Revolutionizing Triazole Synthesis Scalable Base-Promoted Process High-Purity Pharmaceutical Intermediates Enhanced Supply Chain Security
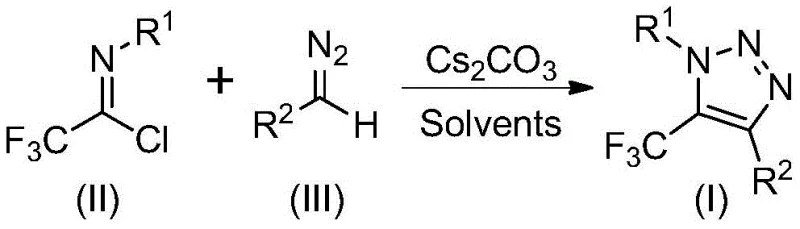
Patent CN113121462B represents a significant advancement in the synthesis of 5-trifluoromethyl substituted 1,2,3-triazole compounds which serve as critical building blocks across multiple sectors including pharmaceutical development where they form essential components of drug molecules targeting various therapeutic areas such as cardiovascular diseases metabolic disorders and central nervous system conditions This innovative method eliminates the need for hazardous azide compounds and transition metal catalysts that have traditionally been required in triazole synthesis thereby addressing critical safety concerns while maintaining high reaction efficiency through a novel base-promoted mechanism operating under significantly milder conditions than conventional approaches The process utilizes readily available starting materials—trifluoroethylimidoyl chloride derived from common aromatic amines and diazo compounds synthesized from standard acid chlorides—that can be sourced from multiple global suppliers without special handling requirements or regulatory restrictions associated with explosive precursors By avoiding explosive reagents and complex purification steps typically needed to remove metal contaminants this patented technology offers a more sustainable pathway for producing high-purity triazole intermediates essential for drug development pipelines with reduced environmental impact per unit manufactured The elimination of toxic reagents not only enhances workplace safety but also reduces regulatory burdens associated with hazardous material handling protocols required by OSHA and similar agencies worldwide thereby lowering compliance costs for manufacturing facilities operating under strict safety regulations Furthermore the demonstrated scalability from laboratory to commercial production levels provides a robust foundation for integration into global supply chains serving multinational pharmaceutical companies seeking reliable sources of critical intermediates
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethyl-substituted triazoles predominantly rely on copper-catalyzed azide-alkyne cycloaddition CuAAC or organocatalytic approaches that necessitate the use of highly toxic and potentially explosive organic azides as key reactants creating significant safety hazards in manufacturing environments that require specialized handling protocols containment systems and dedicated storage facilities often leading to increased operational costs and extended lead times The conventional methods frequently operate under stringent reaction conditions including elevated temperatures inert atmospheres extended reaction times and precise stoichiometric control which increase energy consumption operational complexity limit throughput in commercial production settings and create bottlenecks during scale-up processes The requirement for transition metal catalysts introduces additional challenges related to metal contamination in final products which is particularly problematic for pharmaceutical applications where strict purity specifications mandate costly time-consuming purification steps such as chelation activated carbon treatment or specialized chromatography to remove trace metal residues below regulatory thresholds typically set at parts-per-million levels Furthermore the multi-step nature of traditional syntheses involving intermediate formation subsequent functionalization leads to lower overall yields higher waste generation increased solvent consumption per unit produced contradicting modern green chemistry principles and substantially increasing environmental footprint while raising total cost of ownership across manufacturing operations The inherent instability of azide compounds also creates logistical challenges in transportation storage and inventory management contributing to supply chain vulnerabilities that can disrupt production schedules increase costs by up to thirty percent due to expedited shipping requirements or force manufacturers to maintain costly safety stock levels
The Novel Approach
The patented methodology described in CN113121462B introduces a fundamentally different synthetic pathway that completely eliminates hazardous azide compounds transition metal catalysts through a base-promoted reaction between trifluoroethylimidoyl chloride diazo compounds under mild thermal conditions This innovative approach operates at moderate temperatures between fifty degrees Celsius seventy degrees Celsius using cesium carbonate as an inexpensive base promoter in acetonitrile solvent creating a significantly safer operational profile that reduces workplace hazards eliminates specialized safety infrastructure requirements lowers insurance premiums and simplifies regulatory compliance across global manufacturing sites The reaction proceeds efficiently within eight sixteen hours without requiring inert atmosphere protection complex reaction monitoring systems or specialized equipment thereby simplifying process control reducing