Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles with enhanced pharmacokinetic profiles. Patent CN111423381A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in modern medicinal chemistry. Imidazole scaffolds are ubiquitous in bioactive molecules, serving as key structural motifs in drugs targeting various therapeutic areas, from antifungals to kinase inhibitors. The strategic incorporation of a trifluoromethyl group into these heterocyclic systems is known to drastically improve metabolic stability and membrane permeability, yet traditional synthetic routes often suffer from harsh conditions or limited substrate scope. This patent presents a transition metal palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions, utilizing cheap and readily available starting materials to efficiently construct these valuable architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on the use of highly reactive and potentially hazardous synthons such as trifluorodiazoethane. While effective in certain contexts, these reagents pose significant safety risks due to their explosive nature and require specialized handling equipment that increases operational complexity and cost. Furthermore, alternative methods involving direct trifluoromethylation often necessitate strong bases, elevated temperatures, or stoichiometric amounts of expensive copper or silver promoters, which complicate downstream purification and waste management. The reliance on high-pressure carbon monoxide gas for carbonylation steps also introduces severe safety hazards and engineering constraints, limiting the scalability of these processes for industrial applications. Consequently, there is a persistent demand for safer, more atom-economical, and operationally simple protocols that can tolerate a wide range of functional groups without compromising yield or purity.
The Novel Approach
The methodology described in CN111423381A represents a paradigm shift by employing a palladium-catalyzed cascade reaction that integrates carbonylation and cyclization in a single pot. This novel approach utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts as the core building blocks, reacting them in the presence of a PdCl2/PPh3 catalyst system. Crucially, the reaction generates carbon monoxide in situ from a mixture of formic acid and acetic anhydride, thereby eliminating the need for external CO gas cylinders and high-pressure reactors. The process proceeds efficiently at a mild temperature of 30°C over 16 to 24 hours, demonstrating exceptional functional group tolerance. As illustrated in the reaction scheme below, this strategy allows for the modular assembly of diverse 2-trifluoromethyl imidazole derivatives by simply varying the aryl groups on the starting materials.
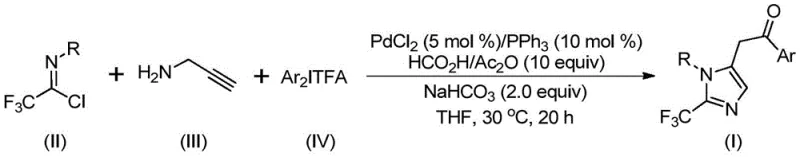
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The reaction mechanism involves a sophisticated sequence of organometallic transformations initiated by base-promoted intermolecular carbon-nitrogen bond formation to generate a trifluoroacetamidine intermediate. This species undergoes isomerization followed by palladium-catalyzed aminopalladation of the alkyne moiety, yielding a vinyl-palladium intermediate. Subsequent isomerization leads to an alkyl-palladium species, which then undergoes carbonylation upon exposure to the carbon monoxide released from the formic acid/acetic anhydride mixture. The resulting acyl-palladium intermediate is then subjected to oxidative addition by the diaryliodonium salt, forming a high-valent Pd(IV) species. The catalytic cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole product and regenerates the active Pd(II) catalyst. This intricate pathway highlights the dual role of the palladium center in facilitating both C-N and C-C bond formations while incorporating the carbonyl functionality seamlessly.
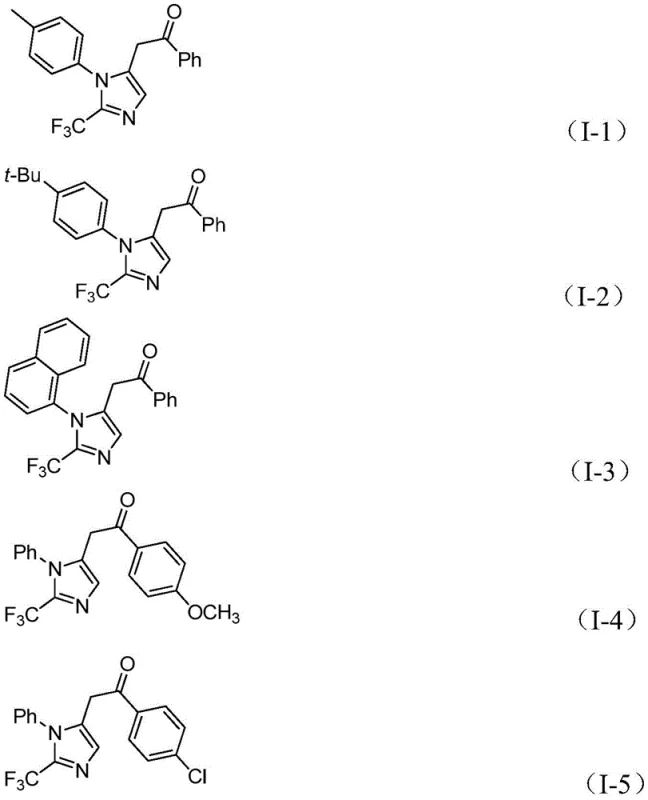
From a quality control perspective, the use of diaryliodonium salts as oxidants offers distinct advantages in impurity profiling. Unlike traditional oxidants that may leave behind difficult-to-remove metal residues or inorganic salts, the byproducts of diaryliodonium salt oxidation are typically neutral organic species that are easily separated during standard workup procedures. The mild reaction temperature of 30°C further minimizes the formation of thermal degradation byproducts or polymerization side reactions that often plague high-temperature heterocycle syntheses. The broad substrate compatibility, encompassing electron-rich and electron-deficient aryl groups as well as various substituents like halogens, alkyls, and nitro groups, ensures that the impurity profile remains consistent and predictable across different analogues. This predictability is vital for regulatory filings where the identification and quantification of process-related impurities are mandatory.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The synthesis protocol outlined in the patent provides a standardized framework for producing high-purity 2-trifluoromethyl imidazoles suitable for drug discovery and development. The procedure is designed to be operationally simple, requiring only standard laboratory glassware and common reagents, which facilitates rapid adoption in both academic and industrial settings. By adhering to the specified molar ratios and reaction times, chemists can reliably achieve high conversion rates while minimizing the formation of side products. The detailed标准化 synthesis steps见下方的指南 ensure reproducibility and safety, making this method an attractive option for process chemists looking to optimize their synthetic routes for complex heterocyclic targets.
