Advanced Metal-Free Synthesis of Pyrimidoindazole Compounds for Pharmaceutical Applications
Introduction to Next-Generation Pyrimidoindazole Synthesis
The pharmaceutical industry continuously seeks robust and economical pathways for constructing complex heterocyclic scaffolds, particularly those serving as core structures for bioactive molecules. A significant breakthrough in this domain is documented in Chinese patent CN109912606B, which discloses a highly efficient synthetic method for pyrimidoindazole compounds. These derivatives are increasingly recognized for their potent biological activities, functioning as potential protein kinase inhibitors and therapeutic agents for conditions ranging from senile dementia to various cancers. The disclosed technology represents a paradigm shift by utilizing a transition-metal-free, one-pot tandem reaction strategy that directly couples aromatic aldehydes with 3-aminoindazoles. This approach not only simplifies the operational workflow but also addresses critical environmental and economic concerns associated with traditional multi-step syntheses. By leveraging ammonium iodide as a catalyst and common oxidants, the process achieves high yields under relatively mild thermal conditions, establishing a new benchmark for the sustainable manufacturing of high-value pharmaceutical intermediates.
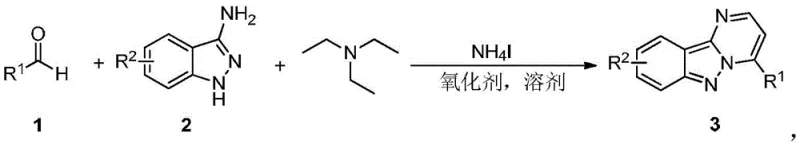
The strategic importance of this methodology lies in its ability to bypass the limitations of conventional routes that often rely on corrosive reagents or precious metal catalysts. The reaction system is remarkably versatile, accommodating a wide array of substrates including substituted benzaldehydes, naphthaldehydes, and heteroaromatic aldehydes. This flexibility is crucial for medicinal chemists aiming to generate diverse libraries for structure-activity relationship (SAR) studies. Furthermore, the use of triethylamine as a readily available promoter significantly lowers the barrier to entry for production, making it an attractive option for both laboratory-scale discovery and industrial-scale supply. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative protocols is essential for maintaining competitiveness in the global market, ensuring that clients receive high-purity materials with optimized lead times and cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrimidoindazole core has been fraught with synthetic challenges that hinder efficient production. Traditional literature methods typically involve the condensation of 3-aminoindazole with 1,3-dicarbonyl compounds or propiolates, or alternatively, Grieco condensation reactions involving alkenes or alkynes. These legacy processes suffer from several inherent drawbacks that impact both the economic viability and the environmental footprint of the synthesis. Firstly, they frequently necessitate the use of corrosive and toxic reagents, which pose significant safety hazards to personnel and require specialized waste treatment infrastructure. Secondly, many established routes depend on expensive transition metal catalysts, which not only inflate the raw material costs but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing trace metals to meet stringent regulatory standards often requires additional purification steps, such as chromatography or specialized scavenging treatments, further driving up processing time and expense. Moreover, these multi-step sequences often result in lower overall yields due to cumulative losses at each stage, generating substantial amounts of chemical waste and limiting the practical application of these methods in large-scale manufacturing settings.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the method outlined in patent CN109912606B introduces a streamlined, one-pot series reaction that fundamentally redefines the synthesis landscape for this compound class. By directly reacting aromatic aldehydes with 3-aminoindazoles in the presence of ammonium iodide and an oxidant, the process eliminates the need for pre-functionalized starting materials or complex intermediate isolations. This telescoped approach drastically reduces the number of unit operations, thereby minimizing solvent consumption and energy usage. The absence of transition metals is a particularly compelling feature, as it removes the necessity for costly metal removal protocols and ensures a cleaner impurity profile from the outset. The reaction conditions are notably mild, typically operating between 110°C and 150°C, which allows for the use of standard glass-lined or stainless-steel reactors without requiring exotic high-pressure equipment. This simplicity translates directly into operational efficiency, enabling manufacturers to produce high-purity pyrimidoindazoles with greater consistency and reliability. The adoption of such a green and atom-economical route aligns perfectly with modern principles of sustainable chemistry, offering a clear pathway for cost reduction in API manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into NH4I-Catalyzed Oxidative Cyclization
The success of this synthetic transformation hinges on the unique catalytic role of ammonium iodide (NH4I) in conjunction with an oxidant to facilitate the oxidative cyclization. Mechanistically, the reaction likely proceeds through an initial condensation between the amino group of the 3-aminoindazole and the carbonyl group of the aromatic aldehyde to form an imine intermediate. The iodide ion, generated in situ from the ammonium salt, acts as a nucleophilic catalyst or a redox mediator, potentially activating the C-H bond adjacent to the nitrogen or facilitating the formation of reactive iodine species under oxidative conditions. When oxidants such as di-tert-butyl peroxide (DTBP) or molecular oxygen are employed, they serve to regenerate the active catalytic species and drive the thermodynamic equilibrium towards the formation of the fused pyrimidine ring. This oxidative aromatization step is critical for establishing the stable heterocyclic core characteristic of the pyrimidoindazole scaffold. The presence of triethylamine serves a dual purpose: it acts as a base to neutralize acidic byproducts and may also assist in deprotonation steps essential for the cyclization cascade. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as oxidant selection and temperature, to maximize conversion and minimize the formation of side products like over-oxidized species or polymerization byproducts.