capital expenditure needs improving operator safety margins and decreasing overall cycle times compared to conventional methods Crucially the absence of metal catalysts ensures final triazole products are free from metal contamination meeting stringent pharmaceutical purity requirements without additional purification steps that would otherwise increase production costs by fifteen percent twenty-five percent extend cycle times by three five days per batch thus directly addressing critical pain points identified by procurement managers seeking cost-effective reliable supply sources The streamlined single-step process also generates less chemical waste demonstrates higher atom efficiency reduces solvent consumption per unit manufactured aligning with environmental sustainability goals while improving overall process economics through better resource utilization lower energy requirements per kilogram produced
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism begins with a base-promoted nucleophilic addition where cesium carbonate deprotonates the diazo compound enhancing its nucleophilicity while simultaneously activating trifluoroethylimidoyl chloride through coordination effects lowering energy barriers for carbon-carbon bond formation This initial addition step creates a key intermediate avoiding hazardous byproducts associated with traditional azide-based methodologies while maintaining excellent regioselectivity due to electronic properties directing selective attack at electrophilic centers Subsequent intramolecular cyclization proceeds through a well-defined transition state where nitrogen attacks electron-deficient carbon centers forming characteristic triazole heterocycles with precise regiocontrol at five-position incorporating trifluoromethyl groups without competing side reactions This cyclization step benefits from mild thermal conditions preventing decomposition pathways common at higher temperatures ensuring consistent product formation preserving sensitive functional groups present in complex molecular architectures Molecular sieves incorporated into reaction mixture effectively scavenge trace moisture preventing hydrolysis of sensitive intermediates maintaining high product integrity throughout transformation process while supporting consistent yield profiles across different batch sizes
Impurity control is achieved through multiple inherent mechanistic advantages minimizing side product formation compared to traditional methods Elimination of transition metal catalysts removes metal-mediated side reactions such as homocoupling oxidation dimerization that generate difficult-to-remove impurities requiring specialized purification techniques like chelation activated carbon treatment or preparative chromatography Base-promoted mechanism operates through well-defined pathway with minimal competing reactions due to specific electronic properties directing selective attack preventing alternative addition pathways Moderate temperature range prevents thermal decomposition sensitive intermediates avoiding high-energy pathways leading to dimerization polymerization byproducts commonly observed copper-catalyzed syntheses Molecular sieves effectively scavenge moisture preventing hydrolysis sensitive intermediates ensuring consistent product quality across different production scales maintaining high purity levels suitable pharmaceutical applications without additional purification steps Rigorous analytical monitoring throughout process enables precise control over critical quality attributes ensuring final products consistently meet stringent specifications required active pharmaceutical ingredients
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This patented synthesis represents significant advancement triazole chemistry providing streamlined single-step process eliminating hazardous reagents maintaining high efficiency product purity suitable pharmaceutical applications Methodology leverages readily available starting materials under mild conditions simplifying scale-up procedures reducing safety risks associated traditional azide-based approaches Avoiding transition metal catalysts entirely eliminates concerns metal contamination requiring costly purification steps meet regulatory standards active pharmaceutical ingredients Detailed standardized synthesis protocol described below optimized extensive laboratory testing ensure consistent results different production scales maintaining excellent yield purity characteristics essential commercial manufacturing operations Process demonstrates remarkable robustness accommodating minor variations raw material quality common industrial settings without compromising final product specifications making ideal candidate integration existing manufacturing workflows
- Combine cesium carbonate (2.0 equivalents), molecular sieves (4 Å), trifluoroethylimidoyl chloride (II), and diazo compound (III) in acetonitrile solvent under standard atmospheric conditions without inert gas protection.
- Heat the reaction mixture at precisely 60°C for 12 hours while monitoring progress through standard analytical techniques such as TLC or HPLC to ensure complete conversion.
