Advanced Indole Phosphine Ligands for Efficient Cross-Coupling and Pharmaceutical Manufacturing
Advanced Indole Phosphine Ligands for Efficient Cross-Coupling and Pharmaceutical Manufacturing
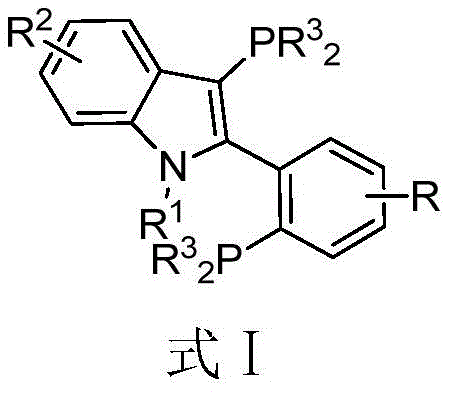
The landscape of transition metal catalysis is constantly evolving, driven by the relentless demand for more efficient, selective, and cost-effective synthetic methodologies in the pharmaceutical and fine chemical industries. A significant breakthrough in this domain is documented in Chinese Patent CN107445989B, which introduces a novel class of 3-(disubstituted phosphino)-2-(2-(disubstituted phosphino) substituted phenyl)-1-alkyl-indole skeleton phosphine ligands. These specialized organophosphorus compounds represent a substantial leap forward in ligand design, offering a unique combination of steric bulk and electronic tunability that addresses many of the limitations inherent in earlier generations of catalysts. For R&D directors and process chemists, the ability to access such high-performance ligands through a streamlined synthetic route is paramount, as it directly impacts the feasibility of complex molecule assembly. The patent details a robust preparation method that leverages classic organic transformations, ensuring that these advanced tools are not just theoretical constructs but practical assets for industrial application.
From a supply chain and procurement perspective, the introduction of this technology signals a shift towards more reliable and economically viable catalytic systems. The core innovation lies in the specific structural architecture of the ligand, which features an indole backbone substituted with phosphine groups at strategic positions. This configuration allows for the formation of exceptionally stable complexes with transition metals, particularly palladium, thereby enhancing catalytic turnover and longevity. As we delve deeper into the technical specifics, it becomes evident that this invention is not merely an incremental improvement but a foundational advancement that supports the synthesis of diverse sulfur-containing compounds and biaryl structures essential for modern drug discovery. The implications for reducing lead time for high-purity pharmaceutical intermediates are profound, offering a pathway to accelerate development timelines while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of cross-coupling reactions has relied heavily on a variety of phosphine ligands, such as the tri-tert-butylphosphine developed by the Fu research group or the biaryl phosphine ligands from the Buchwald research group. While these ligands have served the industry well, they often present significant challenges when applied to specific, sterically demanding substrates or sensitive functional groups. Conventional ligands may suffer from issues related to oxidative instability, leading to degradation during storage or reaction, which compromises the consistency of the catalytic performance. Furthermore, the synthesis of some high-performance ligands can be prohibitively complex, involving multiple steps with low overall yields and the use of hazardous reagents, which escalates the cost of goods sold (COGS) and complicates waste management protocols. In the context of large-scale manufacturing, these inefficiencies translate into higher production costs and increased supply chain vulnerability, as the availability of such specialized ligands can be inconsistent.
The Novel Approach
The novel approach detailed in the patent circumvents these traditional bottlenecks by utilizing an indole scaffold that is both robust and easily modifiable. The synthesis strategy employs a Fischer indole synthesis as the foundational step, a reaction renowned for its reliability and scalability. By starting with commercially abundant precursors like 2'-bromoacetophenone and phenylhydrazines, the process ensures a steady supply of raw materials, mitigating the risks associated with sourcing exotic starting materials. The subsequent functionalization steps, including bromination and phosphorylation, are optimized to maximize yield and purity, resulting in a final ligand product that exhibits superior air and moisture stability compared to many existing alternatives. This stability is a critical commercial advantage, as it simplifies logistics and storage requirements, allowing procurement teams to manage inventory more effectively without the need for stringent inert atmosphere conditions. The result is a ligand system that not only performs exceptionally in catalytic applications but also aligns with the economic and operational realities of modern chemical manufacturing.
