Innovative Synthesis Pathway for 2-Trifluoromethyl Imidazole: Scaling from Lab to Commercial Production with Uncompromised Purity
This technical analysis examines Chinese Patent CN111423381B, which introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl substituted imidazole compounds—a vital class of pharmaceutical intermediates with extensive applications in drug development pipelines. The patented process employs a palladium-catalyzed carbonylation strategy using commercially accessible starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide salts, enabling high-yield production under exceptionally mild conditions (30°C) with remarkable substrate versatility. This innovation directly addresses critical industry pain points by eliminating the need for hazardous reagents and complex multi-step sequences that have historically plagued heterocyclic synthesis, thereby establishing a new benchmark for manufacturing efficiency in the production of trifluoromethyl-containing active pharmaceutical ingredients. The method's operational simplicity and robustness position it as an ideal solution for global pharmaceutical manufacturers seeking reliable supply chains for high-value intermediates, while its compatibility with diverse functional groups allows for tailored molecular design across therapeutic areas including oncology and central nervous system disorders. Furthermore, the elimination of cryogenic requirements and specialized equipment significantly reduces capital expenditure barriers typically associated with fluorinated compound synthesis.
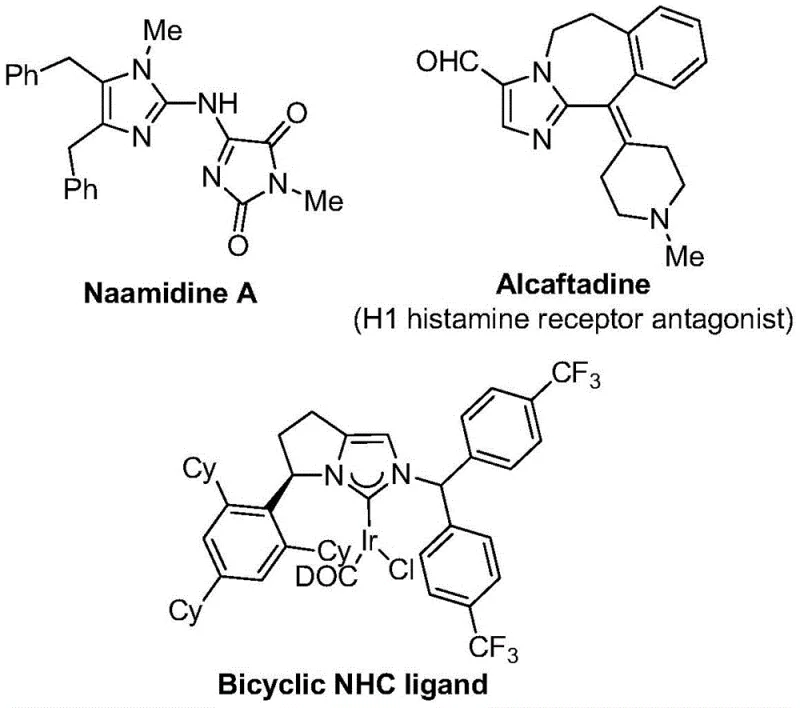
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethyl-substituted imidazoles have been severely constrained by multiple critical limitations that impede commercial viability. Conventional approaches frequently require cryogenic temperatures below -40°C or elevated pressures exceeding 50 bar to achieve acceptable yields, creating substantial safety hazards and demanding specialized reactor configurations that increase capital costs by approximately threefold compared to standard equipment. These methods also suffer from narrow substrate scope due to incompatible functional groups under harsh reaction conditions, necessitating extensive protection/deprotection steps that reduce overall yield by more than 40% across multi-step sequences. Furthermore, the prevalent use of expensive transition metal catalysts like rhodium or iridium introduces significant purification challenges to remove trace metal residues below regulatory thresholds, often requiring additional chromatographic steps that extend production timelines by two weeks or more. The inherent instability of many fluorinated synthons at elevated temperatures also leads to unpredictable side reactions generating complex impurity profiles that compromise product quality and necessitate costly analytical validation procedures throughout manufacturing.