From an impurity control perspective, the metal-free nature of this catalysis offers distinct advantages for downstream processing. In traditional metal-catalyzed reactions, trace metals can coordinate with product molecules or catalyze degradation pathways during storage, leading to stability issues. By avoiding transition metals entirely, the impurity profile of the crude reaction mixture is significantly simplified, primarily consisting of unreacted starting materials or simple condensation byproducts that are easier to separate via crystallization or standard extraction techniques. The broad substrate tolerance observed in the patent data suggests that the electronic properties of the substituents on the aromatic aldehyde do not severely inhibit the catalytic cycle, indicating a robust mechanism capable of handling both electron-rich and electron-deficient systems. For R&D teams, this implies that the process is resilient to variations in raw material quality and can be adapted for the synthesis of analogues with diverse pharmacological profiles. The ability to predict and control the formation of specific impurities based on the choice of oxidant and solvent further enhances the reliability of the process, ensuring that the final product meets the stringent purity specifications required for clinical and commercial applications.
How to Synthesize Pyrimidoindazole Efficiently
Implementing this synthesis route in a practical setting requires careful attention to reagent stoichiometry and reaction conditions to ensure optimal performance. The patent provides a detailed framework where aromatic aldehydes, 3-aminoindazoles, and triethylamine are dissolved in a solvent such as chlorobenzene, followed by the addition of ammonium iodide and an oxidant. The mixture is then heated to temperatures ranging from 110°C to 150°C, typically for a duration of 12 hours, to achieve high conversion rates. Work-up procedures involve standard aqueous quenching, extraction with organic solvents like ethyl acetate, and purification via silica gel chromatography or recrystallization. The detailed standardized synthesis steps for specific derivatives are outlined in the structured guide below, providing a clear roadmap for laboratory execution.
- Dissolve aromatic aldehyde, 3-aminoindazole, and triethylamine in a suitable solvent such as chlorobenzene.
- Add ammonium iodide (NH4I) and a selected oxidant (e.g., DTBP, DMSO, or air) to the reaction mixture.
- Heat the mixture to 110-150°C and stir for approximately 12 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic methodology presents a compelling value proposition centered on cost efficiency and supply security. The elimination of expensive transition metal catalysts represents a direct reduction in raw material expenditure, while the simplified one-pot process reduces labor costs and facility occupancy time. Furthermore, the use of commodity chemicals like triethylamine and common aromatic aldehydes ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized or proprietary reagents. This resilience is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients. The environmental benefits of the process also translate into lower waste disposal costs and reduced regulatory burden, contributing to a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of pyrimidoindazole production. By removing the dependency on precious metals such as palladium or rhodium, which are subject to volatile market pricing and supply constraints, manufacturers can achieve significant savings on input costs. Additionally, the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps, which are often the most resource-intensive parts of a synthetic sequence. This consolidation of steps reduces solvent consumption, energy usage for heating and cooling, and the manpower required for monitoring and handling multiple vessels. The cumulative effect of these efficiencies is a drastically simplified production process that lowers the overall cost of goods sold (COGS), allowing for more competitive pricing strategies in the marketplace without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, where delays can have cascading effects on drug development and patient access. This synthesis method relies on widely available and commercially mature starting materials, such as substituted benzaldehydes and 3-aminoindazoles, which are produced by numerous suppliers globally. This abundance reduces the risk of single-source bottlenecks and ensures that production can be scaled up rapidly in response to increased demand. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further enhancing reliability. For supply chain heads, this translates to reduced lead times for high-purity pharmaceutical intermediates and a greater ability to forecast inventory needs accurately, fostering stronger partnerships with downstream API manufacturers who depend on timely deliveries.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but the simplicity of this oxidative cyclization makes it highly amenable to commercial scale-up. The reaction operates at moderate temperatures and does not require high-pressure equipment or cryogenic conditions, allowing it to be run in standard multipurpose reactors found in most fine chemical facilities. From an environmental compliance standpoint, the absence of heavy metals simplifies wastewater treatment and effluent management, aligning with increasingly strict global environmental regulations. The reduction in chemical waste generation supports corporate sustainability goals and minimizes the ecological footprint of the manufacturing operation. This combination of scalability and environmental stewardship positions the technology as a future-proof solution for the long-term production of complex heterocyclic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of pyrimidoindazole compounds. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing supply chains and R&D pipelines.
Q: What are the key advantages of this pyrimidoindazole synthesis method?
A: The method described in patent CN109912606B offers a transition-metal-free, one-pot tandem reaction that avoids expensive catalysts and complex purification steps, significantly reducing production costs and environmental impact.
Q: What types of substituents are tolerated in this reaction?
A: The process demonstrates broad substrate scope, accommodating various substituted phenyl groups including methyl, tert-butyl, methoxy, halogens (F, Cl, Br, I), trifluoromethyl, and nitro groups, as well as naphthyl and heteroaryl moieties.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of simple raw materials, mild reaction conditions (110-150°C), and the elimination of transition metals make this protocol highly amenable to commercial scale-up and industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidoindazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN109912606B for the production of high-value pharmaceutical intermediates. Our team of expert process chemists is dedicated to translating such innovative academic and patent discoveries into robust, commercial-scale manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch. Whether you require custom synthesis of novel kinase inhibitor scaffolds or large-scale supply of established intermediates, our infrastructure is designed to meet the exacting demands of the global pharmaceutical industry.
We invite you to collaborate with us to leverage these technological advancements for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule and volume requirements. Our technical procurement team is ready to discuss your needs in detail and provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain. Contact us today to explore how our expertise in metal-free catalysis and green chemistry can drive efficiency and innovation in your drug development programs.
