Advanced Metal-Free Photochemical Acylation for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking innovative synthetic routes that balance efficiency with environmental sustainability. Patent CN109293491B presents a groundbreaking advancement in organic synthesis by introducing a novel method for the dediazonation and acylation of aryl diazonium salts. This technology leverages visible light irradiation to drive the transformation of aryl diazonium salts and their derivatives into corresponding aryl acyl products without the need for transition metal catalysts. For R&D directors and process chemists, this represents a significant shift away from traditional heavy-metal-dependent protocols towards greener, photochemical methodologies. The patent details a robust protocol that achieves high yields, with some examples reaching up to 72%, while maintaining exceptional operational simplicity. By utilizing readily available ortho-dicarbonyl compounds as acyl sources, this invention fills a critical gap in the functionalization of aromatic amines, offering a versatile tool for constructing complex molecular architectures found in modern drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of acyl groups onto aromatic rings has relied heavily on classical Friedel-Crafts acylation or transition-metal-catalyzed cross-coupling reactions. Friedel-Crafts chemistry, while foundational, suffers from severe drawbacks including the requirement for stoichiometric amounts of corrosive Lewis acids like anhydrous aluminum chloride, which generates substantial hazardous waste and complicates downstream processing. Furthermore, these reactions often lack precise regioselectivity, leading to difficult-to-separate isomeric mixtures that reduce overall process efficiency. Alternatively, transition-metal-catalyzed methods involving copper or palladium, such as those reported by the Citterio or Cordia teams, introduce the risk of heavy metal contamination. For pharmaceutical applications, removing trace metals to meet stringent regulatory limits adds costly purification steps and extends production timelines. These conventional approaches also frequently demand harsh reaction conditions, such as elevated temperatures or inert atmospheres, which increase energy consumption and limit the compatibility with sensitive functional groups present in complex intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN109293491B utilizes a metal-free photochemical strategy that operates under remarkably mild conditions. By employing aryl diazonium salts and ortho-dicarbonyl compounds as substrates, the reaction proceeds efficiently upon exposure to visible light, typically blue light, at room temperature. This approach eliminates the need for expensive and toxic transition metal catalysts, thereby inherently reducing the risk of metal impurities in the final product. The process demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and esters without degradation. Crucially, the method offers superior positional selectivity, particularly for pyridine derivatives where the diazo group is positioned meta to the nitrogen atom, a transformation that is notoriously difficult with traditional electrophilic substitution. This innovation not only streamlines the synthetic workflow but also aligns perfectly with the industry's push towards greener chemistry and cost reduction in API manufacturing.
Mechanistic Insights into Photochemical Dediazonation Acylation
The core of this technological breakthrough lies in the unique interaction between the aryl diazonium cation and the ortho-dicarbonyl compound under photonic excitation. Unlike thermal reactions that rely on high activation energy, this photochemical process initiates a radical pathway where the diazonium group serves as a leaving group, generating an aryl radical intermediate. This highly reactive species then couples with the dicarbonyl compound to form the desired carbon-carbon bond, effectively installing the acyl group at the site previously occupied by the diazonium moiety. Research indicates that the electronic nature of the substituents on the aromatic ring plays a pivotal role in reaction efficiency. Specifically, electron-donating groups at the para-position, such as methoxy or alkyl groups, significantly enhance the yield, likely by stabilizing the radical intermediate or facilitating the electron transfer process. Conversely, steric hindrance at the ortho-position can impede the reaction, highlighting the importance of substrate design in maximizing conversion rates.
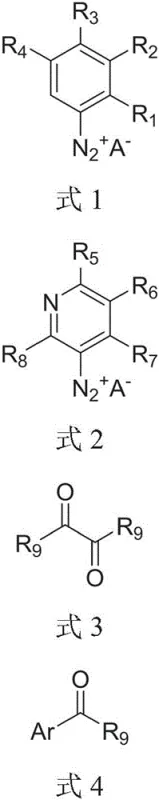
Furthermore, the mechanistic study reveals critical structural requirements for successful acylation, particularly regarding heterocyclic systems. For pyridine-based diazonium salts, the position of the diazo group relative to the ring nitrogen is determinant; only meta-substituted diazonium salts undergo efficient acylation, while ortho or para isomers fail to react. This specificity suggests a complex interplay between the electronic distribution of the heterocycle and the radical propagation steps. The addition of auxiliary agents, such as sodium trifluoromethanesulfinate or sodium acetate, further optimizes the reaction environment. These additives appear to suppress competing side reactions, such as simple dediazonation without acylation, thereby channeling the reaction pathway towards the desired ketone product. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, ensuring robust performance across a diverse range of substrates and paving the way for the commercial scale-up of complex organic molecules.
