Advanced Photochemical Acylation of Aryl Diazonium Salts for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective synthetic routes for complex intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN109293491B, which introduces a novel method for the dediazoniation and subsequent acylation of aryl diazonium salts. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a greener, metal-free photochemical approach. By leveraging visible light irradiation, specifically blue light, this method enables the direct transformation of readily available aryl amines into valuable aryl ketones with high regioselectivity. For R&D directors and procurement managers alike, this innovation offers a compelling solution for producing high-purity pharmaceutical intermediates while drastically simplifying the supply chain by removing the dependency on scarce noble metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of acyl groups onto aromatic rings has relied heavily on classical Friedel-Crafts acylation or transition-metal catalyzed cross-coupling reactions. The Friedel-Crafts methodology, while foundational, suffers from significant drawbacks including the requirement for stoichiometric amounts of harsh Lewis acids like anhydrous aluminum chloride, which generates substantial acidic waste and poses severe corrosion challenges for industrial reactors. Furthermore, these reactions often lack precise regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. Alternatively, metal-catalyzed approaches using copper or palladium, such as those reported by the Citterio and Cordia teams, introduce their own set of logistical burdens. These methods necessitate the use of expensive catalysts, rigorous exclusion of oxygen and moisture, and, critically, extensive downstream processing to remove trace metal residues to meet stringent pharmaceutical purity standards. The environmental footprint and the cost associated with metal recovery and waste treatment make these conventional routes increasingly unattractive for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the technology disclosed in patent CN109293491B utilizes a mild, metal-free photochemical strategy that overcomes the inherent limitations of prior art. By reacting aryl diazonium salts with ortho-dicarbonyl compounds under simple blue light irradiation at room temperature, this method achieves direct acylation without the need for toxic Lewis acids or precious metal catalysts. The process operates under ambient pressure and temperature conditions, significantly reducing energy consumption and safety risks associated with high-pressure or high-temperature reactors. Moreover, the reaction exhibits exceptional positional selectivity, particularly favoring para-substituted products when electron-donating groups are present, thereby minimizing the formation of unwanted isomers. This streamlined one-pot procedure not only accelerates the synthesis timeline but also aligns perfectly with modern green chemistry principles, offering a robust pathway for the cost reduction in pharmaceutical intermediate manufacturing.
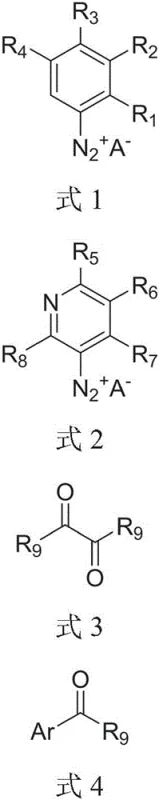
Mechanistic Insights into Photochemical Dediazoniation and Acylation
The core of this innovative synthesis lies in the unique reactivity of aryl diazonium salts under photolytic conditions. Upon exposure to blue light, the diazonium species undergoes homolytic cleavage to generate highly reactive aryl radicals. These radicals then interact with the ortho-dicarbonyl compound, such as dimethyldione, initiating a radical addition sequence that ultimately leads to the formation of the carbon-carbon bond and the installation of the acyl group. The presence of specific additives, such as sodium trifluoromethanesulfinate, plays a crucial mechanistic role by acting as a radical mediator or stabilizer, which helps to suppress competing side reactions like simple dediazoniation (reduction to the parent arene). This careful balancing of radical kinetics ensures that the desired acylation pathway dominates, resulting in the high yields observed across a diverse range of substrates. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for new analogues.
Furthermore, the structural requirements of the substrates provide deep insights into the steric and electronic factors governing the reaction efficiency. Research indicates that for benzene-based diazonium salts, having at least one hydrogen atom at the ortho-position relative to the diazo group is favorable for successful acylation. In the case of heterocyclic systems like pyridine, the position of the diazo group is critical; acylation proceeds efficiently only when the diazo group is located at the meta-position relative to the ring nitrogen. Ortho or para substitution on the pyridine ring hinders the reaction, likely due to electronic deactivation or steric interference with the radical intermediate. This level of specificity allows chemists to predict outcomes accurately and design synthetic routes that maximize yield while minimizing impurity profiles, ensuring the production of high-purity OLED material or API precursors with consistent quality.
