Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Manufacturing
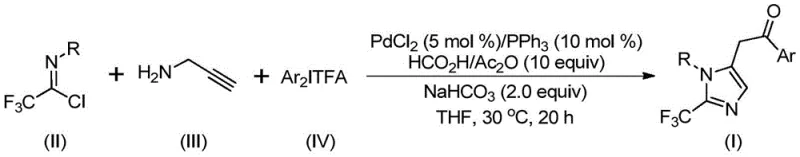
The pharmaceutical industry continuously seeks robust methodologies for constructing fluorinated heterocycles, particularly 2-trifluoromethyl substituted imidazoles, due to their profound impact on the metabolic stability and lipophilicity of drug candidates. Patent CN111423381A introduces a groundbreaking preparation method that addresses long-standing synthetic challenges in this domain. This innovation leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts as key building blocks. The significance of this technology lies in its ability to operate under exceptionally mild conditions, specifically at 30°C, while maintaining high reaction efficiency and broad substrate compatibility. For R&D directors and process chemists, this represents a pivotal shift away from hazardous reagents towards safer, more controllable synthetic pathways that facilitate the rapid generation of diverse molecular libraries for drug discovery programs.
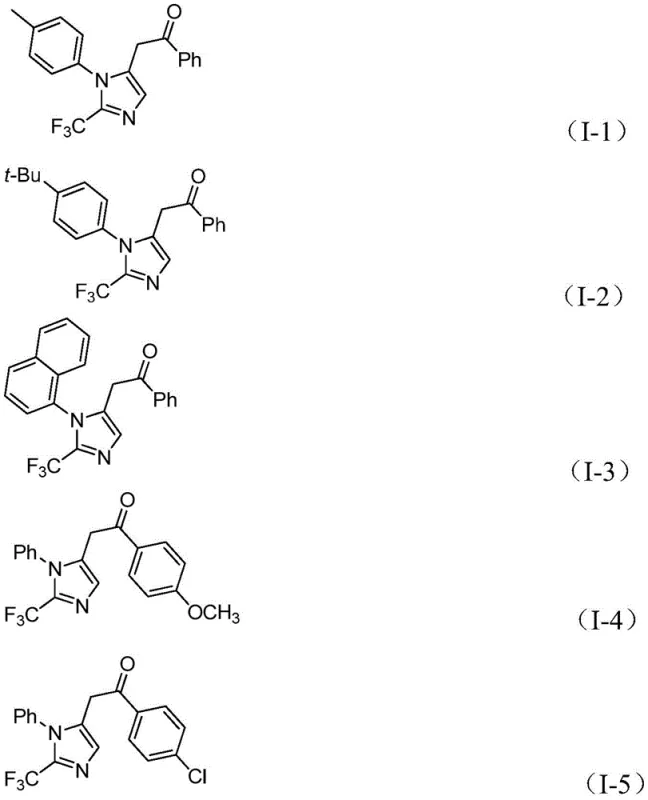
Historically, the construction of nitrogen-containing heterocycles bearing trifluoromethyl groups has been fraught with difficulties, often relying on synthons like trifluorodiazoethane which pose significant safety risks due to their explosive nature. Conventional literature methods frequently require harsh reaction conditions, specialized equipment, or expensive catalysts that hinder scalability and increase the overall cost of goods. Furthermore, the limited functional group tolerance of older protocols often necessitates complex protection and deprotection strategies, elongating the synthetic timeline and reducing overall yield. These limitations create substantial bottlenecks for procurement managers and supply chain heads who require reliable, cost-effective, and safe manufacturing processes for active pharmaceutical ingredient (API) intermediates. The reliance on unstable precursors also introduces variability in batch-to-batch consistency, complicating quality control measures in a regulated environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for introducing trifluoromethyl groups into imidazole scaffolds often suffer from poor atom economy and severe safety hazards associated with handling gaseous or unstable liquid reagents. The use of trifluorodiazoethane, while effective in some academic settings, is impractical for large-scale industrial application due to the rigorous safety protocols required to mitigate explosion risks. Additionally, many existing methods utilize stoichiometric amounts of heavy metal oxidants or require high temperatures that can degrade sensitive functional groups present in complex drug molecules. This lack of chemoselectivity forces process chemists to design longer linear syntheses, increasing waste generation and solvent consumption. From a commercial perspective, these inefficiencies translate into higher production costs and extended lead times, making it difficult to compete in the fast-paced generic and specialty pharmaceutical markets where speed to market is critical.
