Advanced Palladium-Catalyzed Carbonylation for Scalable 3-Arylquinolin-2(1H)-one Production
Introduction to Next-Generation Quinolinone Synthesis
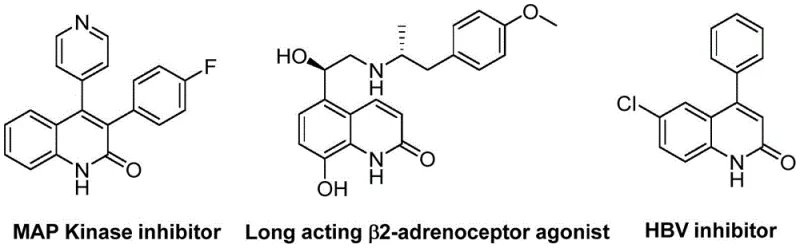
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to access biologically active scaffolds. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinolin-2(1H) ketone derivatives. These quinolinone cores are ubiquitous in medicinal chemistry, serving as critical pharmacophores in a wide array of therapeutic agents ranging from antibiotics and antiplatelet drugs to potent antitumor candidates and endothelin receptor antagonists. The traditional reliance on multi-step syntheses often hampers the rapid development of these valuable intermediates. However, the methodology described in this patent leverages a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm. By utilizing benzisoxazole as a unique dual-source reagent—providing both the nitrogen atom and the essential formyl group—the process achieves remarkable atom economy and operational simplicity. This innovation not only streamlines the synthetic route but also opens new avenues for constructing complex heterocycles with high precision and efficiency, addressing the critical needs of modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one skeleton has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods have served the community well for decades, they are increasingly viewed through the lens of modern green chemistry as suboptimal for large-scale industrial applications. Conventional routes frequently necessitate harsh reaction conditions, including the use of strong acids, high temperatures, or toxic reagents like phosphorus oxychloride, which generate substantial hazardous waste streams. Furthermore, many traditional approaches suffer from limited substrate scope, often failing to tolerate sensitive functional groups that are prevalent in advanced pharmaceutical intermediates. The requirement for pre-functionalized starting materials can also lead to lengthy linear syntheses, driving up costs and extending lead times. Additionally, older transition-metal catalyzed methods often struggled with regioselectivity issues or required expensive and air-sensitive catalysts that were difficult to handle outside of specialized laboratory environments, creating bottlenecks for reliable supply chains.
The Novel Approach
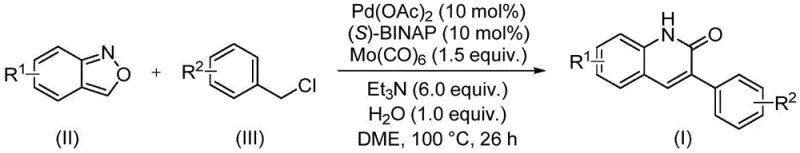
In stark contrast, the novel approach outlined in the patent data presents a robust and versatile solution that overcomes these historical hurdles. This method employs a palladium-catalyzed carbonylation coupling reaction that operates under relatively mild conditions, typically around 100°C, using ethylene glycol dimethyl ether (DME) as the solvent. The true genius of this protocol lies in the strategic use of benzisoxazole derivatives, which act as stable, solid surrogates for carbon monoxide and ammonia equivalents. This eliminates the safety hazards associated with handling high-pressure CO gas. The reaction demonstrates exceptional functional group tolerance, successfully accommodating electron-withdrawing groups like cyano and trifluoromethyl, as well as electron-donating alkoxy and alkyl substituents on both the benzisoxazole and benzyl chloride partners. This breadth of compatibility allows chemists to access a diverse library of 3-arylquinolin-2(1H)one derivatives from readily available commercial building blocks, significantly accelerating the optimization of structure-activity relationships (SAR) in drug development programs.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The mechanistic underpinning of this transformation involves a complex catalytic cycle centered around a palladium species coordinated with a chiral bisphosphine ligand, specifically (S)-1,1'-binaphthyl-2,2'-bis-diphenylphosphine ((S)-BINAP). The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active Pd(0) species, generating an organopalladium intermediate. Concurrently, molybdenum hexacarbonyl (Mo(CO)6) serves as a solid carbon monoxide source, releasing CO in situ under the thermal conditions to facilitate the carbonylation step. The benzisoxazole ring then undergoes a nucleophilic attack or insertion sequence, effectively cleaving the N-O bond to deliver the nitrogen atom into the growing heterocyclic framework while simultaneously providing the carbonyl carbon. This intricate dance of bond-breaking and bond-forming events is meticulously balanced by the presence of triethylamine as a base and a stoichiometric amount of water, which appears to play a crucial role in the hydrolysis or proton transfer steps necessary to finalize the lactam ring closure. The result is the formation of the thermodynamically stable quinolinone core with high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy thermal cyclizations. The use of a well-defined homogeneous catalyst system ensures that the reaction proceeds through a controlled organometallic pathway, minimizing the formation of random polymeric byproducts or isomeric impurities that are common in less selective processes. The specific choice of the (S)-BINAP ligand not only enhances the catalytic turnover but also helps in stabilizing the reactive intermediates, preventing premature decomposition of the sensitive benzisoxazole starting material. Furthermore, the reaction conditions are optimized to favor the desired intramolecular cyclization over intermolecular side reactions, leading to crude reaction profiles that are significantly cleaner. This inherent selectivity reduces the burden on downstream purification processes, allowing for simpler workup procedures such as filtration and standard column chromatography to yield high-purity products suitable for further biological evaluation or formulation.