Advancing Oncology Intermediates: Scalable Synthesis of N-N Axis Chiral Bisindoles via Organocatalysis
Advancing Oncology Intermediates: Scalable Synthesis of N-N Axis Chiral Bisindoles via Organocatalysis
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved therapeutic indices, particularly in the challenging field of oncology. Patent CN116082217B introduces a groundbreaking methodology for the asymmetric synthesis of N-N axis chiral bisindole compounds, a structural motif that has historically been underexplored compared to traditional N-aryl or C-aryl axial chiral indoles. This innovation addresses a critical gap in medicinal chemistry by providing a robust route to access these complex architectures with exceptional stereocontrol. The disclosed technology leverages advanced organocatalysis to construct the sterically hindered N-N bond, resulting in derivatives that demonstrate remarkable cytotoxic potency against human prostate cancer PC-3 cells. For R&D teams focused on expanding their oncology pipeline, this patent represents a significant leap forward in accessing privileged structures that were previously difficult to synthesize with high optical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of axially chiral indole compounds has relied heavily on transition metal catalysis or resolution of racemic mixtures, both of which present substantial drawbacks for large-scale manufacturing. Metal-catalyzed processes often suffer from issues related to residual metal contamination, which is strictly regulated in pharmaceutical intermediates and requires costly purification steps such as chromatography or specialized scavenging resins. Furthermore, conventional methods for constructing N-N bonds frequently struggle with low regioselectivity and poor enantiocontrol, leading to complex mixtures that are difficult to separate. The reliance on harsh reaction conditions, including high temperatures and strong bases, can also limit the functional group tolerance, thereby restricting the diversity of analogues that can be generated for structure-activity relationship (SAR) studies. These limitations collectively increase the cost of goods sold (COGS) and extend the lead time for bringing new drug candidates to preclinical evaluation.
The Novel Approach
The methodology described in CN116082217B offers a transformative solution by employing a chiral phosphoric acid catalyzed condensation reaction under mild conditions. This organocatalytic approach operates effectively at moderate temperatures around 70°C, utilizing 1,1,2,2-tetrachloroethane as a solvent and hexafluoroisopropanol as a crucial additive to enhance reactivity and selectivity. By avoiding transition metals entirely, the process inherently produces cleaner reaction profiles, significantly reducing the burden on downstream purification teams. The use of readily available indole-derived enamines and 2,3-diketone esters as starting materials ensures that the supply chain remains robust and cost-effective. This novel route not only achieves high yields but also delivers exceptional enantiomeric excess, often exceeding 90%, which is critical for ensuring the biological efficacy of the final drug substance. The simplicity and safety of this operation make it highly suitable for industrial scale-up, addressing the key pain points of conventional synthetic strategies.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
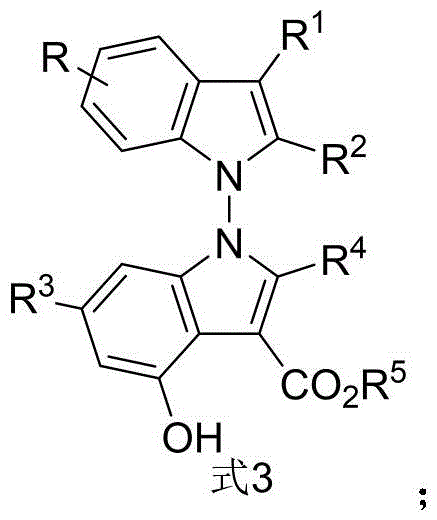
The success of this synthesis lies in the sophisticated activation mode provided by the chiral phosphoric acid catalyst, specifically the binaphthyl skeleton derivatives such as Formula 5a. The catalyst functions through a dual hydrogen-bonding mechanism, simultaneously activating the electrophilic carbonyl group of the 2,3-diketone ester and organizing the nucleophilic indole enamine within a well-defined chiral pocket. This precise spatial arrangement directs the approach of the reactants, ensuring that the new N-N bond is formed with the desired axial chirality. The addition of hexafluoroisopropanol plays a pivotal role by strengthening the hydrogen-bonding network and stabilizing the transition state, which is essential for achieving the observed high levels of stereoselectivity. Understanding this mechanistic nuance allows process chemists to fine-tune reaction parameters, such as catalyst loading and solvent choice, to optimize outcomes for different substrate combinations.
