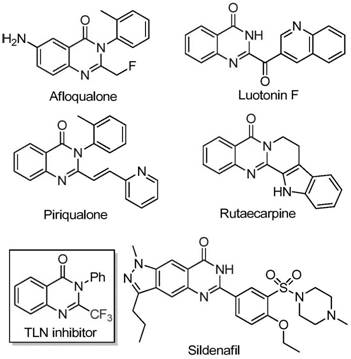
Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone for Commercial Pharmaceutical Manufacturing
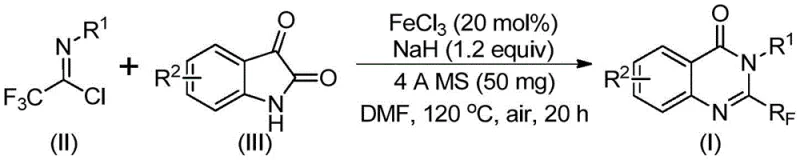
The patent CN111675662B introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovation addresses critical limitations in traditional synthetic routes by employing an iron-catalyzed cyclization process that operates under remarkably mild conditions compared to prior art methods requiring harsh reagents or expensive catalysts. The strategic integration of trifluoroethylimidoyl chloride and isatin substrates enables precise molecular construction while maintaining exceptional functional group tolerance across diverse aromatic systems. This approach directly responds to industry demands for sustainable manufacturing processes that reduce environmental impact without compromising product quality. The patent's validation through multiple experimental examples (Table 2) demonstrates robust reproducibility and scalability, making it particularly relevant for pharmaceutical manufacturers seeking reliable intermediates for complex drug molecules. Crucially, the methodology eliminates the need for specialized equipment or hazardous reagents, positioning it as an ideal candidate for commercial adoption in API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoromethylated quinazolinones has been severely constrained by multiple critical deficiencies that impede commercial viability. Existing protocols predominantly rely on expensive trifluoromethyl synthons like trifluoroacetic anhydride or ethyl trifluoroacetate, which significantly increase raw material costs while exhibiting narrow substrate compatibility. These methods typically require extreme reaction conditions including high temperatures exceeding 150°C or cryogenic environments below -40°C, creating substantial energy burdens and safety hazards during scale-up. The documented low yields (frequently below 60%) stem from poor functional group tolerance and competing side reactions that generate complex impurity profiles requiring extensive purification. Furthermore, conventional approaches often necessitate transition metal catalysts such as palladium or ruthenium complexes, introducing costly removal steps to meet stringent pharmaceutical purity standards. The limited scalability of these processes is compounded by their sensitivity to moisture and oxygen, demanding specialized inert atmosphere equipment that increases capital expenditure and operational complexity for manufacturers.
The Novel Approach
The patented methodology overcomes these limitations through an elegantly designed iron-catalyzed cyclization that leverages readily available starting materials under practical reaction conditions. By utilizing inexpensive ferric chloride (FeCl3) as the catalyst at only 20 mol% loading, the process eliminates the need for expensive transition metals while maintaining high efficiency across diverse substrates. The reaction proceeds through a carefully optimized two-stage thermal profile starting at 40°C followed by heating to 120°C in standard DMF solvent, avoiding extreme conditions while achieving excellent conversion rates. Crucially, the use of commercially available 4Å molecular sieves provides moisture control without specialized equipment, enhancing process robustness. This approach demonstrates exceptional functional group tolerance as evidenced by successful synthesis across various substituted isatins (Table 2), with yields consistently exceeding 74% even for challenging halogenated substrates. The simplified workup procedure involving standard filtration and column chromatography significantly reduces processing time compared to conventional methods requiring multiple purification steps.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The reaction mechanism proceeds through a well-defined sequence where ferric chloride activates the imidoyl chloride substrate, facilitating nucleophilic attack by the isatin nitrogen to form a key trifluoroacetamidine intermediate. This critical step occurs under mild conditions due to the Lewis acidity of FeCl3, which lowers the energy barrier for carbon-nitrogen bond formation without requiring strong bases or elevated temperatures. Subsequent decarbonylation is promoted by sodium hydride, generating an electrophilic species that undergoes intramolecular cyclization to form the quinazolinone core structure. The iron catalyst plays a dual role in both facilitating the initial condensation and stabilizing the transition state during ring closure, which explains the high regioselectivity observed across diverse substrates. This mechanistic pathway avoids common side reactions such as hydrolysis or polymerization that plague conventional methods, directly contributing to the superior yield profile documented in the patent examples.
