Revolutionizing Triazole Synthesis: Iodine-Catalyzed Process Enables Scalable Production of High-Purity Pharmaceutical Intermediates
Patent CN113105402B introduces a groundbreaking methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses critical limitations in current pharmaceutical intermediate production. This innovative approach leverages iodine-catalyzed chemistry to construct complex triazole frameworks containing both trifluoromethyl and acyl functionalities—molecular features essential for enhancing drug bioavailability and metabolic stability as demonstrated in landmark pharmaceuticals like sitagliptin and maraviroc. The process operates under standard atmospheric conditions without requiring anhydrous or oxygen-free environments, representing a significant departure from conventional synthetic routes that often demand specialized equipment and handling procedures. By eliminating the need for toxic heavy metal catalysts while maintaining high reaction efficiency, this methodology offers pharmaceutical manufacturers a more sustainable and economically viable pathway to produce these valuable heterocyclic building blocks. The patent demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining stringent purity specifications required for pharmaceutical applications. This advancement comes at a critical time when the industry seeks more environmentally responsible synthetic methodologies that can be readily implemented across global manufacturing networks without extensive retooling or process validation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing multi-substituted triazole frameworks typically rely on transition metal-catalyzed methodologies that present significant challenges for pharmaceutical manufacturing scale-up. These conventional routes often require strictly anhydrous and oxygen-free conditions that necessitate specialized equipment and extensive process validation before implementation in commercial production facilities. The use of expensive and toxic heavy metal catalysts such as palladium or copper creates additional downstream processing complexities including costly metal removal steps and associated waste treatment procedures that substantially increase overall production costs while generating environmental concerns. Furthermore, many existing methodologies struggle with the simultaneous incorporation of both trifluoromethyl and acyl functional groups into the triazole molecular architecture—a critical limitation given the importance of these structural features in enhancing drug bioavailability and metabolic stability as evidenced by blockbuster pharmaceuticals like sitagliptin and maraviroc. The narrow substrate scope and functional group tolerance of conventional methods also restrict their applicability across diverse molecular scaffolds required by modern drug discovery programs. These combined limitations create substantial barriers to efficient commercial production of triazole-based pharmaceutical intermediates at the scale required by global pharmaceutical supply chains.
The Novel Approach
The patented methodology presented in CN113105402B overcomes these critical limitations through an innovative iodine-catalyzed synthetic pathway that operates under standard atmospheric conditions without requiring specialized equipment or handling procedures. This approach utilizes readily available aryl ethyl ketones and trifluoroethylimide hydrazides as starting materials that can be sourced from multiple global suppliers at competitive prices while maintaining consistent quality specifications required for pharmaceutical manufacturing. The elimination of toxic heavy metal catalysts not only reduces raw material costs but also eliminates expensive metal removal steps and associated waste treatment processes that significantly impact overall production economics. The reaction demonstrates exceptional functional group tolerance across diverse substrate combinations while maintaining high yields across a broad range of molecular architectures relevant to pharmaceutical applications. Crucially, this methodology enables the simultaneous incorporation of both trifluoromethyl and acyl functionalities into the triazole framework—a capability that addresses a significant gap in existing synthetic approaches as highlighted in the patent's background section referencing Science (2007) literature on trifluoromethyl group benefits. The process has been successfully demonstrated at gram-scale with straightforward purification protocols that can be readily adapted to commercial manufacturing environments without extensive revalidation requirements.
Mechanistic Insights into Iodine-Catalyzed Triazole Synthesis
The reaction mechanism begins with iodine-mediated Kornblum oxidation of aryl ethyl ketones in dimethyl sulfoxide solvent to form aryl diketone intermediates—a critical transformation that occurs efficiently at elevated temperatures (90–110°C) without requiring specialized reaction conditions. This initial step generates the necessary electrophilic species that subsequently undergo condensation with trifluoroethylimide hydrazides to form hydrazone intermediates through dehydration reactions facilitated by the pyridine base present in the reaction mixture. The sodium dihydrogen phosphate serves as a crucial buffer system that maintains optimal pH conditions throughout the reaction sequence while preventing unwanted side reactions that could compromise product purity or yield. The iodine catalyst then promotes an intramolecular cyclization process through electrophilic activation of the hydrazone nitrogen atoms, enabling nucleophilic attack by the carbonyl oxygen to form the characteristic triazole ring structure with simultaneous elimination of water molecules. This cascade reaction proceeds through a well-defined mechanistic pathway that avoids the formation of persistent reactive intermediates that could lead to impurity generation—a critical advantage for pharmaceutical manufacturing where strict impurity profiles must be maintained.
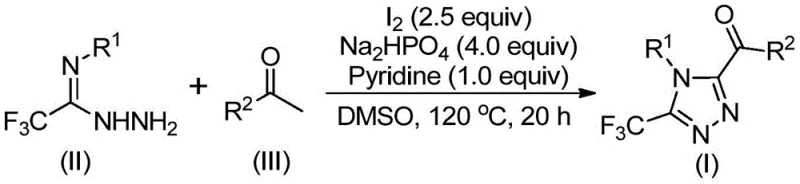
The impurity control mechanism is particularly noteworthy as it leverages the inherent selectivity of the iodine-catalyzed cyclization process to minimize unwanted side products that commonly plague traditional triazole syntheses. The carefully optimized molar ratio of iodine (2.5 equivalents), sodium dihydrogen phosphate (4 equivalents), and pyridine (1 equivalent) creates a balanced reaction environment that suppresses competing pathways while promoting high regioselectivity in the ring closure step—ensuring consistent formation of the desired 3,4,5-trisubstituted isomer without significant formation of regioisomeric impurities. The use of dimethyl sulfoxide as both solvent and participant in the initial oxidation step provides additional control over reaction kinetics by modulating intermediate stability and reactivity profiles throughout the transformation sequence. Post-reaction purification through standard column chromatography on silica gel effectively removes any residual starting materials or minor byproducts without requiring specialized techniques or equipment that would complicate scale-up efforts. This comprehensive impurity management strategy ensures that final products consistently meet the stringent purity specifications required for pharmaceutical intermediates while maintaining excellent process robustness across multiple production batches.