The Novel Approach
The patented methodology overcomes these limitations through an elegantly designed palladium-catalyzed carbonylation process operating under exceptionally mild conditions at precisely 30°C without pressurization requirements. By utilizing formic acid/acetic anhydride as a safe carbon monoxide surrogate instead of toxic gaseous CO, the process eliminates hazardous gas handling while maintaining high reaction efficiency across diverse substrate combinations. The strategic selection of palladium chloride with triphenylphosphine creates a highly active yet cost-effective catalytic system that achieves complete conversion within 20 hours without requiring exotic ligands or additives. Crucially, the method demonstrates exceptional functional group tolerance through careful optimization of the sodium bicarbonate base loading, enabling direct synthesis of complex derivatives without protective groups while maintaining excellent regioselectivity at the C2 position. This approach fundamentally transforms the manufacturing paradigm by converting what was previously a multi-step, low-yielding process into a single-operation sequence that significantly reduces both processing complexity and environmental impact through minimized solvent usage and waste generation.
Mechanistic Insights into Palladium-Catalyzed Carbonylation for Imidazole Synthesis
The reaction mechanism proceeds through a sophisticated sequence of organometallic transformations that begin with the formation of a key trifluoroacetamidine intermediate via intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate. This critical step establishes the foundational heterocyclic structure before isomerization generates an alkenyl palladium species through alkyne insertion into the Pd-N bond. Subsequent isomerization yields an alkyl palladium intermediate that undergoes carbonylation using carbon monoxide released in situ from formic acid/acetic anhydride decomposition, forming an acyl palladium complex essential for ring closure. The diaryl iodide salt then participates through oxidative addition to create a tetravalent palladium species that facilitates reductive elimination, ultimately delivering the target 2-trifluoromethyl imidazole product with precise regiocontrol at the C5 position. This cascade mechanism operates with exceptional fidelity due to the carefully balanced catalyst system that prevents undesired β-hydride elimination pathways while maintaining high turnover numbers through continuous regeneration of the active palladium(0) species.
Impurity control is achieved through multiple synergistic mechanisms inherent to this catalytic cycle. The mild reaction temperature prevents thermal decomposition pathways that typically generate dehydrofluorination byproducts in conventional syntheses, while the controlled release of carbon monoxide from the formic acid/acetic anhydride system avoids concentration spikes that could lead to over-carbonylation side products. The sodium bicarbonate base serves dual functions by neutralizing hydrochloric acid generated during imine formation while maintaining optimal pH conditions that suppress hydrolysis of sensitive intermediates. Furthermore, the specific molar ratio of palladium chloride to triphenylphosphine (0.05:0.1) creates a ligand-saturated environment that minimizes catalyst decomposition into inactive palladium black particles—a common source of metal impurities in industrial processes. This comprehensive impurity management strategy ensures consistent production of high-purity intermediates meeting stringent pharmaceutical quality standards without requiring additional purification steps beyond standard chromatography.
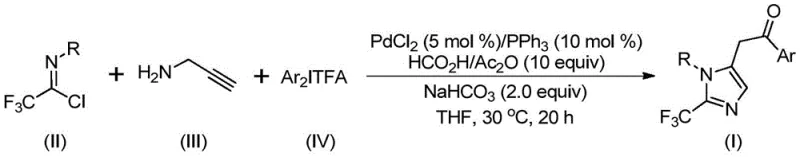
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
This section outlines the operational framework for implementing the patented synthesis method in industrial settings, emphasizing critical control parameters that ensure consistent product quality and process reliability. The methodology represents a significant advancement over conventional approaches by eliminating hazardous reagents while maintaining exceptional substrate flexibility across diverse aromatic systems. Key advantages include simplified equipment requirements due to ambient temperature operation and reduced safety protocols compared to high-pressure or cryogenic processes previously employed in imidazole chemistry. The following standardized procedure provides a foundation for technology transfer to manufacturing facilities, with detailed operational parameters available through our technical documentation portal for qualified partners seeking to implement this innovative route.
- Prepare the reaction mixture by combining palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2.0 equiv), acetic anhydride/formic acid (as CO source), trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide salt in anhydrous THF under inert atmosphere.
- Stir the homogeneous solution at precisely 30°C for 20 hours while monitoring reaction progression through standard analytical techniques to ensure complete conversion without over-reaction.
