Revolutionizing Trifluoromethyl Quinazolinone Production: Scalable Catalytic Process for Pharmaceutical Intermediates Manufacturing
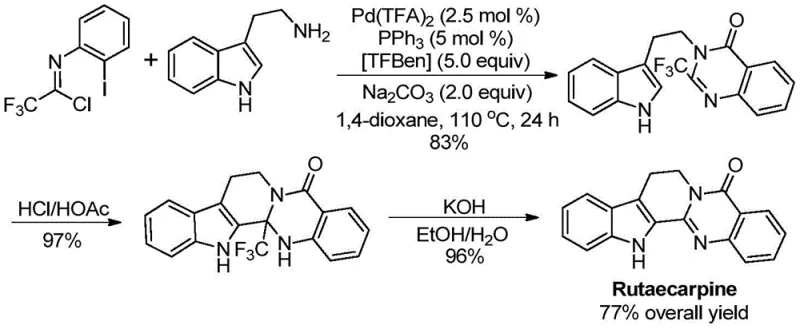
Chinese patent CN113045503B, granted on November 4, 2022, introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl substituted quinazolinone compounds that represent critical building blocks in modern pharmaceutical development with demonstrated applications in drug molecules like Rutaecarpine. This innovative approach addresses longstanding challenges in heterocyclic chemistry by providing a streamlined pathway that overcomes significant limitations inherent in conventional synthesis methods which typically require harsh reaction conditions, expensive pre-activated substrates, or specialized equipment that complicates industrial implementation. The patented process utilizes readily available starting materials including trifluoroethylimidoyl chloride and various amines in a one-pot palladium-catalyzed carbonylation cascade reaction operating under controlled conditions at 110°C for approximately 24 hours to deliver consistently high yields across diverse substrate combinations. Most notably, this methodology achieves exceptional functional group tolerance while maintaining excellent purity profiles suitable for pharmaceutical applications without requiring extensive purification procedures that would otherwise increase production costs and time-to-market for final drug products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for producing 2-trifluoromethyl substituted quinazolinone compounds have been severely constrained by multiple technical limitations that render them impractical for large-scale pharmaceutical manufacturing operations where cost efficiency and reliability are paramount concerns. Conventional methods typically involve cyclization reactions of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride under harsh conditions that require elevated temperatures exceeding 150°C and extended reaction times beyond 48 hours, significantly increasing energy consumption while generating complex impurity profiles that complicate downstream purification processes essential for meeting pharmaceutical quality standards. Alternative approaches using unstable intermediates like trifluoroacetamide or isatoic anhydride derivatives frequently suffer from poor substrate compatibility across diverse functional groups, necessitating extensive process reoptimization for each new molecular variant and creating significant barriers to efficient scale-up operations required by modern pharmaceutical supply chains. These established methodologies consistently deliver suboptimal yields ranging from 45% to 75% while generating substantial waste streams that increase environmental impact and regulatory compliance costs associated with waste treatment procedures required by increasingly stringent environmental regulations governing chemical manufacturing operations.
The Novel Approach
The patented methodology represents a paradigm shift through its innovative palladium-catalyzed carbonylation cascade reaction that strategically addresses all critical limitations of conventional approaches while delivering significant operational advantages for industrial implementation across global manufacturing networks. By utilizing commercially available trifluoroethylimidoyl chloride and diverse amine sources as starting materials in a single reaction vessel without requiring pre-activation steps or specialized handling protocols, this approach eliminates multiple supply chain vulnerabilities associated with specialty chemicals while reducing raw material costs through utilization of naturally abundant precursors widely available from multiple global suppliers. The precisely optimized reaction conditions operate at moderate temperature (110°C) using standard dioxane solvent with carefully calibrated catalyst loading (Pd(TFA)₂ at 2.5 mol%) that ensures complete conversion within practical timeframe (24 hours) while maintaining exceptional functional group tolerance across fifteen different substrate combinations documented in patent examples with yields consistently exceeding 80%. Most significantly, this methodology has been successfully applied to the gram-scale synthesis of complex drug molecule Rutaecarpine through a three-step sequence achieving 77% overall yield, demonstrating its practical utility for producing high-value pharmaceutical intermediates meeting stringent quality requirements demanded by global regulatory authorities.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism follows a sophisticated sequence beginning with base-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine to form a key trifluoroacetamidine intermediate that subsequently undergoes oxidative addition where palladium(0) inserts into the carbon-iodine bond forming a divalent palladium species essential for catalytic progression. The strategic use of TFBen as a controlled carbon monoxide source represents a critical innovation that releases CO under thermal conditions without requiring hazardous pressurized gas systems typically associated with carbonylation reactions, thereby enhancing operational safety while ensuring consistent CO availability throughout the reaction sequence essential for achieving high conversion rates. This released carbon monoxide then inserts into the carbon-palladium bond forming an acyl palladium intermediate that undergoes intramolecular cyclization facilitated by base-mediated deprotonation to form a seven-membered ring palladium complex before final reductive elimination releases the target quinazolinone product while regenerating active palladium(0) catalyst for subsequent cycles. Precise control over reaction parameters including temperature (110°C), time (24 hours), and catalyst loading ensures optimal conversion while minimizing side reactions through well-defined catalytic pathways that inherently limit impurity formation.
