Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
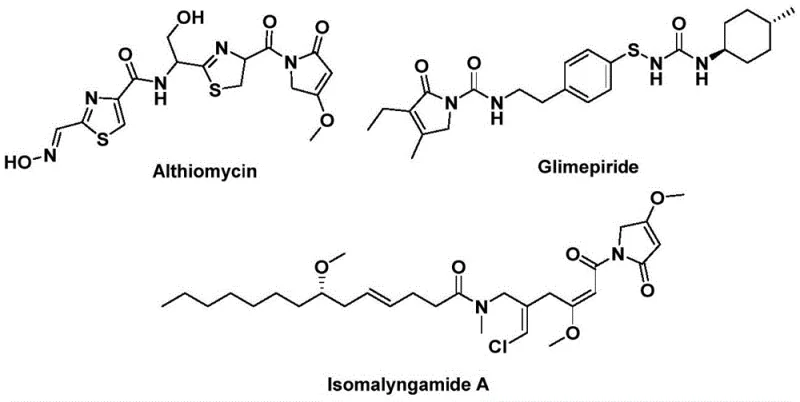
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and therapeutic agents. As illustrated in the biological context, compounds such as Althiomycin exhibit potent antibacterial properties, while Glimepiride functions as a critical hypoglycemic agent, and Isomalyngamide A demonstrates promising anticancer activity. Recognizing the immense pharmacological potential of this heterocyclic system, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargylamine and benzyl chloride derivatives as accessible starting materials to construct the five-membered lactam ring with remarkable efficiency. By shifting away from complex multi-step sequences, this technology offers a streamlined pathway that enhances both atom economy and operational simplicity, positioning it as a vital tool for modern pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied on classical cyclization reactions that often suffer from significant drawbacks regarding step count and reagent availability. Traditional routes frequently necessitate the use of pre-functionalized cyclic precursors or require harsh reaction conditions that limit substrate scope and functional group tolerance. Furthermore, many conventional carbonylation methods depend on the direct use of gaseous carbon monoxide, which poses severe safety hazards and logistical challenges for scale-up in standard laboratory or pilot plant settings. These limitations result in prolonged development timelines, increased waste generation, and higher overall production costs, creating a bottleneck for the rapid synthesis of diverse analog libraries required for structure-activity relationship (SAR) studies in drug discovery programs.
The Novel Approach
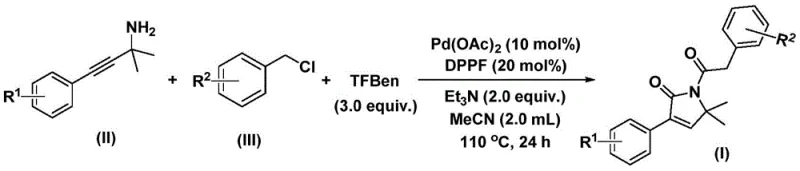
In stark contrast, the methodology disclosed in the patent introduces a robust one-pot synthesis that elegantly bypasses these obstacles through the use of a solid carbon monoxide substitute and a highly active palladium catalyst system. By employing benzyl chlorides and propargylamines as simple, commodity-grade feedstocks, the process achieves the formation of two carbon-carbon bonds and one carbon-nitrogen bond in a single operational sequence. This bis-carbonylation cascade not only simplifies the synthetic design but also dramatically improves the reaction efficiency, allowing for the rapid assembly of complex molecular architectures. The use of a solid CO source eliminates the need for specialized high-pressure equipment for gas handling, thereby enhancing the safety profile and making the technology inherently more suitable for commercial scale-up and continuous flow applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle underpinning this transformation is a sophisticated example of transition metal-mediated C-H functionalization and carbonylative coupling. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzylpalladium(II) intermediate. Subsequently, carbon monoxide, which is released in situ from the decomposition of the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acylpalladium species. This electrophilic acyl complex then undergoes nucleophilic attack by the propargylamine, followed by a second carbonyl insertion event that extends the carbon chain and sets the stage for cyclization. The final step involves an intramolecular reductive elimination that closes the five-membered pyrrol-2-one ring and regenerates the active palladium catalyst, completing the cycle with high turnover frequency.