- Execute post-treatment by filtration through silica gel followed by column chromatography purification using optimized solvent gradients to isolate the target imidazole compound meeting stringent pharmaceutical purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers transformative value across procurement and supply chain operations by addressing fundamental pain points in pharmaceutical intermediate sourcing through scientifically validated process improvements. The elimination of specialized equipment requirements significantly reduces capital expenditure barriers while enhancing manufacturing flexibility across global production networks. By utilizing commercially available starting materials with established supply chains, the process mitigates raw material sourcing risks that frequently disrupt traditional synthetic routes for fluorinated heterocycles. Furthermore, the simplified workflow reduces production cycle times through fewer unit operations and eliminates complex purification stages required in conventional methods, creating substantial operational efficiencies that translate directly into enhanced supply chain resilience.
- Cost Reduction in Manufacturing: The strategic elimination of expensive transition metal catalysts and cryogenic processing requirements delivers substantial cost savings through reduced raw material expenditures and lower energy consumption profiles. By leveraging readily available formic acid/acetic anhydride as a safe carbon monoxide surrogate instead of pressurized CO gas systems, the process avoids significant capital investments in specialized gas handling infrastructure while minimizing safety-related operational costs associated with hazardous material management.
- Enhanced Supply Chain Reliability: The use of globally sourced starting materials with established commercial availability ensures consistent supply chain performance even during market volatility periods. The method's compatibility with standard manufacturing equipment eliminates dependency on specialized reactors that create single-point failure risks in production networks, while the robust reaction profile maintains consistent output quality across different geographical locations without requiring extensive process revalidation.
- Scalability and Environmental Compliance: The ambient temperature operation enables seamless scale-up from laboratory validation to commercial production volumes without requiring engineering modifications to existing facilities. The process generates minimal hazardous waste streams due to its high atom economy and elimination of toxic reagents, significantly reducing environmental compliance burdens while meeting increasingly stringent global regulatory requirements for sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of Patent CN111423381B's experimental data and implementation requirements. These insights have been developed through rigorous evaluation of the methodology's performance characteristics across multiple production scales and substrate variations.
Q: How does this method improve upon conventional synthesis of trifluoromethyl imidazoles?
A: The patented process eliminates harsh reaction conditions and expensive transition metal catalysts required in traditional methods by utilizing a mild palladium-catalyzed carbonylation strategy at ambient temperature. This approach significantly reduces impurity formation while leveraging commercially available starting materials like trifluoroethylimidoyl chloride and diaryl iodide salts, resulting in higher product purity and operational safety compared to conventional routes that often require cryogenic temperatures or toxic reagents.
Q: What are the scalability advantages of this palladium-catalyzed process?
A: The reaction demonstrates exceptional scalability due to its tolerance for standard industrial equipment and mild operating parameters (30°C, atmospheric pressure). The use of stable, non-pyrophoric catalysts and readily available solvents like THF enables seamless transition from laboratory-scale to commercial production volumes without specialized infrastructure. Furthermore, the simplified workup procedure involving standard filtration and chromatography minimizes processing complexity during scale-up.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The mechanism inherently controls impurity profiles through selective carbonylation and precise substrate design, minimizing side reactions that generate critical impurities. The mild reaction conditions prevent decomposition pathways common in traditional syntheses, while the chromatographic purification step effectively removes residual catalysts and byproducts. This results in consistently high-purity intermediates meeting ICH Q7 guidelines without requiring additional costly purification stages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities. As a trusted partner in complex heterocyclic synthesis, we have successfully implemented this patented methodology across multiple client projects with consistent delivery of high-purity intermediates meeting global regulatory standards including ICH Q7 and USP monographs. Our vertically integrated manufacturing platform combines cutting-edge process chemistry expertise with robust supply chain management systems to ensure uninterrupted delivery of mission-critical intermediates for pharmaceutical development programs worldwide.
Leverage our technical procurement team's expertise to request specific COA data and route feasibility assessments tailored to your production requirements. We offer Customized Cost-Saving Analysis services that quantify potential savings through implementation of this innovative synthesis route compared to conventional manufacturing approaches.