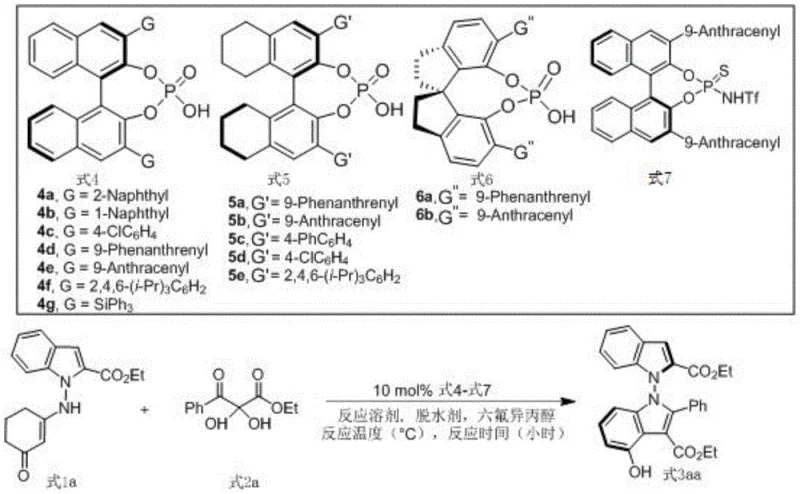
Impurity control is another critical aspect where this mechanistic understanding provides value. The mild nature of the organocatalytic cycle minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in indole chemistry. The specific choice of 5A molecular sieves as a dehydrating agent ensures that water generated during the condensation is efficiently removed, driving the equilibrium towards product formation without degrading sensitive functional groups. This results in a crude reaction mixture with a high purity profile, simplifying the isolation process. For quality control departments, this means fewer unknown impurities to identify and quantify, accelerating the regulatory filing process. The robustness of the catalyst system across a wide range of substrates, including those with electron-withdrawing or electron-donating groups, further demonstrates the reliability of this mechanistic framework for diverse chemical spaces.
How to Synthesize N-N Axis Chiral Bisindole Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for producing these high-value intermediates with consistent quality. The process begins with the precise weighing of indole-derived enamine and 2,3-diketone ester derivatives, which are then dissolved in the optimized solvent system. The addition of the chiral catalyst and additives must be performed under controlled conditions to ensure reproducibility. While the general procedure is straightforward, attention to detail regarding reaction time and temperature is paramount to achieving the reported high enantiomeric excess. The following section outlines the standardized steps derived from the optimal conditions identified in the patent examples, serving as a foundational guide for laboratory implementation.
- Combine indole-derived enamine (Formula 1) and 2,3-diketone ester derivative (Formula 2) in 1,1,2,2-tetrachloroethane solvent.
- Add chiral phosphoric acid catalyst (Formula 5a), 5A molecular sieves as dehydrating agent, and hexafluoroisopropanol additive.
- Stir the reaction mixture at 70°C for 48 hours, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The shift from metal-based catalysis to organocatalysis fundamentally alters the cost structure of the manufacturing process by eliminating the need for expensive precious metals and the associated validation of metal removal protocols. This transition not only reduces raw material costs but also shortens the production cycle time by streamlining the workup procedure. Furthermore, the use of commercially available starting materials mitigates supply chain risks, ensuring that production schedules are not disrupted by the scarcity of specialized reagents. The scalability of this method means that manufacturers can confidently plan for increased volumes without encountering the nonlinear cost increases often associated with complex chiral syntheses.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of the metal and the disposal of metal-contaminated waste streams. Additionally, the high atom economy of the reaction ensures that a greater proportion of the starting materials are converted into the desired product, minimizing waste generation. The simplified purification process, which relies on standard silica gel chromatography rather than specialized metal scavengers, further drives down operational expenses. These factors combine to create a more lean and efficient manufacturing process that enhances overall profit margins for the final API.
- Enhanced Supply Chain Reliability: The reliance on stable, off-the-shelf reagents such as indole enamines and diketone esters ensures a resilient supply chain that is less susceptible to geopolitical or market fluctuations. Unlike proprietary ligands or rare earth metals, these building blocks are produced by multiple global suppliers, fostering competition and price stability. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who demand strict adherence to delivery timelines.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Scaling this process from gram to kilogram quantities does not require significant re-engineering of the reactor setup, as the heat transfer and mixing requirements are manageable with standard equipment. The reduced environmental footprint facilitates easier permitting and compliance with local waste disposal laws, avoiding potential fines or shutdowns. This sustainability angle is increasingly important for pharmaceutical companies aiming to meet their corporate social responsibility goals while maintaining operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing development pipelines.
Q: What is the primary advantage of using chiral phosphoric acid for N-N axis construction?
A: Chiral phosphoric acids provide a highly effective hydrogen-bonding network that enables precise stereocontrol over the N-N axis formation, achieving enantiomeric excess values up to 96% without requiring toxic transition metals.
Q: How does this synthesis method impact downstream purification costs?
A: By utilizing an organocatalytic system instead of heavy metal catalysts, the process eliminates the need for expensive and complex metal scavenging steps, significantly simplifying the purification workflow and reducing overall production costs.
Q: What is the biological significance of these N-N axis chiral bisindoles?
A: These compounds exhibit potent cytotoxic activity against human prostate cancer PC-3 cells, with IC50 values demonstrating high sensitivity, making them valuable candidates for developing next-generation anticancer therapeutics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bisindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral intermediates in the development of next-generation oncology therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from discovery to market. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity and optical purity. By partnering with us, you gain access to a supply chain that prioritizes consistency, compliance, and technical excellence.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current route or need to evaluate the feasibility of this novel organocatalytic method, we are ready to assist. Please reach out to request specific COA data and route feasibility assessments tailored to your target molecules, and let us help you accelerate your drug development timeline with confidence.
