Revolutionizing Asymmetric Catalysis With Novel N-N Axis Chiral Bis-Indole Phosphine Ligands
Revolutionizing Asymmetric Catalysis With Novel N-N Axis Chiral Bis-Indole Phosphine Ligands
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for higher enantiomeric purity and more efficient catalytic systems in the pharmaceutical industry. A significant breakthrough in this domain is documented in patent CN116041393B, which introduces a novel class of N-N axis chiral bis-indole phosphine ligands. Unlike traditional ligands derived from binaphthyl skeletons, these new structures leverage the unique electronic and steric properties of the indole moiety to create a distinct chiral environment. This innovation addresses critical limitations in current catalytic technologies, offering a robust platform for constructing complex chiral molecules. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic advantages and commercial viability of this technology is paramount for future-proofing supply chains and enhancing process efficiency.
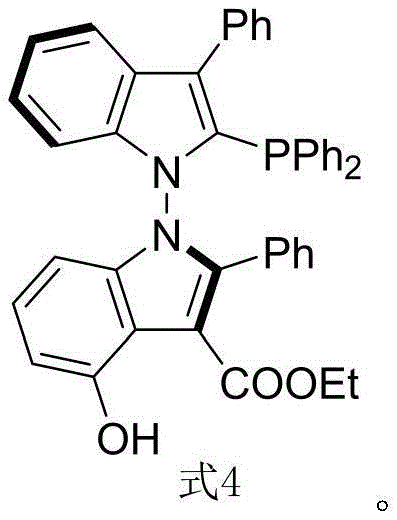
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has been dominated by axially chiral ligands based on the binaphthyl scaffold, such as BINAP and BINOL derivatives. While effective, these conventional systems often suffer from inherent rigidity that limits the tunability of the dihedral angle, a critical parameter for optimizing stereoselectivity in diverse substrate classes. Furthermore, the synthesis of these traditional ligands frequently involves harsh conditions, expensive resolving agents, or multi-step sequences that generate substantial chemical waste. The lack of functional group diversity on the binaphthyl core also restricts the ability to introduce secondary interactions, such as hydrogen bonding, which are increasingly recognized as vital for activating challenging substrates. Consequently, there is a pressing need for alternative scaffolds that offer greater conformational flexibility and enhanced interaction capabilities without compromising stability or ease of synthesis.
The Novel Approach
The methodology outlined in the patent presents a paradigm shift by utilizing an N-N axial chirality framework built upon a bis-indole skeleton. This approach capitalizes on the rich chemistry of indoles to construct a ligand backbone that provides a wider space for dihedral angle regulation compared to its binaphthyl counterparts. The synthesis begins with a highly enantioselective organocatalytic coupling between an indole-derived enamine and a 2,3-diketone ester, facilitated by a chiral phosphoramide catalyst. This step is remarkable for its mild conditions, operating at 70°C in 1,1,2,2-tetrachloroethane with hexafluoroisopropanol as an additive. The subsequent conversion to the final phosphine ligand involves a straightforward deoxygenative reduction, preserving the high optical purity established in the first step. This two-step route represents a significant simplification over traditional methods, drastically reducing the complexity and cost associated with producing high-performance chiral ligands.
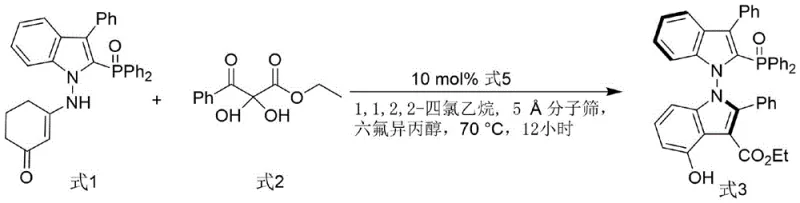
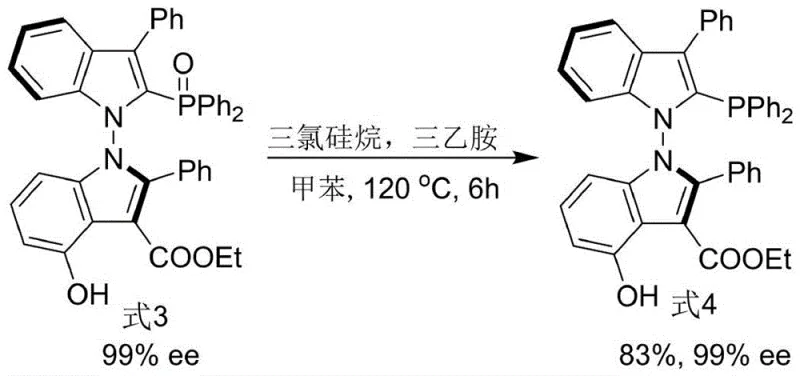
Mechanistic Insights into Organocatalytic Axial Chirality Construction
The core innovation lies in the first synthetic step, where axial chirality is induced via an organocatalytic reaction rather than traditional resolution or metal catalysis. The chiral phosphoramide catalyst activates the electrophilic 2,3-diketone ester through hydrogen bonding, while simultaneously organizing the nucleophilic indole enamine within a well-defined chiral pocket. The use of 5Å molecular sieves is critical here, acting as a dehydrating agent to drive the equilibrium towards the condensation product by removing water generated during the reaction. Hexafluoroisopropanol (HFIP) serves as a crucial additive, likely enhancing the acidity of the catalyst and stabilizing transition states through strong hydrogen bond donor interactions. This synergistic effect allows the reaction to proceed with high stereocontrol, yielding the intermediate bis-indole derivative with an initial enantiomeric excess of 82%, which can be upgraded to 99% ee through simple recrystallization. This mechanism underscores the power of non-covalent interactions in dictating stereochemical outcomes.
