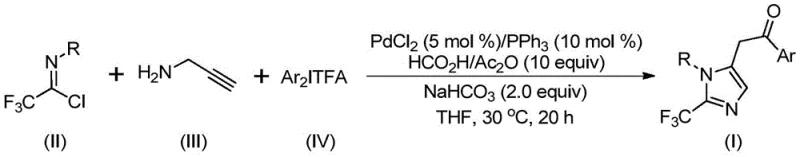
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which are pivotal for enhancing metabolic stability and lipophilicity in drug candidates. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical limitations in current synthetic strategies. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. Unlike conventional approaches that often require harsh conditions or unstable reagents, this novel protocol operates under remarkably mild conditions at 30°C, ensuring high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline the production of high-purity pharmaceutical intermediates while mitigating safety risks associated with traditional diazo chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of synthons like trifluorodiazoethane. While effective in certain contexts, these traditional methods present substantial challenges for commercial scale-up and operational safety. Trifluorodiazoethane is inherently unstable and poses significant explosion hazards, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure and operational costs. Furthermore, the narrow substrate scope of many legacy methods often necessitates complex protection and deprotection strategies, leading to increased step counts and reduced overall atom economy. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to meet the demands of global drug development pipelines.
The Novel Approach
The methodology disclosed in CN111423381A offers a transformative alternative by employing a palladium-catalyzed carbonylation cascade that bypasses the need for hazardous diazo compounds. By utilizing trifluoroethylimidoyl chloride and diaryliodonium salts as key building blocks, the process achieves efficient construction of the imidazole core with high regioselectivity. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF) at a mild temperature of 30°C, demonstrating remarkable tolerance for various functional groups including halogens, alkyls, and nitro groups. This broad compatibility allows for the rapid generation of diverse chemical libraries, facilitating the discovery of new bioactive molecules. The simplicity of the operation, combined with the use of commercially available catalysts like palladium chloride and triphenylphosphine, makes this approach highly attractive for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps that ensure high fidelity and yield. Initially, the reaction likely undergoes a base-promoted intermolecular carbon-nitrogen bond formation to generate a trifluoroacetamidine intermediate. This species then experiences isomerization followed by palladium-catalyzed alkyne aminopalladation, yielding an alkenyl palladium intermediate. Subsequent isomerization converts this into an alkyl palladium species, which is primed for the crucial carbonylation step. In the presence of carbon monoxide released in situ from the formic acid and acetic anhydride mixture, an acyl palladium intermediate is formed. The cycle concludes with oxidative addition of the diaryliodonium salt to generate a tetravalent palladium intermediate, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product. Understanding this detailed catalytic cycle is essential for optimizing reaction parameters and ensuring consistent quality in commercial production.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The use of well-defined starting materials and a controlled catalytic cycle minimizes the formation of side products often associated with radical pathways or uncontrolled decomposition of diazo precursors. The specific choice of ligands and additives, such as sodium bicarbonate, helps maintain the pH balance and stabilizes the active palladium species, further suppressing unwanted byproducts. This results in a cleaner crude reaction profile, simplifying downstream purification processes like column chromatography. For quality assurance teams, this means achieving stringent purity specifications with less effort, directly impacting the cost of goods sold and accelerating time-to-market for new drug substances.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to maximize yield and reproducibility. The process involves mixing the palladium catalyst system with the specific ratio of trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt in an aprotic solvent. Maintaining the reaction temperature at 30°C for a duration of 16 to 24 hours is critical to ensure complete conversion without degrading sensitive functional groups. Following the reaction, a straightforward workup procedure involving filtration and silica gel treatment allows for easy isolation of the target compound. Detailed standardized synthesis steps see the guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid mixture, trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at 30°C for 16 to 24 hours to ensure complete conversion of starting materials.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented method offers compelling advantages that address common pain points in the sourcing of complex heterocyclic intermediates. The reliance on commercially available and inexpensive starting materials significantly reduces raw material costs and mitigates supply risk. Unlike specialized diazo reagents that may have limited suppliers and long lead times, trifluoroethylimidoyl chlorides and diaryliodonium salts are accessible from multiple global vendors, ensuring supply continuity even during market fluctuations. Furthermore, the mild reaction conditions reduce energy consumption and eliminate the need for expensive high-pressure or cryogenic equipment, contributing to substantial cost savings in manufacturing overhead.