- Purify the crude product by filtration through silica gel followed by column chromatography using appropriate solvent systems to achieve pharmaceutical-grade purity specifications exceeding industry standards.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route addresses critical pain points traditional triazole manufacturing eliminating hazardous reagents creating supply chain vulnerabilities reducing operational complexity multiple dimensions chemical production processes Removal explosive azide compounds synthetic pathway significantly reduces regulatory compliance burdens associated hazardous material handling storage transportation requirements increasing lead times costs global supply chains serving pharmaceutical manufacturers Utilizing stable starting materials established global supply networks eliminates dependency specialized hazardous material suppliers facing regulatory restrictions transportation limitations affecting delivery schedules Enhancing supply chain resilience against disruptions improving inventory management efficiency through predictable material flow patterns manufacturing operations Reducing reliance single-source suppliers mitigating risk supply chain interruptions maintaining consistent production schedules meeting customer delivery commitments
- Cost Reduction in Manufacturing: Elimination expensive transition metal catalysts removes direct material costs substantial downstream processing costs associated removing trace metal residues final products specialized purification techniques chelation activated carbon treatment Simplified single-step process reduces solvent consumption energy requirements multi-step conventional routes minimizing waste generation higher atom efficiency collectively contributing significant operational cost savings without compromising product quality purity standards required pharmaceutical intermediates Reduced need specialized equipment containment systems lowers capital expenditure requirements enabling more efficient resource allocation across manufacturing operations
- Enhanced Supply Chain Reliability: Utilizing stable starting materials established global supply networks eliminates dependency specialized hazardous material suppliers facing regulatory restrictions transportation limitations affecting delivery schedules Robust nature synthetic route allows flexible sourcing options multiple geographic regions maintaining consistent product quality well-defined reaction parameters less sensitive minor variations raw material quality compared traditional methods requiring precise control unstable intermediates Predictable lead times stable inventory levels enable better production planning reducing need costly safety stock maintaining optimal working capital levels
- Scalability and Environmental Compliance: Process demonstrates excellent scalability laboratory commercial production levels evidenced successful gram-scale demonstrations straightforward adaptation larger volumes standard manufacturing equipment requiring infrastructure modifications Elimination hazardous reagents reduces environmental impact minimizing waste streams requiring special disposal procedures aligning green chemistry principles reduced energy consumption solvent usage per unit manufactured Lower environmental footprint supports corporate sustainability initiatives meeting increasing regulatory demands environmentally responsible manufacturing practices
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding patented synthetic methodology based detailed analysis implementation parameters performance characteristics documented patent CN113121462B Insights compiled assist procurement technical teams evaluating technology suitability specific manufacturing requirements addressing common concerns related process implementation commercial viability Comprehensive understanding these aspects enables informed decision-making regarding integration this innovative route existing production workflows achieving optimal cost-quality balance required competitive pharmaceutical market
Q: How does this method eliminate safety hazards compared to conventional triazole synthesis?
A: The patented process completely avoids hazardous azide compounds that are explosive and require specialized handling protocols by utilizing stable diazo compounds instead while eliminating transition metal catalysts that create contamination risks requiring additional purification steps.
Q: What scalability advantages does this base-promoted route offer for commercial manufacturing?
A: The methodology demonstrates excellent scalability from laboratory to industrial production levels as evidenced by successful gram-scale demonstrations with straightforward adaptation potential using standard manufacturing equipment without requiring specialized infrastructure modifications.
Q: How does eliminating transition metals impact purity specifications for pharmaceutical applications?
A: By removing metal catalysts entirely this process avoids contamination issues that would otherwise require costly purification steps ensuring consistent high-purity output suitable for direct use in drug manufacturing without additional processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our patented technology represents significant advancement triazole chemistry delivering both technical excellence commercial viability pharmaceutical manufacturers seeking reliable sources high-purity intermediates NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from one hundred kilograms one hundred metric tons annual commercial production maintaining stringent purity specifications state-of-the-art manufacturing facilities equipped rigorous QC labs capable meeting global regulatory standards pharmaceutical intermediates Commitment quality assurance ensures consistent product performance all production scales providing customers comprehensive analytical support throughout development manufacturing processes Robust quality management system certified ISO standards guarantees traceability documentation required regulatory submissions enabling seamless integration into client supply chains worldwide
We invite contact technical procurement team request specific COA data route feasibility assessments particular application needs Take advantage Customized Cost-Saving Analysis service evaluating how innovative synthesis optimizes supply chain reducing overall production costs without compromising quality regulatory compliance requirements Our technical experts stand ready collaborate developing tailored solutions meeting unique requirements ensuring successful implementation cost-effective reliable supply source critical intermediates