From an impurity control perspective, the mechanism offers distinct advantages due to the high chemoselectivity of the palladium catalyst towards the specific activation of the benzyl chloride and alkyne moieties. The use of 1,1'-bis(diphenylphosphino)ferrocene (DPPF) as a bidentate ligand stabilizes the palladium center, preventing the formation of inactive palladium black and minimizing side reactions such as homocoupling of the alkyne or premature hydrolysis of the acyl intermediate. Furthermore, the controlled release of carbon monoxide from the solid surrogate ensures a steady, low concentration of CO in the reaction medium, which suppresses the formation of dicarbonylation byproducts or over-carbonylated species. This precise kinetic control results in a clean reaction profile, facilitating downstream purification and ensuring the delivery of high-purity intermediates that meet stringent pharmaceutical quality standards.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The experimental protocol outlined in the patent provides a clear and reproducible roadmap for executing this transformation with optimal results. The procedure involves charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and substrates in acetonitrile, followed by heating to promote the cascade reaction. The choice of acetonitrile as the solvent is critical, as it provides the necessary polarity to dissolve the ionic intermediates while maintaining thermal stability at the required reaction temperature of 110°C. For researchers aiming to implement this chemistry, adherence to the specified molar ratios and reaction times is essential to maximize yield and minimize the formation of trace impurities. Detailed standardized synthesis steps are provided in the guide below to ensure consistent batch-to-batch reproducibility.
- Combine palladium acetate, DPPP ligand, triethylamine, solid CO source (TFBen), propargylamine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition driven by raw material accessibility and process safety. The reliance on benzyl chlorides and propargylamines, which are bulk chemicals produced on a massive global scale, insulates the supply chain from the volatility associated with exotic or custom-synthesized reagents. This stability in sourcing translates directly into more predictable lead times and reduced risk of production stoppages due to material shortages. Additionally, the elimination of gaseous carbon monoxide removes a significant regulatory and safety burden, lowering the barrier for manufacturing in facilities that may not be equipped for high-pressure gas handling.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the convergence of multiple synthetic steps into a single pot, which drastically reduces solvent consumption, labor hours, and waste disposal costs. By avoiding the use of expensive pre-functionalized cyclic starting materials and replacing hazardous gas cylinders with a stable solid powder, the overall cost of goods sold (COGS) is significantly optimized. The high reaction efficiency reported in the patent data suggests that minimal raw material is wasted, further enhancing the cost-effectiveness of the route for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in substrate electronics ensures that a wide range of derivatives can be produced using the same standard operating procedure. This flexibility allows manufacturers to respond rapidly to changing demand for different analogs without requiring extensive process re-validation or retooling. The use of common organic solvents and standard heating equipment means that the process can be easily transferred between different manufacturing sites, ensuring continuity of supply even in the face of regional disruptions.
- Scalability and Environmental Compliance: From an environmental standpoint, the atom-economical nature of the carbonylation reaction minimizes the generation of stoichiometric byproducts, aligning with green chemistry principles. The simplified workup procedure, which involves basic filtration and chromatography, reduces the volume of aqueous waste streams typically associated with acidic or basic quenching steps in traditional methods. This cleaner profile facilitates easier compliance with increasingly strict environmental regulations, making the process sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed methodology. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, providing clarity for R&D teams evaluating this technology for their pipelines. Understanding these nuances is crucial for successful technology transfer and process optimization.
Q: What are the primary advantages of this carbonylation method over traditional cyclization routes?
A: This method utilizes a one-pot palladium-catalyzed bis-carbonylation strategy that eliminates the need for pre-functionalized cyclic precursors. It operates under relatively mild thermal conditions (100-120°C) using commercially available benzyl chlorides and propargylamines, significantly simplifying the synthetic workflow compared to multi-step traditional approaches.
Q: How does the process handle substrate diversity for pharmaceutical applications?
A: The catalytic system demonstrates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing substituents such as halogens and trifluoromethyl groups. This broad compatibility allows for the rapid generation of diverse libraries of bioactive pyrrol-2-one scaffolds essential for drug discovery.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the protocol relies on inexpensive and readily available starting materials like benzyl chloride and uses a solid carbon monoxide substitute, avoiding the safety hazards associated with handling gaseous CO. The straightforward workup involving filtration and chromatography supports scalable manufacturing processes for high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in accelerating drug development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-one derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed carbonylations allows us to troubleshoot complex reaction parameters and optimize yields for your specific target molecules.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data for reference standards and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain.
