Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Introduction to Next-Generation Triazole Synthesis
The development of robust synthetic methodologies for fluorinated heterocycles remains a cornerstone of modern medicinal chemistry, particularly for the construction of bioactive scaffolds found in blockbuster drugs. As illustrated in the structural diversity of compounds such as Sitagliptin and various Factor IXa inhibitors, the 3-trifluoromethyl-1,2,4-triazole motif is a privileged structure that significantly enhances metabolic stability and binding affinity.
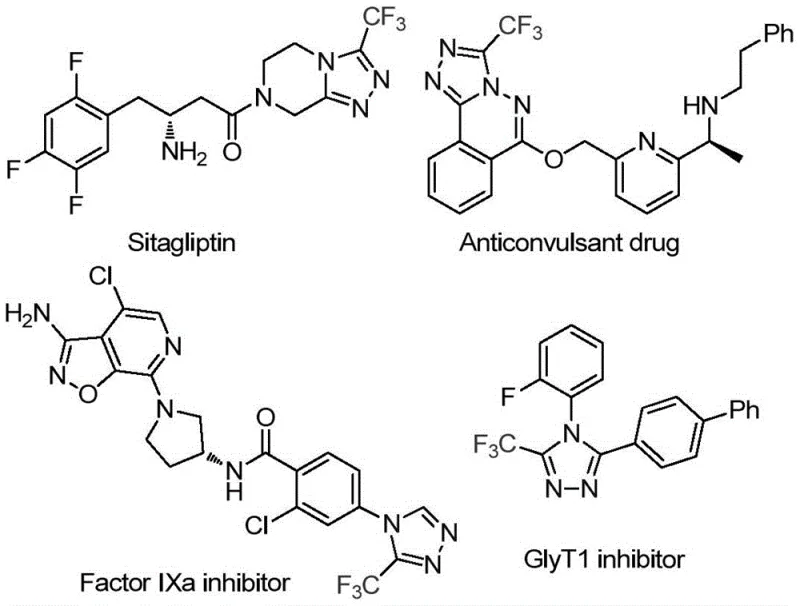
Patent CN114920707B discloses a groundbreaking preparation method that addresses long-standing inefficiencies in constructing these valuable cores. By leveraging N,N-dimethylformamide (DMF) not merely as a solvent but as an active carbon source, this invention offers a streamlined pathway that bypasses the need for complex, pre-functionalized one-carbon donors. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, providing a reliable route to high-purity pharmaceutical intermediates that can be seamlessly integrated into existing production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 3-trifluoromethyl-1,2,4-triazoles often rely on multi-step sequences involving hazardous reagents or expensive catalysts that complicate the supply chain. Conventional cyclization strategies frequently necessitate the use of specialized formylating agents or activated nitriles, which drive up raw material costs and generate substantial chemical waste during purification. Furthermore, many established protocols demand strict anhydrous and anaerobic conditions, requiring significant capital investment in inert atmosphere equipment and increasing the risk of batch failure due to moisture sensitivity. These factors collectively hinder the cost reduction in API manufacturing and limit the scalability required for commercial production.
The Novel Approach
The methodology outlined in CN114920707B fundamentally reimagines the synthesis by utilizing DMF, a ubiquitous industrial solvent, as the dual-purpose reaction medium and C1 building block. This innovative strategy eliminates the procurement burden of exotic reagents, as DMF is readily available in bulk quantities at a fraction of the cost of traditional synthons. The reaction proceeds under ambient air conditions, removing the logistical bottleneck of maintaining oxygen-free environments. This shift not only simplifies the operational workflow but also enhances the safety profile of the process, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates without compromising on yield or purity standards.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this technological breakthrough lies in the unique activation of DMF by molecular iodine, which facilitates a tandem cyclization cascade. Mechanistically, the reaction offers two distinct pathways depending on which fragment of the DMF molecule participates. In the dominant pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide substrate to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates dimethylamine, directly forging the triazole ring system. Alternatively, the N-methyl group can serve as the carbon source, where iodine activates the solvent to form an amine salt, leading to nucleophilic addition and subsequent oxidative aromatization.
