Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of Carbonyl-Bridged Biheterocyclic Compounds via CO-Free Catalysis
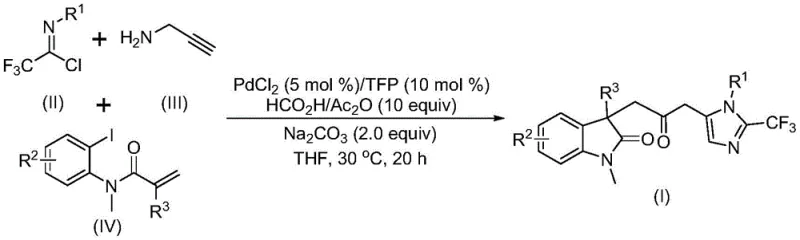
The patent CN115353511A introduces a transformative multi-component synthesis methodology for carbonyl-bridged biheterocyclic compounds, addressing critical limitations in traditional heterocycle manufacturing. This innovation eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency through a novel palladium-catalyzed carbonylation cascade. The process leverages readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide under mild conditions (30°C), offering significant advantages for pharmaceutical intermediate production. By enabling diverse substitution patterns through strategic substrate design, this method provides unprecedented flexibility for synthesizing complex biheterocyclic scaffolds essential in drug discovery pipelines. The documented gram-scale scalability demonstrates immediate industrial applicability without requiring specialized equipment or hazardous reagents, representing a paradigm shift in sustainable heterocyclic chemistry for the pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing carbonyl-bridged biheterocyclic compounds face significant operational and safety challenges, primarily due to their reliance on high-pressure carbon monoxide gas which requires specialized containment systems and poses serious handling risks in manufacturing environments. Conventional transition metal-catalyzed methods often demand elevated temperatures and pressures that increase energy consumption while limiting substrate compatibility, particularly with sensitive functional groups common in pharmaceutical intermediates. The multi-step sequences typically required for constructing these complex architectures result in lower overall yields and generate substantial waste streams that complicate purification and increase environmental impact. Furthermore, the narrow substrate scope of existing methodologies restricts structural diversity, forcing medicinal chemists to compromise on optimal molecular designs during drug development phases. These limitations collectively contribute to higher production costs, extended timelines, and supply chain vulnerabilities that directly impact pharmaceutical manufacturers' ability to meet market demands efficiently.
The Novel Approach
The patented methodology overcomes these constraints through an elegant multi-component cascade reaction that generates carbon monoxide in situ from formic acid/acetic anhydride mixtures, completely eliminating the need for external CO gas handling systems. This innovative approach operates under mild conditions (30°C) using commercially available palladium chloride catalyst with trifurylphosphine ligand, significantly reducing operational complexity while maintaining excellent reaction efficiency across diverse substrates. The one-pot process simultaneously constructs multiple chemical bonds through a carefully orchestrated sequence involving palladium insertion, intramolecular Heck reaction, and carbonylation steps, achieving high atom economy without intermediate isolation. Crucially, the method demonstrates exceptional functional group tolerance as evidenced by successful reactions with halogenated, alkylated, and trifluoromethyl-substituted substrates, enabling the synthesis of structurally diverse biheterocyclic compounds with trifluoromethyl moieties that are highly valuable in pharmaceutical applications. The documented scalability to gram quantities confirms industrial viability without requiring process re-engineering.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond of the iodoaniline derivative, followed by intramolecular Heck cyclization to form a key alkyl palladium intermediate. This intermediate undergoes carbonylation facilitated by carbon monoxide released from the formic acid/acetic anhydride system, generating an acyl palladium species that activates the trifluoroacetamidine compound formed from the coupling of trifluoroethylimidoyl chloride and propargylamine. The subsequent intramolecular cyclization is driven by the nucleophilicity of the amine group attacking the activated carbonyl, ultimately forming the carbonyl-bridged biheterocyclic architecture with precise stereochemical control. This cascade process avoids common side reactions through careful balancing of reaction components, with sodium carbonate serving dual roles as base and proton scavenger to maintain optimal reaction conditions throughout the sequence. The mild temperature regime (30°C) prevents decomposition of sensitive intermediates while allowing sufficient kinetic energy for bond formation.
