Advanced One-Pot Synthesis of Pyrimidoindazole Compounds for Pharmaceutical Applications
Introduction to Next-Generation Pyrimidoindazole Synthesis
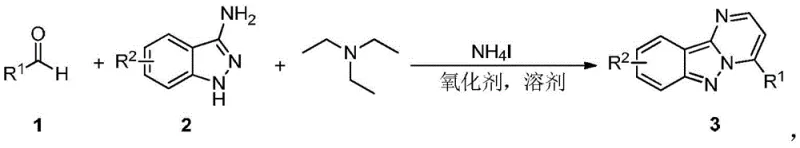
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds, particularly those exhibiting significant biological activity such as protein kinase inhibition. A groundbreaking approach detailed in patent CN109912606B introduces a highly efficient, transition metal-free methodology for constructing the pyrimidoindazole core. This innovation addresses critical bottlenecks in traditional organic synthesis by replacing complex multi-step sequences with a streamlined one-pot tandem reaction. By leveraging ammonium iodide (NH4I) as a catalyst in conjunction with simple oxidants, this technique achieves direct cyclization under mild thermal conditions. For R&D directors and process chemists, this represents a paradigm shift towards greener, more atom-economical manufacturing of high-value pharmaceutical intermediates. The ability to access these privileged structures without the burden of heavy metal contamination significantly simplifies downstream purification and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrimidoindazole derivatives has relied on cumbersome methodologies that pose significant challenges for commercial scale-up. Traditional routes often involve the condensation of 3-aminoindazoles with 1,3-dicarbonyl compounds or propiolates, which frequently necessitate harsh reaction conditions and corrosive reagents. Alternatively, Grieco condensation reactions or addition cyclizations with Michael acceptors have been employed, but these methods suffer from low atom economy and the generation of substantial chemical waste. A major drawback for procurement and supply chain teams is the reliance on expensive transition metal catalysts, which not only inflate raw material costs but also introduce stringent requirements for metal scavenging and residual analysis. These multi-step processes inherently increase the risk of yield loss at each stage and complicate the overall impurity profile, making the production of high-purity API intermediates both technically difficult and economically inefficient.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct oxidative cyclization strategy that merges simplicity with high efficiency. The core transformation involves the reaction of an aromatic aldehyde, a 3-aminoindazole derivative, and triethylamine in a suitable solvent, facilitated by NH4I and an oxidant. This one-pot series reaction operates effectively at temperatures between 110°C and 150°C, eliminating the need for cryogenic conditions or exotic reagents. The versatility of this system is demonstrated by its compatibility with a broad spectrum of oxidants, including di-tert-butyl peroxide (DTBP), dimethyl sulfoxide (DMSO), and even molecular oxygen or air. This flexibility allows manufacturers to optimize the process based on availability and cost, ensuring a reliable supply chain. Furthermore, the use of triethylamine as a promoter significantly reduces production costs compared to specialized bases, while the absence of transition metals streamlines the work-up procedure.
Mechanistic Insights into NH4I-Catalyzed Oxidative Cyclization
The mechanistic elegance of this synthesis lies in the dual role of ammonium iodide as both an iodine source and a catalyst for oxidative C-H functionalization. Under thermal conditions, the interaction between NH4I and the oxidant generates reactive iodine species in situ, which activate the aromatic aldehyde towards nucleophilic attack by the 3-aminoindazole. This initiates a cascade of condensation and cyclization events that construct the fused pyrimidoindazole ring system with high regioselectivity. The presence of triethylamine serves to neutralize acidic byproducts and facilitate the deprotonation steps essential for ring closure. For process chemists, understanding this mechanism is crucial for troubleshooting and optimization, as it highlights the importance of maintaining precise stoichiometric ratios—typically 1:1:2.5:1.5:3 for aldehyde, indazole, amine, NH4I, and oxidant respectively. This controlled environment minimizes side reactions and ensures consistent batch-to-batch reproducibility, a key metric for commercial scale-up of complex pharmaceutical intermediates.