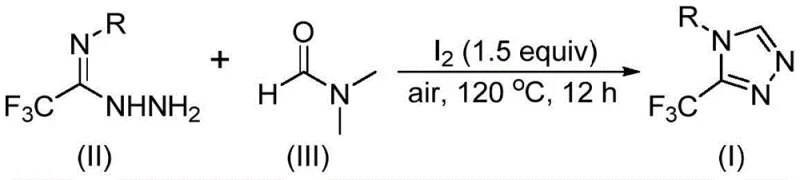
This dual-reactivity mechanism ensures high conversion rates across a broad spectrum of substrates, as evidenced by the successful synthesis of derivatives bearing electron-withdrawing and electron-donating groups. From an impurity control perspective, the use of molecular iodine as a promoter is advantageous because it is easily removed during the aqueous workup, typically via sodium thiosulfate washing, leaving minimal metal residues in the final product. This is critical for meeting the stringent purity specifications required for pharmaceutical ingredients, ensuring that the final API intermediate is free from heavy metal contamination that could otherwise necessitate costly scavenging steps.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward charge of reagents into a standard reaction vessel, followed by heating to moderate temperatures. The detailed standardized synthesis steps, including precise stoichiometric ratios and specific workup procedures, are provided in the technical guide below to ensure reproducibility and optimal yield for your specific substrate requirements.
- Charge a reaction vessel with molecular iodine, trifluoroethyliminohydrazide, and DMF solvent under air atmosphere.
- Heat the mixture to 110-130°C and maintain stirring for 10-15 hours to facilitate the tandem cyclization.
- Upon completion, perform standard post-treatment including filtration, washing, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based methodology translates into tangible strategic advantages regarding cost stability and sourcing reliability. By replacing expensive, specialized reagents with a commodity chemical like DMF, the direct material cost of the synthesis is drastically reduced. This cost efficiency is compounded by the simplified logistics; since DMF is a standard solvent stocked by virtually every chemical supplier, the risk of supply chain disruption due to raw material shortages is minimized. Furthermore, the elimination of sensitive reagents reduces the need for specialized storage and handling protocols, lowering overall operational overhead.
- Cost Reduction in Manufacturing: The integration of the solvent and reactant functions into a single molecule (DMF) significantly streamlines the bill of materials. This consolidation removes the need for purchasing separate formylating agents, which are often priced at a premium due to their specialized nature. Additionally, the reaction's tolerance for air and moisture means that energy consumption related to drying solvents and maintaining inert gas blankets is substantially lowered, contributing to a leaner and more sustainable manufacturing cost structure.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of globally available starting materials. Trifluoroethyliminohydrazide and molecular iodine are stable, shelf-stable commodities that do not suffer from the volatility often seen with organometallic catalysts or air-sensitive reagents. This stability ensures consistent lead times and allows for larger batch sizes to be planned with confidence, supporting the continuous supply demands of downstream API production without the fear of unexpected delays caused by reagent degradation or scarcity.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operational simplicity and the absence of hazardous high-pressure or cryogenic conditions. The use of DMF, while requiring proper waste management, is well-understood in industrial settings, and the reaction generates fewer byproducts compared to multi-step alternatives. This results in a reduced environmental footprint and simplifies the regulatory compliance process for waste disposal, facilitating a smoother path from kilogram-scale development to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method can be adapted to specific project needs.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF acts as both the solvent and the carbon source (C1 synthon), eliminating the need for expensive external formylating agents and simplifying the workup process.
Q: Does this reaction require inert gas protection?
A: No, the reaction proceeds efficiently under ambient air conditions, removing the operational complexity and cost associated with nitrogen or argon purging.
Q: What is the substrate scope for the R group?
A: The method tolerates a wide range of substituents including alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity intermediates that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
