Advanced Metal-Free Synthesis of Pyrimidoindazole Compounds for Commercial Scale-Up
Introduction to Novel Pyrimidoindazole Synthesis Technology
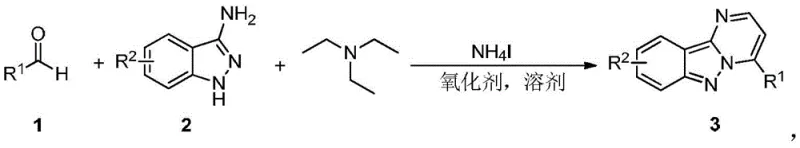
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery. A significant breakthrough in this domain is detailed in Chinese patent CN109912606B, which discloses a highly efficient, transition-metal-free method for synthesizing pyrimidoindazole compounds. These fused heterocyclic systems are increasingly recognized for their potent biological activities, functioning as potential protein kinase inhibitors and therapeutic agents for neurodegenerative diseases and viral infections. The core innovation lies in a one-pot tandem reaction that directly couples aromatic aldehydes with 3-aminoindazoles using ammonium iodide as a catalyst and triethylamine as a key reagent. This approach fundamentally shifts the paradigm from traditional multi-step syntheses, offering a streamlined pathway that minimizes waste and maximizes atom economy. By leveraging simple, commercially available starting materials and avoiding expensive heavy metal catalysts, this technology presents a compelling value proposition for both research and development teams and supply chain strategists looking to optimize their API intermediate sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrimidoindazole core has relied on methodologies that introduce significant operational complexity and environmental burden. Traditional routes often involve the condensation of 3-aminoindazole with 1,3-dicarbonyl compounds or propiolates, which frequently require harsh reaction conditions and generate substantial byproduct streams. Furthermore, Grieco condensation reactions involving alkenes or alkynes often necessitate the use of corrosive reagents and stoichiometric amounts of activating agents that complicate downstream processing. A major bottleneck in these legacy processes is the reliance on transition metal catalysts, which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding residual metal limits in final pharmaceutical products. The purification of intermediates at each step of a multi-step sequence leads to cumulative yield losses and increased solvent consumption, creating a sustainability challenge that modern green chemistry principles aim to resolve. Consequently, the search for a direct, catalytic, and metal-free alternative has become a priority for process chemists aiming to enhance the viability of these bioactive scaffolds.
The Novel Approach
The methodology outlined in the patent data represents a decisive leap forward by integrating the cyclization and oxidation steps into a single operational unit. By utilizing ammonium iodide in conjunction with an oxidant such as di-tert-butyl peroxide or even molecular oxygen, the reaction proceeds through a radical-mediated or iodine-catalyzed pathway that efficiently constructs the pyrimidine ring onto the indazole core. The use of triethylamine serves a dual purpose, acting both as a base to facilitate condensation and as a solvent component that stabilizes the reaction medium. This one-pot strategy effectively bypasses the need for isolating sensitive intermediates, thereby reducing the overall processing time and labor intensity. The reaction conditions are remarkably mild, typically operating between 110°C and 150°C, which allows for the tolerance of a wide array of functional groups including halogens, nitro groups, and ethers. This robustness ensures that diverse analogues can be generated rapidly without the need for protecting group strategies, accelerating the lead optimization phase in drug discovery programs while simultaneously laying the groundwork for cost-effective manufacturing.
Mechanistic Insights into NH4I-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its reproducibility and scope. The reaction likely initiates with the formation of an imine intermediate between the aromatic aldehyde and the amino group of the 3-aminoindazole, facilitated by the basic environment provided by triethylamine. The ammonium iodide then plays a pivotal role, potentially generating reactive iodine species in situ upon interaction with the oxidant. These active iodine species can promote the subsequent cyclization by activating the C-H bond adjacent to the nitrogen or by facilitating a radical addition pathway depending on the specific oxidant employed. For instance, when peroxides like DTBP are used, a radical mechanism involving hydrogen abstraction is plausible, leading to the closure of the six-membered pyrimidine ring. The final aromatization step yields the stable pyrimidoindazole system. This mechanistic elegance ensures high selectivity, as the reaction is driven by the thermodynamic stability of the fused aromatic system, minimizing the formation of regioisomers or polymeric side products that often plague condensation reactions.