Impurity control is inherently superior in this metal-free system compared to transition metal-catalyzed alternatives. Without palladium, copper, or iron residues, the risk of forming difficult-to-remove organometallic complexes is entirely eliminated. The primary byproducts are typically benign organic salts or reduced forms of the oxidant, which can be easily removed during standard aqueous work-up and silica gel chromatography. This results in a cleaner crude reaction profile, reducing the burden on purification units and increasing the overall throughput of the manufacturing line. The robustness of the reaction across various substrates, including electron-deficient and electron-rich aldehydes, further attests to the stability of the catalytic cycle. This reliability is paramount for producing high-purity pyrimidoindazole compounds required for preclinical and clinical studies, where strict impurity thresholds must be met.
How to Synthesize Pyrimidoindazole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The general protocol involves charging a reaction vessel with the aromatic aldehyde and 3-aminoindazole substrate, followed by the addition of triethylamine and a solvent such as chlorobenzene or toluene. Upon the introduction of ammonium iodide and the chosen oxidant, the mixture is heated to the target temperature, typically around 120°C, and stirred for approximately 12 hours. The reaction progress can be monitored via TLC or HPLC, with completion indicated by the consumption of the starting aldehyde. Following the reaction, a standard quench with water and extraction with ethyl acetate allows for the isolation of the crude product, which is then purified via column chromatography. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Dissolve aromatic aldehyde, 3-aminoindazole, and triethylamine in a solvent such as chlorobenzene.
- Add ammonium iodide (NH4I) and an oxidant (e.g., DTBP, DMSO, or air) to the reaction mixture.
- Heat the mixture to 110-150°C for approximately 12 hours, then quench and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. By shifting away from precious metal catalysis, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through the use of commodity chemicals like ammonium iodide and triethylamine. The elimination of expensive ligands and metal scavengers removes a significant cost center, while the simplified one-pot design reduces labor hours and equipment occupancy time. For supply chain heads, the reliance on readily available starting materials mitigates the risk of shortages associated with specialized reagents. The mild reaction conditions also enhance safety profiles, lowering insurance and compliance costs related to hazardous operations. Overall, this technology enables a more agile and cost-effective production model.
- Cost Reduction in Manufacturing: The substitution of transition metal catalysts with ammonium iodide drastically lowers raw material expenses. Additionally, the one-pot nature of the reaction consolidates multiple synthetic steps into a single operation, which significantly reduces solvent consumption, energy usage, and waste disposal costs. This streamlined workflow translates to substantial operational savings without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: The substrates required for this synthesis, such as benzaldehydes and aminoindazoles, are commercially abundant and stable, ensuring a consistent supply flow. The flexibility to use air or oxygen as an oxidant in certain variations further decouples the process from reliance on specific chemical suppliers. This robustness ensures reducing lead time for high-purity intermediates and maintains production continuity even during market fluctuations.
- Scalability and Environmental Compliance: The absence of toxic heavy metals simplifies the environmental permitting process and reduces the complexity of wastewater treatment. The reaction conditions are温和 (mild) enough to be safely scaled from gram to kilogram quantities without requiring specialized high-pressure equipment. This ease of scale-up facilitates rapid technology transfer from R&D to pilot and commercial plants, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on feasibility and application. Understanding these details helps stakeholders make informed decisions about integrating this route into their development pipelines.
Q: Does this synthesis method require transition metal catalysts?
A: No, the process described in patent CN109912606B is specifically designed to be transition metal-free, utilizing NH4I as the catalyst, which eliminates heavy metal residue concerns.
Q: What is the substrate scope for this pyrimidoindazole synthesis?
A: The method supports a wide range of aromatic aldehydes including substituted phenyls, naphthyls, pyridyls, and thienyls, as well as various 3-aminoindazole derivatives.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the one-pot nature, mild reaction conditions (110-150°C), and use of inexpensive reagents like triethylamine make it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidoindazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methodologies like the NH4I-catalyzed cyclization can be seamlessly translated to industrial volumes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrimidoindazole intermediate meets the highest global standards. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking reliable supply chains.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your manufacturing budget.