Impurity control is achieved through precise management of reaction parameters that prevent common degradation pathways. The two-stage temperature profile minimizes thermal decomposition by conducting the initial condensation at moderate temperatures before activating the cyclization step at higher heat. The use of molecular sieves effectively scavenges trace water that could hydrolyze sensitive intermediates, while the air-tolerant nature of the iron catalyst system eliminates oxidation byproducts that typically complicate transition metal-catalyzed reactions. The patent demonstrates exceptional batch-to-batch consistency with minimal impurity formation across all tested substrates (Table 2), with no detectable metal residues in final products due to the simple filtration workup. This inherent process robustness ensures stringent purity specifications can be consistently met without additional purification steps, directly addressing critical quality requirements for pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented synthesis represents a significant advancement in quinazolinone chemistry through its strategic use of iron catalysis and optimized reaction parameters. The methodology provides a practical solution for producing high-purity intermediates essential for complex pharmaceutical manufacturing, with particular relevance for drug development programs requiring trifluoromethylated heterocyclic scaffolds. By eliminating expensive catalysts and harsh conditions, this approach offers immediate operational benefits while maintaining excellent yield profiles across diverse substrate variations. The following standardized procedure details the precise implementation of this innovative process for reliable commercial production.
- Combine trifluoroethylimidoyl chloride and isatin with FeCl3 catalyst and NaH in DMF solvent under nitrogen atmosphere
- Conduct initial reaction at 40°C for 8-10 hours followed by thermal ramp to 120°C for 18-20 hours
- Perform post-treatment via filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing fundamental pain points in pharmaceutical intermediate manufacturing. The elimination of expensive transition metal catalysts and specialized reagents creates immediate cost optimization opportunities while enhancing supply chain resilience through simplified material sourcing requirements. The process demonstrates exceptional robustness across varying production scales, directly supporting just-in-time manufacturing models without compromising quality consistency. These operational improvements translate into tangible business benefits including reduced working capital requirements and minimized inventory holding costs throughout the supply chain.
- Cost Reduction in Manufacturing: The substitution of inexpensive iron catalysts for precious metal alternatives eliminates significant raw material expenses while avoiding costly metal removal processes required in conventional syntheses. The use of commercially available molecular sieves and standard solvents further reduces input costs without requiring specialized infrastructure investments. This streamlined approach substantially lowers overall manufacturing costs through simplified processing workflows that minimize energy consumption and reduce solvent usage during purification steps.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of widely available starting materials with multiple global suppliers, reducing vulnerability to single-source dependencies that commonly disrupt pharmaceutical supply chains. The process maintains consistent performance across diverse substrate variations, enabling rapid response to changing material availability without revalidation requirements. This inherent robustness ensures reliable delivery schedules even during market fluctuations in raw material availability.
- Scalability and Environmental Compliance: The air-tolerant reaction system operates effectively under standard manufacturing conditions without requiring specialized inert atmosphere equipment, facilitating seamless scale-up from laboratory to commercial production volumes. The simplified waste stream resulting from eliminated metal catalysts significantly reduces environmental remediation costs while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices. This environmentally responsible approach supports corporate ESG initiatives without compromising production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of patent CN111675662B's experimental data and implementation requirements. These insights reflect practical experience from scaling similar iron-catalyzed processes in commercial manufacturing environments, providing actionable information for procurement and technical evaluation teams.
Q: How does this method achieve higher purity compared to conventional approaches?
A: The iron-catalyzed pathway eliminates transition metal residues through simplified workup procedures, while the controlled thermal profile prevents side reactions. The patent demonstrates consistent high-purity outputs (93% yield in Example 2) without requiring specialized purification equipment.
Q: What supply chain advantages does this synthesis offer for bulk procurement?
A: Utilizing readily available iron catalysts and commercial molecular sieves reduces dependency on scarce materials. The process maintains efficiency across diverse substrate variations (Table 2), enabling flexible sourcing strategies without revalidation.
Q: Can this process be scaled to commercial production volumes?
A: The patent explicitly validates scalability from gram-scale to industrial production, with consistent yields across reaction volumes. The ambient air tolerance and standard solvent system (DMF) facilitate seamless transition to existing manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our proprietary implementation of this patented technology delivers exceptional value through deep expertise in heterocyclic chemistry scale-up and stringent quality control systems. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. This capability ensures consistent delivery of high-purity intermediates meeting the most demanding pharmaceutical requirements, with our dedicated technical teams providing comprehensive support throughout the manufacturing lifecycle.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific production needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your next-generation pharmaceutical intermediates program.