From an impurity control perspective, the absence of transition metals eliminates a whole class of potential contaminants related to metal-ligand complexes. The primary impurities are likely to be unreacted starting materials or over-oxidized byproducts, both of which are generally easier to remove via standard crystallization or chromatography compared to metal residues. The use of chlorobenzene or toluene as solvents further aids in purification, as these high-boiling solvents allow for high-temperature reactions that drive equilibrium towards the product while remaining compatible with standard workup procedures involving aqueous washes. The broad substrate scope demonstrated in the patent examples, ranging from electron-rich trimethoxyphenyl aldehydes to electron-deficient nitro-substituted variants, confirms the versatility of this mechanistic pathway. This predictability is essential for scaling up, as it reduces the risk of unexpected side reactions when switching between different batches of raw materials or when introducing slight modifications to the molecular structure.
How to Synthesize Pyrimidoindazole Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and temperature control to maximize yield and purity. The standard protocol involves dissolving the aromatic aldehyde and 3-aminoindazole in a solvent like chlorobenzene, followed by the addition of triethylamine and ammonium iodide. The mixture is then heated to the optimal temperature range, typically around 120°C, under an atmosphere of air or oxygen if using aerobic oxidation, or sealed if using peroxide oxidants. The reaction progress can be monitored via TLC or HPLC, with completion usually achieved within 12 hours. Workup involves quenching with water, extraction with ethyl acetate, and purification via silica gel chromatography. Detailed standardized synthesis steps see the guide below.
- Dissolve aromatic aldehyde, 3-aminoindazole, and triethylamine in a suitable solvent such as chlorobenzene.
- Add ammonium iodide (NH4I) and a selected oxidant (e.g., DTBP, DMSO, or air) to the reaction mixture.
- Heat the mixture to 110-150°C under stirring for approximately 12 hours to yield the target pyrimidoindazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic route are profound. The elimination of precious metal catalysts such as palladium or rhodium removes a significant variable cost driver and mitigates supply chain risks associated with the volatility of precious metal markets. Furthermore, the use of commodity chemicals like triethylamine and ammonium iodide ensures a stable and low-cost supply of reagents that are readily available from multiple global vendors. The one-pot nature of the reaction drastically reduces the number of unit operations required, translating to lower energy consumption, reduced solvent usage, and decreased labor costs per kilogram of product. This efficiency gain is critical for maintaining competitive margins in the production of high-volume pharmaceutical intermediates. Additionally, the simplified workflow shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive ammonium iodide results in a direct reduction in raw material expenditure. Moreover, the avoidance of multi-step purification processes for intermediates significantly lowers the cost of goods sold by minimizing solvent waste and processing time. The ability to use air as an oxidant in certain variations further eliminates the cost of specialized chemical oxidants, contributing to a leaner and more economical manufacturing process that enhances overall profitability.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of widely available starting materials such as substituted benzaldehydes and 3-aminoindazoles, which are produced at scale by numerous chemical suppliers globally. This diversification of the supply base reduces dependency on single-source vendors and mitigates the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when supply chains face logistical challenges.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the simplicity of the reaction setup, which does not require specialized high-pressure equipment or inert atmosphere gloveboxes for all variations. The reduced generation of hazardous waste, particularly heavy metal sludge, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only simplifies waste disposal logistics but also enhances the company's reputation as a responsible manufacturer, which is a key factor for long-term partnerships with major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrimidoindazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and reliability for decision-makers. Understanding these details helps in assessing the feasibility of adopting this route for specific project requirements and in planning the necessary resource allocation for process development.
Q: What are the key advantages of this pyrimidoindazole synthesis method?
A: The method described in patent CN109912606B eliminates the need for transition metal catalysts, utilizes inexpensive raw materials like triethylamine, and operates under mild conditions with a wide substrate scope, significantly simplifying purification and reducing environmental impact.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the one-pot tandem reaction design avoids complex intermediate isolation steps, uses readily available solvents like chlorobenzene, and tolerates open-air conditions with certain oxidants, making it highly amenable to commercial scale-up.
Q: What types of substituents are tolerated on the aromatic aldehyde?
A: The protocol demonstrates excellent functional group tolerance, accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl on the phenyl ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidoindazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN109912606B for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and reliable. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry allows us to offer customized solutions that optimize both cost and quality for our global partners.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your drug development timeline while reducing overall production costs. Let us be your trusted partner in bringing innovative medicines to market faster and more efficiently.