Following the construction of the chiral backbone, the transformation of the phosphine oxide intermediate into the active phosphine ligand is achieved using trichlorosilane and triethylamine in toluene at 120°C. This deoxygenation step is chemically robust and does not erode the axial chirality established earlier, demonstrating the thermal stability of the N-N axis under these reducing conditions. The resulting ligand features a tertiary phosphine group strategically positioned to coordinate with transition metals, such as palladium, creating a highly congested chiral environment. In application, specifically in asymmetric allylation reactions, this ligand forms a cationic pi-allyl palladium complex where the bulky indole substituents effectively shield one face of the allyl system. This steric differentiation forces the nucleophile to attack from the less hindered trajectory, resulting in products with excellent enantioselectivity, as evidenced by the 90% ee achieved in the model allylation reaction described in the patent data.
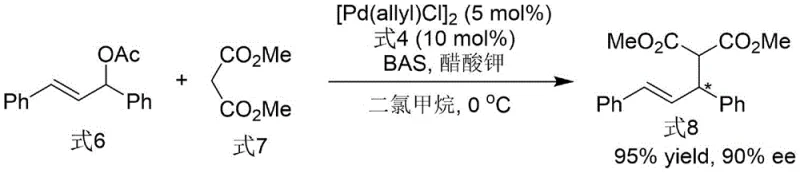
How to Synthesize N-N Axis Chiral Bis-Indole Phosphine Ligand Efficiently
The synthesis of this advanced ligand is designed for operational simplicity and scalability, making it an attractive candidate for industrial adoption. The process avoids the use of sensitive organometallic reagents in the initial bond-forming step, relying instead on stable organic catalysts and readily available starting materials. The reaction conditions are moderate, avoiding cryogenic temperatures or ultra-high pressures that often complicate scale-up. Purification is streamlined through standard silica gel chromatography and recrystallization techniques, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications. For technical teams looking to implement this chemistry, the following standardized protocol outlines the critical parameters for achieving optimal yield and stereocontrol.
- Condense indole-derived enamine with 2,3-diketone ester using chiral phosphoramide catalyst and molecular sieves at 70°C.
- Purify the intermediate bis-indole derivative via silica gel column chromatography and recrystallization to enhance enantiomeric excess.
- Perform deoxygenative reduction of the phosphine oxide intermediate using trichlorosilane and triethylamine in toluene at 120°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-N axis chiral ligand technology offers compelling advantages for cost reduction in pharmaceutical intermediate manufacturing. The synthetic route eliminates the need for expensive transition metal catalysts during the ligand construction phase, relying instead on organocatalysis which significantly lowers raw material costs. Furthermore, the high enantioselectivity achieved directly in the synthesis minimizes the need for costly downstream purification processes such as preparative chiral HPLC, which are often the bottleneck in producing optically pure intermediates. The use of commodity chemicals like toluene, triethylamine, and trichlorosilane ensures that the supply chain remains resilient and less susceptible to fluctuations in the pricing of specialized reagents. This robustness translates into a more predictable cost structure for large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in the ligand synthesis step represents a direct saving on bill of materials. Additionally, the high efficiency of the organocatalytic step reduces the overall processing time and energy consumption compared to multi-step resolution protocols. By achieving high enantiomeric excess early in the sequence, the process minimizes material loss associated with discarding unwanted enantiomers, thereby improving the overall mass balance and atom economy of the operation. These factors collectively contribute to a substantially lower cost of goods sold for the final chiral ligand.
- Enhanced Supply Chain Reliability: The starting materials, including indole derivatives and diketone esters, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The reaction conditions are not sensitive to trace moisture or oxygen to the same extent as many organometallic processes, allowing for more flexible manufacturing schedules and reduced requirements for specialized inert atmosphere equipment. This operational flexibility ensures consistent delivery timelines and enhances the reliability of the supply chain for downstream customers who depend on steady flows of high-quality chiral building blocks.
- Scalability and Environmental Compliance: The process utilizes solvents that are amenable to recovery and recycling, aligning with modern green chemistry principles and environmental regulations. The absence of heavy metal residues in the ligand synthesis simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing facility. The thermal stability of the intermediates allows for safe handling and storage, facilitating the logistics of transporting bulk quantities. These attributes make the technology highly suitable for commercial scale-up of complex pharmaceutical intermediates, ensuring that production can be ramped up to meet market demand without compromising safety or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what potential users can expect. Understanding these details is crucial for evaluating the fit of this technology within existing process workflows and for making informed decisions about technology transfer and licensing.
Q: What distinguishes this N-N axis ligand from traditional BINAP ligands?
A: Unlike rigid binaphthyl scaffolds, the N-N axis bis-indole structure offers tunable dihedral angles and additional hydrogen-bonding sites, enabling superior stereocontrol in challenging transformations.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes mild conditions (70°C and 120°C), commercially available solvents like toluene and tetrachloroethane, and avoids expensive transition metals in the ligand construction phase, facilitating scale-up.
Q: What level of enantioselectivity can be achieved?
A: The patented method achieves up to 99% ee after recrystallization of the intermediate, maintaining high optical purity (99% ee) in the final phosphine ligand product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bis-Indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this N-N axis chiral ligand technology in advancing asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and trace metal analysis. We are committed to delivering high-purity OLED material and pharmaceutical intermediates that meet the exacting standards of the global market, leveraging our deep expertise in process optimization and quality assurance.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of this technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the integration of this superior ligand system into your supply chain with confidence. Together, we can drive efficiency and innovation in the production of next-generation chiral compounds.