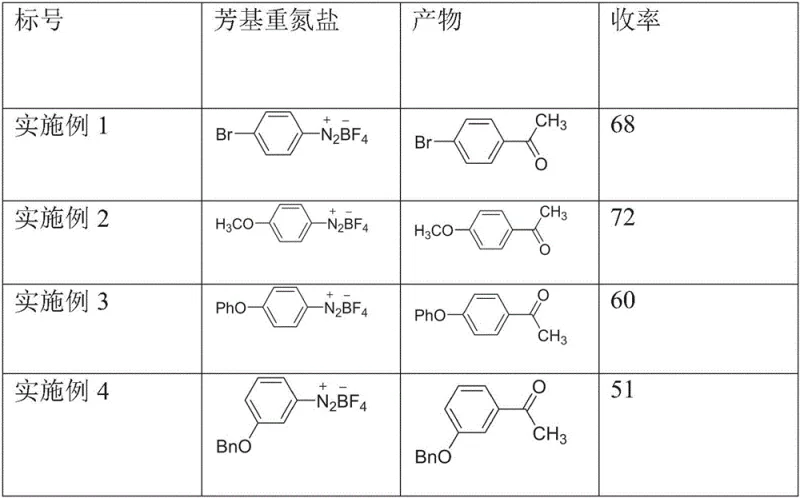
How to Synthesize Acylated Aryl Compounds Efficiently
Implementing this photochemical acylation protocol requires attention to specific operational details to ensure reproducibility and safety. The standard procedure involves dissolving the aryl diazonium tetrafluoroborate salt and the auxiliary agent in a polar aprotic solvent like acetonitrile, followed by the addition of a large excess of the dicarbonyl coupling partner. The reaction mixture is then subjected to continuous irradiation using a standard blue light source, typically a 36W LED array, for a period ranging from 2 to 20 hours depending on the substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the aryl diazonium tetrafluoroborate salt and sodium trifluoromethanesulfinate additive in acetonitrile solvent within a glass vessel.
- Add a significant excess of the ortho-dicarbonyl compound, such as dimethyldione, to the reaction flask using a syringe to ensure complete conversion.
- Irradiate the mixture with blue light (e.g., 36W) at room temperature for approximately 12 hours, followed by extraction and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free photochemical technology translates into tangible strategic advantages beyond mere technical novelty. The elimination of transition metal catalysts removes a major cost driver and a significant supply chain bottleneck, as the prices of palladium and copper can be volatile and their availability subject to geopolitical constraints. Additionally, the absence of heavy metals simplifies the purification process, reducing the number of unit operations required to meet regulatory specifications for residual metals in drug substances. This streamlining of the downstream processing directly contributes to substantial cost savings and shorter lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive noble metal catalysts and the associated ligands which are often required in traditional cross-coupling reactions. By utilizing simple, commodity-grade reagents like aryl amines and dicarbonyl compounds, the raw material costs are significantly lowered. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure equipment or cryogenic cooling systems, allowing the process to be run in standard glass-lined or stainless steel reactors with minimal energy input. The simplified workup procedure, which avoids complex metal scavenging steps, further reduces operational expenditures and waste disposal costs, making the overall manufacturing process economically superior to conventional methods.
- Enhanced Supply Chain Reliability: Relying on a synthetic route that depends on scarce transition metals introduces inherent supply chain risks, especially during periods of global shortage. This photochemical method mitigates such risks by utilizing widely available organic starting materials that can be sourced from multiple suppliers globally. The robustness of the reaction conditions, which tolerate a variety of functional groups and proceed at room temperature, ensures consistent production output regardless of minor fluctuations in utility supplies. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the strict delivery timelines demanded by multinational pharmaceutical clients, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in reactor design now allow for efficient commercial scale-up of complex organic transformations. The simplicity of this one-pot reaction facilitates easy translation from laboratory benchtop to pilot plant and full-scale production. From an environmental perspective, the process generates significantly less hazardous waste compared to Friedel-Crafts acylation, which produces stoichiometric amounts of aluminum waste. The reduced environmental footprint aligns with increasingly stringent global regulations on industrial emissions and waste management, ensuring long-term compliance and sustainability for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical acylation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on substrate compatibility, reaction conditions, and product quality. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: Does this photochemical acylation method require transition metal catalysts?
A: No, the method described in patent CN109293491B is entirely metal-free. It utilizes visible light irradiation to drive the reaction, eliminating the need for expensive copper or palladium catalysts and avoiding heavy metal contamination in the final product.
Q: What is the substrate scope for this acylation reaction?
A: The process demonstrates broad applicability for benzene-based diazonium salts with various substituents including halogens, alkoxy groups, and esters. It is also effective for pyridine rings provided the diazo group is located at the meta-position relative to the nitrogen atom.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal results are achieved using acetonitrile as the solvent at room temperature under blue light irradiation. The addition of auxiliaries like sodium trifluoromethanesulfinate significantly improves yield and suppresses dediazoniation by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Diazonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the photochemical acylation described in CN109293491B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable industrial processes. Our state-of-the-art facilities are equipped with specialized photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aryl ketone intermediates delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this metal-free acylation route can optimize your specific project requirements. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this technology can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for high-value fine chemicals.