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The protocol is designed to be operationally simple, avoiding the need for specialized high-pressure equipment typically associated with carbonylation reactions. The process begins by charging a sealed reaction vessel with the palladium catalyst precursor, the chiral ligand, the solid CO source, and the organic substrates in the appropriate solvent system. Maintaining an inert atmosphere is generally recommended to protect the catalyst integrity, although the specific robustness of this system allows for some flexibility. The reaction is then heated to the optimal temperature range of 90 to 110°C, with 100°C identified as the sweet spot for balancing reaction rate and selectivity. Monitoring the reaction progress is essential, with 26 hours typically sufficient for complete conversion of the starting materials. Upon completion, the mixture is cooled, and the product is isolated through a straightforward workup involving filtration to remove metal residues and silica gel treatment, followed by purification.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant reduction of raw material complexity and cost. By utilizing benzisoxazoles and benzyl chlorides, which are commodity chemicals available in bulk quantities from multiple global suppliers, the dependency on exotic or custom-synthesized precursors is eliminated. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring a more stable and predictable procurement landscape. Moreover, the elimination of gaseous carbon monoxide removes the need for specialized gas handling infrastructure and safety protocols, thereby reducing capital expenditure and operational overheads associated with regulatory compliance and facility maintenance.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the streamlined nature of the reaction sequence. Traditional methods often require multiple isolation steps, protecting group manipulations, and the use of expensive activating agents, all of which accumulate costs in terms of materials, labor, and waste disposal. In contrast, this one-pot aminocarbonylation approach consolidates multiple bond-forming events into a single operation, drastically reducing the number of unit operations required. The use of a solid CO surrogate like Mo(CO)6 avoids the logistical challenges and safety costs of pressurized gas cylinders. Furthermore, the high reaction efficiency and yields reported (often exceeding 90% for various substrates) mean that less raw material is wasted, directly improving the overall mass balance and lowering the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of this chemistry. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical manufacturing facilities, removing the barrier to entry for contract manufacturing organizations (CMOs). The broad substrate scope means that a single platform technology can be used to produce a wide variety of analogues, allowing for agile responses to changing market demands or clinical trial requirements without the need for extensive process re-development. The availability of starting materials like benzisoxazole and substituted benzyl chlorides from established chemical supply chains ensures that production schedules can be maintained without interruption, supporting just-in-time manufacturing models and reducing inventory holding costs.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns well with modern sustainability goals. The reaction generates fewer byproducts compared to classical condensation reactions, resulting in a lower E-factor (mass of waste per mass of product). The solvent system, primarily DME, can be recovered and recycled, further minimizing environmental impact. The simplified workup procedure, which avoids complex extractions or crystallizations in the early stages, reduces solvent consumption and energy usage. As regulatory pressures on pharmaceutical manufacturing increase, having a process that inherently produces less hazardous waste and utilizes safer reagents provides a competitive advantage in obtaining necessary environmental permits and maintaining social license to operate, facilitating smoother scale-up from pilot plant to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this method for industrial applications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for R&D groups planning new synthetic campaigns.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate toxic carbonylating agents like carbon monoxide gas and simplifying the reaction stoichiometry.
Q: What is the functional group tolerance of this Pd-catalyzed method?
A: The method exhibits excellent tolerance for various substituents including halogens (Cl, F), alkoxy groups (OMe), cyano groups, and bulky alkyl groups (t-Bu), allowing for diverse derivative synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of commercially available starting materials, moderate reaction temperatures (100°C), and straightforward post-processing makes this route highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN113045489B for accelerating drug development. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. We understand that the purity of intermediates like 3-arylquinolin-2(1H)-ones is critical for downstream synthesis, which is why our stringent purity specifications and rigorous QC labs are dedicated to delivering materials that meet the highest industry standards, minimizing risks in your final API synthesis.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this efficient route can optimize your budget. We encourage you to reach out today to discuss your requirements, obtain specific COA data for our catalog compounds, or initiate route feasibility assessments for your proprietary targets, ensuring a seamless path from concept to commercial reality.