How to Synthesize Acylated Aryl Compounds Efficiently
Implementing this photochemical acylation protocol requires careful attention to reagent ratios and light source selection to ensure optimal conversion. The standard procedure involves dissolving the aryl diazonium tetrafluoroborate salt and the auxiliary agent in a polar aprotic solvent like acetonitrile, followed by the addition of a large excess of the ortho-dicarbonyl compound, typically dimethyldione. The reaction mixture is then subjected to irradiation using a blue light source, such as a 36W LED array, at ambient temperature for a duration ranging from 2 to 20 hours depending on the substrate reactivity. Post-reaction workup is straightforward, involving aqueous quenching, extraction with dichloromethane, and purification via silica gel chromatography. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below, providing a clear roadmap for laboratory execution.
- Prepare the aryl diazonium salt substrate (Formula 1 or 2) and mix with an ortho-dicarbonyl compound (Formula 3) in a suitable solvent like acetonitrile.
- Add an auxiliary agent such as sodium trifluoromethanesulfinate or sodium acetate to enhance yield and suppress side reactions.
- Irradiate the reaction mixture with blue light (e.g., 36W) at room temperature for 2 to 20 hours, followed by extraction and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free photochemical method offers transformative benefits that directly impact the bottom line and operational resilience. By removing the dependency on precious metal catalysts like palladium or copper, manufacturers can achieve substantial cost savings on raw material procurement. More importantly, the elimination of heavy metals simplifies the purification process, removing the need for expensive scavenging resins or complex recrystallization steps designed to meet strict residual metal specifications. This streamlined workflow translates into shorter production cycles and reduced labor costs, enhancing overall manufacturing efficiency. Additionally, the use of benign reagents and mild reaction conditions minimizes the generation of hazardous waste, lowering disposal costs and ensuring compliance with increasingly stringent environmental regulations. These factors collectively contribute to a more sustainable and economically viable supply chain for high-value chemical intermediates.
- Cost Reduction in Manufacturing: The absence of transition metal catalysts eliminates a major cost driver associated with both the purchase of expensive reagents and the subsequent removal processes. Traditional methods often require specialized equipment and extensive quality control testing to verify metal levels, whereas this photochemical route inherently produces cleaner crude products. The simplified downstream processing reduces solvent consumption and energy usage, as there is no need for high-temperature reflux or high-pressure reactors. Consequently, the overall cost of goods sold (COGS) for acylated intermediates can be significantly optimized, making the final API more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on transition metals exposes the supply chain to geopolitical risks and price volatility, as seen with palladium and platinum markets. This new method utilizes commodity chemicals such as aryl amines and simple diketones, which are widely available from multiple global suppliers, ensuring a stable and diversified supply base. The robustness of the reaction conditions, operating effectively at room temperature and atmospheric pressure, reduces the risk of batch failures due to equipment malfunction or parameter deviation. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risk of production delays that can ripple through the entire pharmaceutical value chain and affect time-to-market for new drugs.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in flow chemistry and reactor design have made this increasingly feasible for industrial applications. The mild nature of this reaction means it can be adapted to continuous flow systems, which offer superior heat and mass transfer compared to batch processing. This facilitates the commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton scales with minimal safety risks. Furthermore, the reduction in hazardous waste and the avoidance of toxic heavy metals align perfectly with green chemistry principles, helping companies meet their corporate sustainability goals and adhere to strict environmental discharge limits imposed by regulatory bodies worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical acylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical implications for industrial adoption. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or new drug development pipelines. The answers highlight the versatility of the method across different substrate classes and its superiority over legacy technologies in terms of purity and operational safety.
Q: What are the advantages of this photochemical method over traditional Friedel-Crafts acylation?
A: Unlike Friedel-Crafts reactions which require harsh Lewis acids and generate significant waste, this photochemical method operates under mild, metal-free conditions with superior positional selectivity.
Q: Does this method work for heterocyclic compounds like pyridine?
A: Yes, the method successfully acylates pyridine rings, provided the diazo group is located at the meta-position relative to the nitrogen atom, overcoming limitations of prior art.
Q: How does this process impact impurity profiles in pharmaceutical intermediates?
A: By eliminating transition metal catalysts like copper or palladium, the process avoids heavy metal residues, significantly simplifying purification and ensuring high-purity specifications for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Diazonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this metal-free photochemical acylation technology to revolutionize the synthesis of key pharmaceutical building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier global pharmaceutical companies. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the highest quality standards, leveraging our deep expertise in organic synthesis to optimize yield and minimize impurities for your specific project needs.
We invite you to collaborate with us to explore how this cutting-edge technology can enhance your supply chain efficiency and product quality. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating the tangible economic benefits of switching to this greener synthetic route. Please contact our technical procurement team today to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive innovation and efficiency in the development of next-generation therapeutics.