The Novel Approach
In stark contrast, the methodology disclosed in CN111423381A offers a streamlined, one-pot solution that dramatically simplifies the synthetic workflow. By employing a palladium-catalyzed carbonylation strategy, the process effectively utilizes formic acid and acetic anhydride as a safe, in situ source of carbon monoxide, eliminating the need for high-pressure CO gas cylinders. The reaction proceeds smoothly at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), ensuring energy efficiency and operational simplicity. This novel approach not only enhances safety profiles but also significantly improves the economic viability of producing 2-trifluoromethyl imidazoles. The use of cheap and commercially available starting materials, such as aromatic amines and propargylamine, ensures a stable supply chain, while the high yields reported across various substrates demonstrate the robustness of the catalytic system for diverse chemical architectures.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated yet efficient palladium catalytic cycle that orchestrates multiple bond-forming events in a single operation. Initially, the reaction likely proceeds through a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species then undergoes isomerization followed by palladium-catalyzed aminopalladation of the alkyne moiety to form a vinyl-palladium intermediate. Subsequent isomerization yields an alkyl-palladium species, which is then subjected to carbonylation driven by carbon monoxide released from the formic acid/acetic anhydride mixture. This step generates a crucial acyl-palladium intermediate that sets the stage for the final ring closure.
The cycle concludes with the oxidative addition of the diaryliodonium salt to the palladium center, forming a high-valent Pd(IV) intermediate, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active Pd(0) catalyst. This intricate mechanism highlights the dual role of the palladium catalyst in facilitating both the carbonylation and the C-C bond formation steps. From an impurity control perspective, the mild reaction temperature of 30°C is instrumental in suppressing side reactions such as polymerization of the alkyne or decomposition of the sensitive imidoyl chloride. The use of sodium bicarbonate as a mild base further ensures that acid-sensitive functionalities remain intact, resulting in a cleaner crude reaction profile that simplifies downstream purification and enhances the overall purity of the final pharmaceutical intermediate.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory glassware and commonly available reagents. The protocol involves mixing the palladium catalyst, ligand, additives, and substrates in an organic solvent, followed by stirring at a controlled temperature. This straightforward procedure minimizes the need for specialized equipment, making it accessible for both small-scale discovery chemistry and larger pilot plant operations. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and optimal yield for your specific application requirements.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards safer, more stable starting materials directly mitigates supply chain risks associated with hazardous reagent sourcing and storage. Furthermore, the operational simplicity of the process reduces the burden on manufacturing facilities, allowing for faster turnaround times and more flexible production scheduling. By eliminating the need for high-pressure equipment or cryogenic conditions, capital expenditure requirements for new production lines are significantly lowered, enhancing the overall return on investment for new product introductions.
- Cost Reduction in Manufacturing: The utilization of palladium chloride (PdCl2) as the catalyst represents a significant cost advantage over more exotic and expensive palladium sources often required in cross-coupling reactions. Additionally, the replacement of hazardous carbon monoxide gas with a liquid mixture of formic acid and acetic anhydride removes the need for specialized gas handling infrastructure and safety monitoring systems. This substitution drastically simplifies the reactor setup and reduces operational overheads. The high atom economy of the cascade reaction means less raw material is wasted as byproducts, leading to substantial savings in raw material costs and waste disposal fees, thereby optimizing the overall cost structure for API intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including aromatic amines, propargylamine, and diaryliodonium salts, are commodity chemicals that are widely available from multiple global suppliers. This abundance ensures a resilient supply chain that is less susceptible to disruptions caused by the scarcity of niche reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups, allows for the use of diverse feedstock sources without compromising product quality. This flexibility empowers procurement teams to negotiate better pricing and secure long-term supply contracts, ensuring continuous availability of critical intermediates for downstream drug production.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable to gram levels and beyond, indicating strong potential for multi-kilogram or ton-scale production. The mild reaction conditions and the use of THF as a solvent align well with existing green chemistry initiatives, as THF can be efficiently recovered and recycled. The avoidance of toxic heavy metal oxidants and explosive diazo compounds significantly reduces the environmental footprint of the manufacturing process. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to safety or environmental violations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for integration into their manufacturing portfolios.
Q: What are the primary safety advantages of this synthesis method compared to traditional routes?
A: Unlike traditional methods that often rely on hazardous trifluorodiazoethane, this patent utilizes stable trifluoroethylimidoyl chloride and mild reaction conditions (30°C), significantly reducing explosion risks and operational hazards.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is scalable to gram levels and beyond, utilizing cheap and commercially available starting materials like PdCl2 and simple aryl amines, making it highly suitable for large-scale manufacturing.
Q: What is the functional group tolerance of this palladium-catalyzed reaction?
A: The reaction demonstrates excellent substrate compatibility, tolerating various substituents including methyl, tert-butyl, halogens (Cl, Br), trifluoromethyl, and nitro groups on both the imidoyl chloride and the iodonium salt components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality, fluorinated heterocyclic intermediates for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our capability to implement complex catalytic cascades, such as the palladium-mediated carbonylation described in CN111423381A, positions us as a strategic partner capable of solving your most challenging synthetic problems.
We invite you to collaborate with us to leverage this innovative technology for your specific drug development projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you accelerate your pipeline with reliable, cost-effective, and scalable chemical solutions.