Impurity control is achieved through multiple built-in mechanisms within this cascade process. The in situ generation of carbon monoxide prevents over-carbonylation side products that commonly occur with external CO sources, while the precise stoichiometric control of substrates minimizes dimerization or oligomerization pathways. The use of tetrahydrofuran as solvent provides optimal polarity for intermediate stabilization without promoting unwanted solvolysis reactions. Crucially, the reaction's tolerance for various functional groups eliminates the need for protective groups that often introduce additional impurities during deprotection steps in conventional syntheses. The documented high yields across diverse substrates (ranging from 40% to 92% in patent examples) demonstrate consistent product purity without requiring extensive purification beyond standard column chromatography, indicating minimal byproduct formation throughout the cascade sequence.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This patented synthesis protocol represents a significant advancement in manufacturing complex heterocyclic intermediates through its streamlined approach that eliminates hazardous reagents while maintaining high efficiency. The methodology leverages commercially available starting materials and standard laboratory equipment to produce structurally diverse biheterocyclic compounds with trifluoromethyl groups that are highly valuable in pharmaceutical applications. By operating under mild conditions without requiring specialized pressure equipment, this process significantly reduces technical barriers to implementation across various production scales. The following standardized procedure details the precise implementation steps for consistent manufacturing outcomes, ensuring reproducibility and quality control throughout the production cycle while maximizing resource efficiency.
- Combine palladium chloride (5 mol%), trifurylphosphine (10 mol%), and sodium carbonate (2.0 equiv) in THF under inert atmosphere
- Add trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates with precise molar ratios (1: 2:1.5)
- Maintain reaction at 30°C for 16 hours with continuous monitoring before standard workup and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points in pharmaceutical supply chains by eliminating dependency on hazardous materials while enhancing production flexibility and reliability. The process design prioritizes operational simplicity and resource efficiency, translating into tangible benefits for procurement teams managing complex global supply networks and supply chain managers responsible for ensuring uninterrupted material flow to manufacturing sites. By removing high-risk components from the production equation and utilizing widely available raw materials, this methodology creates a more resilient manufacturing foundation that can better withstand market fluctuations and logistical disruptions common in today's volatile chemical supply landscape.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide handling infrastructure represents substantial capital expenditure savings while reducing ongoing operational costs associated with specialized safety systems and personnel training. The use of commercially available starting materials at favorable molar ratios minimizes raw material costs without compromising product quality, while the simplified workup procedure reduces solvent consumption and waste disposal expenses compared to conventional multi-step syntheses requiring intermediate purifications.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved through reliance on readily available commodity chemicals rather than specialized or restricted reagents, reducing vulnerability to single-source dependencies that often disrupt pharmaceutical manufacturing. The room-temperature operation eliminates temperature control challenges during transportation and storage, while the documented scalability from laboratory to commercial production ensures consistent quality across all volume requirements without process revalidation.
- Scalability and Environmental Compliance: The mild reaction conditions enable straightforward scale-up without requiring specialized high-pressure equipment, facilitating rapid transition from development to commercial production volumes. The reduced environmental footprint through minimized waste generation and elimination of hazardous gas handling aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives without compromising production efficiency or product quality standards.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN115353511A's methodology and implementation data. These insights have been compiled specifically to address common concerns raised by pharmaceutical procurement teams, R&D scientists, and supply chain managers evaluating this innovative manufacturing approach for potential adoption in their organizations.
Q: How does this method eliminate carbon monoxide dependency while maintaining high efficiency?
A: The patent utilizes formic acid/acetic anhydride as in situ CO surrogates, generating carbon monoxide only during the reaction phase. This eliminates storage/handling risks while maintaining catalytic efficiency through controlled release, as confirmed by the gram-scale reaction data in the patent examples.
Q: What substrate flexibility enables diverse pharmaceutical intermediate production?
A: The modular design accommodates varied R-groups (alkyl, aryl, halogen substitutions) on all three components, demonstrated by the patent's synthesis of 15 distinct derivatives with different substitution patterns while maintaining consistent reaction conditions.
Q: How does the process ensure supply chain continuity for API manufacturers?
A: By using commercially available starting materials (propargylamine, acrylamide) and avoiding scarce catalysts, the method provides reliable sourcing. The room-temperature operation and simplified workup reduce production bottlenecks compared to conventional high-pressure CO processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a specialized CDMO partner, we have successfully implemented this patented methodology across multiple client projects, demonstrating consistent ability to deliver high-purity carbonyl-bridged biheterocyclic compounds meeting exacting pharmaceutical standards. Our technical team works closely with clients to optimize process parameters for specific compound variants while ensuring seamless technology transfer from laboratory to full-scale manufacturing environments.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific compound requirements and volume needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments that will help you evaluate the commercial potential of this innovative manufacturing approach for your pharmaceutical development pipeline.