Mechanistic Insights into Indole-Based Bidentate Phosphine Coordination
The exceptional catalytic performance of these indole-based ligands can be attributed to their unique bidentate coordination mode and the electronic properties imparted by the indole nitrogen and the phosphine substituents. When complexed with a transition metal like palladium, the ligand forms a stable chelate structure that protects the metal center from deactivation pathways such as aggregation or oxidation. The steric environment around the metal is precisely tuned by the substituents on the phosphorus atoms (R3 groups) and the indole nitrogen (R1 group), allowing chemists to fine-tune the catalyst for specific reaction profiles. For instance, bulky cyclohexyl groups on the phosphorus atoms create a protective pocket that facilitates the oxidative addition of aryl halides while preventing unwanted side reactions. This level of control is essential for the synthesis of complex pharmaceutical intermediates where regioselectivity and chemoselectivity are non-negotiable requirements. The mechanistic robustness of this system ensures consistent performance across different batches, a key metric for quality assurance in regulated industries.
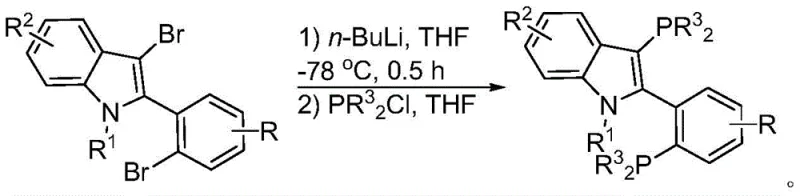
Furthermore, the synthetic pathway itself is a masterclass in efficient process chemistry, designed to minimize impurities and maximize throughput. The initial Fischer indole cyclization creates the core heterocyclic structure with high fidelity, setting the stage for subsequent functionalization. The bromination step, typically achieved using N-bromosuccinimide (NBS), introduces the necessary leaving groups for the final phosphorylation. The critical lithiation step, performed at cryogenic temperatures (-75 to -80°C), generates the reactive organolithium species which then attacks the chlorophosphine electrophiles. This sequence ensures that the phosphine groups are installed at the precise 2 and 3 positions of the indole ring, creating the desired bidentate geometry. The ability to execute these transformations with high selectivity minimizes the formation of regioisomers, thereby simplifying downstream purification and reducing solvent consumption. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the chemical behavior is well-understood and predictable.
How to Synthesize 3-(Disubstituted Phosphino)-2-(2-(Disubstituted Phosphino) Substituted Phenyl)-1-Alkyl-Indole Efficiently
The synthesis of these high-value ligands is achieved through a logical and scalable four-step sequence that balances reaction efficiency with operational simplicity. The process begins with the condensation of 2'-bromoacetophenone and an N-alkyl phenylhydrazine in the presence of phosphoric acid, followed by cyclization using polyphosphoric acid at elevated temperatures (80-120°C) to form the indole core. This intermediate is then subjected to controlled bromination using NBS in DMF to introduce bromine atoms at the 2 and 3 positions of the indole ring. The resulting dibromo intermediate serves as the precursor for the final dual phosphorylation. In the concluding step, the dibromo compound is treated with n-butyllithium at -78°C to generate the dilithio species, which is subsequently quenched with a disubstituted phosphine chloride to yield the target ligand. Detailed standardized synthesis steps for this protocol are provided in the guide below, ensuring reproducibility and safety during implementation.
- Perform Fischer indole synthesis by reacting 2'-bromoacetophenone with N-alkyl phenylhydrazine using phosphoric acid and polyphosphoric acid at 80-120°C.