- Charge a reaction vessel with palladium chloride, triphenylphosphine, sodium bicarbonate, trifluoroethylimidoyl chloride, propargylamine, diaryliodonium salt, and an organic solvent such as THF.
- Add a mixture of formic acid and acetic anhydride as the carbon monoxide source and stir the reaction mixture at 30°C for 16 to 24 hours.
- Upon completion, filter the mixture, treat with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits in terms of cost efficiency and supply reliability. The reliance on commercially available starting materials such as aromatic amines, propargylamine, and aryl boronic acids (precursors to iodonium salts) ensures a stable supply chain不受 geopolitical disruptions affecting exotic reagents. Furthermore, the use of palladium chloride, a relatively inexpensive palladium source compared to specialized ligand-bound complexes, contributes to significant cost reduction in API manufacturing. The elimination of high-pressure equipment and hazardous gas handling reduces capital expenditure on specialized reactor infrastructure and lowers insurance and compliance costs associated with dangerous goods.
- Cost Reduction in Manufacturing: The process utilizes inexpensive catalysts and avoids the need for cryogenic conditions or high-pressure autoclaves, leading to substantial operational savings. The in situ generation of carbon monoxide removes the logistical burden and cost of purchasing and storing compressed gas cylinders. Additionally, the high reaction efficiency and yield reported in the patent examples minimize raw material waste, directly improving the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials are commodity chemicals with robust global supply networks, reducing the risk of shortages that often accompany custom-synthesized intermediates. The mild reaction conditions allow for flexible scheduling and batch processing without the need for extended cooling or heating cycles, thereby increasing throughput capacity. This reliability ensures consistent delivery timelines for downstream customers, supporting just-in-time manufacturing models essential for modern pharmaceutical production.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable to gram levels with potential for kilogram-to-ton scale-up, supported by the use of common organic solvents like THF which are easily recovered and recycled. The avoidance of toxic heavy metal oxidants and explosive diazo compounds aligns with green chemistry principles, simplifying waste disposal and environmental permitting. This environmental compatibility reduces the regulatory burden and enhances the sustainability profile of the manufacturing process, a key metric for modern ESG-conscious supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for large-scale production. Understanding these nuances helps stakeholders make informed decisions about integrating this chemistry into their existing pipelines.
Q: What are the primary benefits of introducing a trifluoromethyl group into imidazole scaffolds?
A: Introducing a trifluoromethyl group significantly enhances the electronegativity, bioavailability, metabolic stability, and lipophilicity of the parent molecule, making it highly valuable for drug design.
Q: How does this method improve safety compared to traditional trifluoromethylation techniques?
A: This method utilizes formic acid and acetic anhydride as a safe, in-situ carbon monoxide source, avoiding the handling of hazardous high-pressure CO gas or unstable trifluorodiazoethane reagents.
Q: Is the catalyst system cost-effective for large-scale manufacturing?
A: Yes, the process employs palladium chloride and triphenylphosphine, which are relatively inexpensive and commercially available compared to specialized noble metal complexes, facilitating cost reduction in API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We are committed to delivering high-purity 2-trifluoromethyl imidazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch conforms to the highest industry standards, providing our partners with the confidence they need to advance their clinical programs.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs. Let us be your partner in transforming complex chemical challenges into commercial successes.
