laboratory equipment (sealed tubes), with a simple workup procedure involving filtration and chromatography, making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed aminocarbonylation technology for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We understand that every molecule is unique, and our team of expert chemists is ready to adapt this robust methodology to your specific substrate requirements, optimizing yields and minimizing impurities to deliver a product that exceeds expectations.
We invite you to collaborate with us to leverage this advanced synthetic route for your upcoming projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs, helping you identify opportunities for further optimization. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your drug development timeline with our reliable supply of high-quality 3-arylquinolin-2(1H)-one derivatives.