- Conduct selective bromination of the indole intermediate using N-bromosuccinimide (NBS) in dimethylformamide at room temperature.
- Execute double lithiation at -78°C followed by quenching with disubstituted phosphine chloride to install the phosphine groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology offers tangible benefits that extend beyond mere catalytic performance. The primary advantage lies in the significant reduction of manufacturing complexity and cost. By utilizing a synthetic route that relies on commodity chemicals and avoids the need for precious metal catalysts during the ligand synthesis itself, the overall production cost is drastically simplified. The high total yield of the process means that less raw material is wasted, directly contributing to cost reduction in fine chemical manufacturing. Moreover, the stability of the final ligand product eliminates the need for specialized cold-chain logistics or glovebox handling during transport and storage, which further lowers the total cost of ownership. This robustness ensures a continuous supply of high-quality catalysts, reducing the risk of production delays caused by reagent degradation or scarcity.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent is inherently economical, utilizing inexpensive starting materials such as bromoacetophenone and simple hydrazines. The elimination of complex purification steps and the high efficiency of the Fischer indole synthesis contribute to a leaner manufacturing process. By avoiding the use of expensive transition metals in the ligand synthesis phase, the process minimizes capital expenditure on catalyst recovery systems. This economic efficiency allows for competitive pricing strategies, making advanced catalytic solutions accessible for a broader range of applications without compromising on quality or performance metrics.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that the supply chain for these ligands is resilient against market fluctuations. Unlike specialized ligands that depend on niche precursors with long lead times, the raw materials for this indole scaffold are produced globally in large volumes. This abundance guarantees a steady flow of inputs, enabling manufacturers to maintain consistent production schedules. Additionally, the air and moisture stability of the final product simplifies inventory management, allowing for longer shelf lives and reduced waste due to expiration. This reliability is crucial for maintaining the continuity of downstream pharmaceutical production lines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as heating, stirring, and extraction that are easily transferable from pilot plant to commercial scale. The use of common solvents like THF and DMF, while requiring proper handling, fits within established waste management frameworks, facilitating compliance with environmental regulations. The high atom economy of the reaction steps minimizes the generation of hazardous byproducts, aligning with green chemistry principles. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity, a key consideration for modern supply chain stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for integrating this new catalytic system into existing workflows and maximizing its potential value. The insights provided here aim to clarify the operational parameters and expected outcomes, ensuring that all stakeholders have a clear understanding of the technology's capabilities and limitations.
Q: What are the key advantages of this indole-based phosphine ligand over traditional biaryl ligands?
A: The indole skeleton provides a rigid yet tunable framework that enhances the stability of the palladium complex. Unlike traditional monodentate ligands, this bidentate structure offers superior catalytic activity in challenging cross-coupling reactions, particularly for carbon-sulfur bond formation, with yields reaching up to 90%.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent outlines a method using readily available raw materials like 2'-bromoacetophenone and standard reagents. The process avoids exotic catalysts and utilizes common solvents like THF and DMF, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Q: How does this ligand improve cost efficiency in pharmaceutical manufacturing?
A: By enabling lower catalyst loadings (as low as 0.5 mol% Pd) and achieving high conversion rates, the ligand reduces the overall consumption of expensive palladium metals. Additionally, the air and moisture stability of the ligand simplifies storage and handling, further reducing operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced ligand technologies like the 3-(disubstituted phosphino)-2-(2-(disubstituted phosphino) substituted phenyl)-1-alkyl-indole scaffold in driving innovation in pharmaceutical synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or intermediate we produce meets the highest international standards. We understand that consistency is key in catalytic applications, and our commitment to quality assurance ensures that your processes run smoothly without interruption.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production needs, demonstrating how switching to this ligand system can optimize your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain and deep technical expertise, empowering you to accelerate your drug development timelines and bring life-saving therapies to market faster.