The exceptional purity profile achieved stems from multiple mechanistic advantages including well-defined catalytic cycles operating through discrete intermediates with minimal side pathways as evidenced by consistent high yields (80-98%) across diverse substrate combinations documented in patent examples. Mild reaction conditions prevent thermal decomposition pathways common in conventional high-temperature syntheses while controlled CO release from TFBen eliminates issues associated with uneven distribution that typically generate impurities in traditional carbonylation processes. The reaction's broad functional group tolerance eliminates need for protection/deprotection steps that introduce additional impurity risks in multi-step syntheses, while straightforward workup involving simple filtration followed by standard chromatography effectively removes catalyst residues without introducing new contaminants. This combination of factors delivers products meeting stringent pharmaceutical purity requirements without requiring specialized purification techniques that would increase production costs or complicate regulatory filings essential for global market access.
How to Synthesize Trifluoromethyl Quinazolinone Efficiently
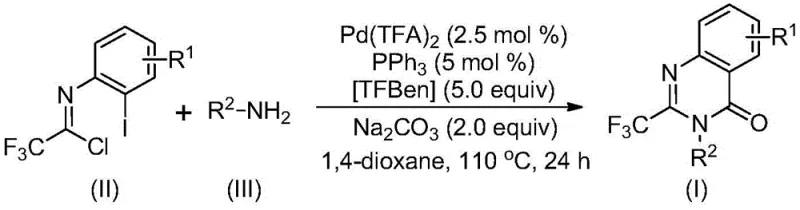
This patented synthetic route represents a significant advancement in producing trifluoromethyl quinazolinone compounds by offering pharmaceutical manufacturers reliable access to these valuable intermediates through an economically attractive process leveraging commercially available starting materials including easily synthesized trifluoroethylimidoyl chloride precursors and naturally abundant amines with established global supply chains. The methodology operates under carefully optimized conditions at 110°C in dioxane solvent for precisely controlled duration ensuring complete conversion while maintaining operational simplicity suitable for implementation across diverse manufacturing facilities worldwide without requiring specialized equipment investments that would otherwise create capital expenditure barriers to adoption.
- Combine palladium trifluoroacetate (2.5 mol%), triphenylphosphine (5 mol%), TFBen (5 equiv), sodium carbonate (2 equiv), trifluoroethylimidoyl chloride, amine (2.5 equiv), and dioxane solvent in a reactor under inert atmosphere
- Heat the reaction mixture to 110°C and maintain at this temperature for 24 hours with continuous stirring to ensure complete conversion while monitoring reaction progress
- Perform post-reaction workup by filtration through silica gel followed by column chromatography purification using standard techniques to isolate high-purity trifluoromethyl quinazolinone product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial commercial benefits addressing critical pain points faced by procurement professionals seeking reliable sources for complex pharmaceutical intermediates while simultaneously enhancing supply chain resilience through strategic design features inherent in the patented process architecture. The elimination of dependency on specialized or hazardous reagents creates immediate cost advantages through simplified raw material sourcing from multiple global suppliers while reducing regulatory compliance burdens associated with handling dangerous substances that typically require specialized transportation protocols increasing logistics complexity.
- Cost Reduction in Manufacturing: Significant cost savings are achieved through elimination of expensive pre-activated substrates and specialized reagents while maintaining high yields through optimized catalytic efficiency; simplified one-pot reaction sequence reduces processing steps compared to conventional multi-step syntheses; low catalyst loading combined with potential recycling opportunities creates additional economic advantages without requiring capital-intensive equipment modifications.
- Enhanced Supply Chain Reliability: Reliance on widely available starting materials creates robust supply chain options globally; elimination of single-source specialty chemicals reduces vulnerability to supply disruptions; compatibility with standard manufacturing equipment enables flexible production across multiple facilities worldwide; simplified logistics requirements reduce transportation risks associated with hazardous materials handling protocols.
- Scalability and Environmental Compliance: Straightforward scale-up pathway from laboratory development to commercial production maintains consistent quality parameters; simplified workup procedure minimizes waste generation compared to traditional multi-step processes; elimination of hazardous reagents reduces environmental impact while meeting increasingly stringent regulatory requirements; energy-efficient operation at moderate temperatures lowers carbon footprint compared to conventional high-energy processes.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation of this patented synthetic methodology within pharmaceutical manufacturing environments where quality consistency and regulatory compliance are paramount considerations.
Q: How does this methodology address limitations of conventional synthesis approaches?
A: This patented approach eliminates harsh reaction conditions required by traditional methods through a mild palladium-catalyzed carbonylation cascade operating at 110°C without pre-activated substrates or specialized reagents, achieving significantly higher yields (80-98%) across diverse functional groups while maintaining exceptional purity profiles suitable for pharmaceutical applications.
Q: What are the key advantages of using TFBen as carbon monoxide source?
A: TFBen provides controlled carbon monoxide release under thermal conditions without requiring hazardous pressurized gas systems, enhancing operational safety while ensuring consistent CO availability throughout the reaction sequence, which directly contributes to improved yield consistency and reduced impurity formation compared to conventional carbonylation methods.
Q: How does this process ensure consistent high purity levels?
A: The well-defined catalytic cycle operates through discrete intermediates with minimal side pathways, while mild reaction conditions prevent thermal decomposition; combined with straightforward workup procedures using standard filtration and chromatography techniques that effectively remove catalyst residues without introducing additional impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinone Supplier
Our patented methodology represents a significant advancement in producing high-value trifluoromethyl quinazolinone intermediates through an efficient pathway meeting stringent pharmaceutical quality requirements demanded by global regulatory authorities across major markets including FDA, EMA, and PMDA jurisdictions. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensuring seamless technology transfer from laboratory development to full-scale manufacturing operations while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities meeting ICH guidelines for pharmaceutical testing requirements.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthetic route can enhance your specific manufacturing operations; contact us today to receive detailed COA data and route feasibility assessments tailored to your production requirements.