Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are pivotal motifs in modern drug design due to their enhanced metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in Chinese Patent CN111423381B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology leverages a sophisticated palladium-catalyzed carbonylation strategy that circumvents the limitations of traditional trifluoromethylation routes. By utilizing readily accessible precursors such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, the process achieves remarkable yields under exceptionally mild conditions. The versatility of this approach is underscored by its ability to generate a wide array of structurally diverse imidazole derivatives, which serve as critical scaffolds for bioactive molecules ranging from antihistamines to complex natural product analogs.
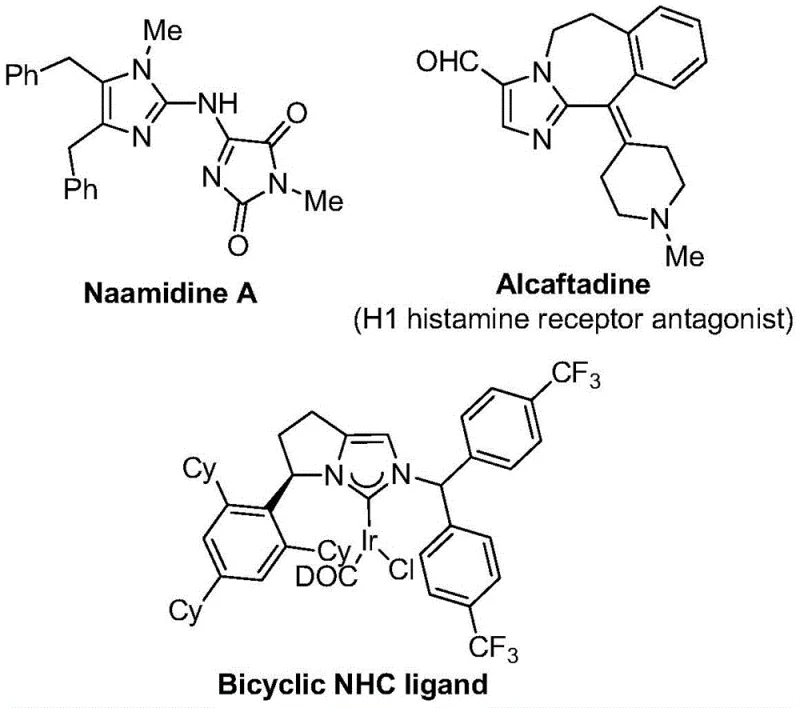
The strategic importance of this synthesis cannot be overstated, as imidazole cores are ubiquitous in high-value active pharmaceutical ingredients (APIs). For instance, compounds like Alcaftadine, an H1 histamine receptor antagonist, rely on similar heterocyclic frameworks. The introduction of the trifluoromethyl group at the 2-position further amplifies the pharmacological potential by modulating electronic properties and binding affinity. As a reliable pharmaceutical intermediate supplier, understanding and implementing such cutting-edge synthetic routes allows us to offer clients superior access to these high-value building blocks. The patent highlights a pathway that is not only chemically elegant but also commercially viable, addressing the growing demand for fluorinated heterocycles in the development of next-generation therapeutics and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant challenges that hinder large-scale adoption. Conventional strategies often rely on the direct reaction of pre-functionalized trifluoromethyl synthons, such as trifluorodiazoethane or specialized hypervalent iodine reagents, with suitable heterocyclic substrates. These reagents are frequently expensive, thermally unstable, and potentially hazardous to handle on an industrial scale. Furthermore, many existing protocols require harsh reaction conditions, including elevated temperatures or the use of strong bases, which can lead to the decomposition of sensitive functional groups and result in complex impurity profiles. The reliance on gaseous carbon monoxide for carbonylation steps in traditional methods also introduces severe safety concerns and necessitates specialized high-pressure equipment, thereby increasing capital expenditure and operational complexity for manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a transition metal palladium-catalyzed multicomponent reaction that operates under remarkably benign conditions. The core innovation lies in the synergistic combination of trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts in the presence of a palladium catalyst and a carbon monoxide surrogate. This methodology eliminates the need for handling toxic CO gas directly, instead generating it in situ from formic acid and acetic anhydride. The reaction proceeds efficiently at a low temperature of 30°C, significantly reducing energy consumption and thermal stress on the reactants. This mild protocol ensures high atom economy and exceptional substrate compatibility, allowing for the seamless integration of diverse functional groups without the need for extensive protecting group strategies.
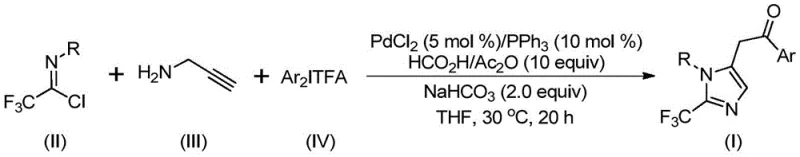
The operational simplicity of this new route is a game-changer for cost reduction in API manufacturing. By employing cheap and commercially available starting materials, the process drastically lowers the raw material costs compared to routes relying on exotic fluorinating agents. The one-pot nature of the reaction minimizes unit operations, reducing solvent usage and waste generation. Moreover, the post-treatment procedure is straightforward, typically involving filtration and standard column chromatography, which facilitates rapid isolation of the pure product. This streamlined workflow not only accelerates the timeline from discovery to production but also enhances the overall sustainability of the manufacturing process, aligning with modern green chemistry principles.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
To fully appreciate the robustness of this synthesis, one must delve into the intricate mechanistic pathway that drives the formation of the 2-trifluoromethyl imidazole core. The reaction initiates with the formation of a trifluoroacetamidine intermediate through an intermolecular carbon-nitrogen bond promotion facilitated by the alkaline environment provided by sodium bicarbonate. This intermediate subsequently undergoes isomerization, setting the stage for the palladium catalytic cycle. The palladium species, generated from palladium chloride and triphenylphosphine, coordinates with the alkyne moiety of the propargylamine derivative. This coordination triggers a palladation event, leading to the formation of an alkenyl palladium intermediate. Through a series of precise isomerization steps, this species converts into a more stable alkyl palladium intermediate, which is poised for the critical carbonylation step.