- Cost Reduction in Manufacturing: The elimination of hazardous diazo compounds removes the need for costly safety infrastructure and specialized waste disposal protocols, leading to significant operational expense reductions. Additionally, the high atom economy and excellent yields observed across diverse substrates minimize material waste, further driving down the cost per kilogram of the final API intermediate. The use of earth-abundant palladium catalysts in low loading amounts also ensures that precious metal recovery costs remain manageable.
- Enhanced Supply Chain Reliability: By utilizing robust and stable reagents, manufacturers can maintain consistent production schedules without the interruptions often caused by the instability of traditional trifluoromethyl sources. The broad substrate scope allows for flexible production planning, enabling the same reactor setup to produce a wide variety of analogues simply by changing the aryl substituents. This flexibility is crucial for CDMOs serving clients with evolving pipeline needs, ensuring rapid response times and reliable delivery of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable to the gram level with potential for ton-scale production, making it suitable for commercial manufacturing. The use of THF as a solvent and the generation of benign byproducts align with green chemistry principles, simplifying environmental compliance and waste treatment. This eco-friendly profile enhances the sustainability credentials of the supply chain, a key factor for modern pharmaceutical companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on hazardous trifluorodiazoethane. This patented process utilizes stable trifluoroethylimidoyl chlorides and diaryliodonium salts, significantly reducing explosion risks and handling hazards associated with diazo compounds.
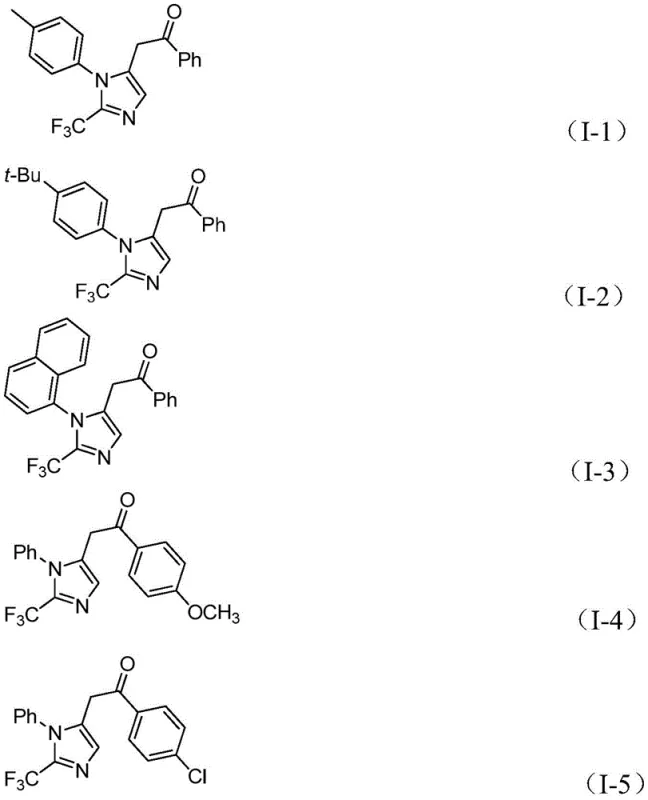
Q: What yields can be expected for diverse substrates in this palladium-catalyzed reaction?
A: The patent demonstrates excellent substrate compatibility with yields ranging from 65% to 97% across various electron-donating and electron-withdrawing groups, including methyl, tert-butyl, methoxy, chloro, bromo, and nitro substituents.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method operates under mild conditions (30°C) using commercially available reagents and standard solvents like THF. The patent explicitly states the process can be expanded to the gram level, providing a viable pathway for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111423381A for accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable industrial supply. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium catalysis and heterocycle chemistry positions us as an ideal partner for bringing these complex molecules to market efficiently.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs.