The carbonylation event is the heart of this transformation, where the in situ generated carbon monoxide inserts into the palladium-carbon bond to form an acyl palladium intermediate. This step is crucial for introducing the carbonyl functionality that becomes part of the final imidazole ring. Following this, the diaryl iodonium salt participates in an oxidative addition to the palladium center, generating a high-valent tetravalent palladium intermediate. This rare oxidation state is key to the success of the reaction, enabling the final reductive elimination step that closes the ring and releases the desired 2-trifluoromethyl-substituted imidazole product while regenerating the active palladium catalyst. This sophisticated cascade ensures high selectivity and minimizes the formation of side products, which is critical for maintaining high purity specifications required in pharmaceutical applications.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in suppressing degradation pathways that are common in high-temperature syntheses. The specific choice of ligands and additives helps to stabilize the palladium intermediates, preventing premature catalyst deactivation or the formation of palladium black. Furthermore, the use of sodium bicarbonate as a mild base avoids the aggressive deprotonation events that could lead to polymerization or decomposition of the sensitive imidoyl chloride starting material. This careful balancing of reaction parameters results in a clean reaction profile, simplifying downstream purification and ensuring that the final product meets stringent quality standards for use as a high-purity pharmaceutical intermediate.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this advanced synthetic route requires strict adherence to the optimized reaction conditions to maximize yield and reproducibility. The process is designed to be user-friendly, utilizing standard laboratory glassware and commercially sourced reagents. The key to success lies in the precise stoichiometric balance of the catalyst system and the effective generation of the carbon monoxide surrogate. Operators should ensure that the reaction mixture is thoroughly degassed and maintained under an inert atmosphere to protect the palladium catalyst from oxidation. The following guide outlines the standardized protocol derived from the patent examples, providing a clear roadmap for laboratory execution and subsequent scale-up efforts.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards this methodology addresses several critical pain points in the sourcing of complex fluorinated intermediates. By leveraging a process that relies on abundant feedstocks and avoids specialized high-pressure infrastructure, organizations can achieve significant cost savings and mitigate supply chain risks. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted production schedules in a regulated environment.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of inexpensive and widely available starting materials such as propargylamine and aniline derivatives, which are commodity chemicals with stable pricing. Unlike traditional methods that depend on costly trifluoromethylating reagents or high-pressure carbon monoxide cylinders, this route generates the necessary carbonyl group in situ from formic acid and acetic anhydride, which are low-cost bulk chemicals. The elimination of expensive heavy metal scavenging steps, often required when using other transition metal catalysts, further contributes to substantial cost savings. Additionally, the mild reaction temperature of 30°C significantly lowers energy consumption compared to processes requiring reflux or cryogenic conditions, resulting in a lower overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the high availability of all key reagents involved in this synthesis. Trifluoroethylimidoyl chloride and diaryl iodonium salts can be sourced from multiple global suppliers, reducing the risk of single-source bottlenecks. The simplicity of the reaction setup means that production can be easily transferred between different manufacturing sites without the need for specialized reactor modifications. This flexibility ensures continuity of supply even in the face of regional disruptions. Furthermore, the short reaction time of approximately 20 hours allows for faster turnover rates, enabling manufacturers to respond more agilely to fluctuating market demands and reducing inventory holding times.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways for expansion to kilogram and ton scales. The absence of hazardous gases and the use of relatively benign solvents like tetrahydrofuran simplify waste management and regulatory compliance. The high atom economy of the multicomponent reaction minimizes the generation of chemical waste, aligning with increasingly strict environmental regulations. The straightforward workup procedure, involving simple filtration and chromatography, reduces the volume of solvent waste and streamlines the purification process. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines.
Q: What are the primary advantages of this novel synthesis method over conventional trifluoromethylation techniques?
A: Unlike conventional methods that often require hazardous trifluoromethylating reagents or harsh conditions, this patent-disclosed process utilizes cheap and readily available starting materials like propargylamine and trifluoroethylimidoyl chloride. It operates at a mild 30°C, offers excellent substrate compatibility, and achieves high reaction efficiency without the need for high-pressure carbon monoxide gas.
Q: What is the scope of substrate compatibility for this palladium-catalyzed reaction?
A: The method demonstrates broad functional group tolerance, accommodating various substituted aryl groups on both the imidoyl chloride and the diaryl iodonium salt components. Substituents such as methyl, tert-butyl, halogens (chlorine, bromine), trifluoromethyl, and nitro groups are well-tolerated, allowing for the synthesis of diverse derivatives suitable for medicinal chemistry optimization.
Q: Is this synthetic route suitable for industrial scale-up and large-scale production?
A: Yes, the process is designed for scalability. The operation is simple, involving a one-pot reaction with easily removable byproducts. The use of stable catalysts and mild temperatures reduces energy consumption and safety risks associated with high-pressure reactions, making it highly viable for transition from gram-level laboratory synthesis to kilogram or ton-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex fluorinated chemistries positions us as a strategic partner for companies seeking to secure a stable supply of critical building blocks for drug development.
We invite you to collaborate with us to leverage this innovative synthesis method for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget without compromising quality. We encourage you to contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom targets. By partnering with us, you gain access to a reliable supply chain backed by deep technical expertise and a commitment to excellence in fine chemical manufacturing.