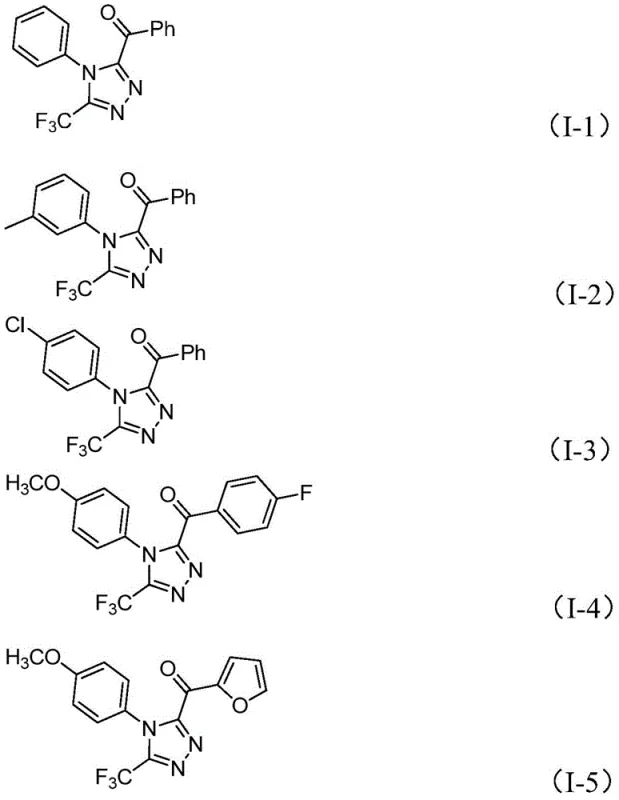
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
This patented methodology represents a significant advancement in triazole synthesis by eliminating traditional barriers to commercial implementation while maintaining high product quality standards required by pharmaceutical manufacturers. The process begins with readily available aryl ethyl ketones that undergo iodination/Kornblum oxidation in dimethyl sulfoxide solvent to form key aryl diketone intermediates—a transformation that occurs efficiently under standard atmospheric conditions without requiring specialized equipment or handling procedures. The subsequent addition of trifluoroethylimide hydrazides with precisely controlled stoichiometry of sodium dihydrogen phosphate and pyridine enables a tandem cyclization process that constructs the complex triazole framework with simultaneous incorporation of both trifluoromethyl and acyl functional groups essential for pharmaceutical applications. Detailed standardized synthesis procedures have been developed based on this patent disclosure to ensure consistent product quality across different manufacturing scales while maintaining optimal process economics.
- Prepare aryl ethyl ketone and iodine in DMSO solvent at 90-110°C for 4-6 hours to form aryl diketone intermediate through iodination/Kornblum oxidation
- Add trifluoroethylimide hydrazide with sodium dihydrogen phosphate and pyridine to the reaction mixture at molar ratio of 4: 1:2.5
- Heat the mixture to 110-130°C for 12-20 hours to complete cyclization reaction and obtain the final triazole product after standard purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach directly addresses multiple pain points experienced by procurement and supply chain professionals managing complex pharmaceutical intermediate sourcing strategies across global manufacturing networks. The elimination of toxic heavy metal catalysts removes significant supply chain vulnerabilities associated with specialized catalyst procurement and handling requirements while reducing regulatory compliance burdens across different geographical regions with varying environmental regulations. The use of readily available starting materials from multiple global suppliers creates enhanced supply chain resilience by avoiding single-source dependencies that could disrupt production schedules during market fluctuations or geopolitical events.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts removes substantial raw material costs while simultaneously eliminating costly metal removal steps and associated waste treatment processes that significantly impact overall production economics. The use of standard laboratory equipment without requiring specialized anhydrous or oxygen-free environments reduces capital investment requirements and simplifies facility qualification processes across different manufacturing sites globally.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials from multiple global suppliers creates enhanced supply chain resilience by avoiding single-source dependencies that could disrupt production schedules during market fluctuations or geopolitical events. The straightforward reaction conditions enable rapid technology transfer between manufacturing sites with minimal revalidation requirements—ensuring consistent product quality across different geographical locations while maintaining flexible production capacity allocation.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production volumes without requiring significant process modifications enables seamless transition from development to manufacturing phases while maintaining stringent quality specifications required for pharmaceutical applications. The elimination of toxic heavy metal catalysts significantly reduces environmental impact by avoiding hazardous waste streams associated with metal removal processes—aligning with global sustainability initiatives while simplifying regulatory compliance across different geographical markets.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams evaluating new synthetic methodologies for pharmaceutical intermediate production. These responses are based on detailed analysis of patent CN113105402B's technical disclosures and experimental data supporting the commercial viability of this innovative synthetic approach.
Q: How does this iodine-catalyzed method improve upon traditional heavy metal-catalyzed approaches for triazole synthesis?
A: This novel method eliminates the need for toxic heavy metal catalysts while maintaining high reaction efficiency. The iodine-based catalytic system avoids costly metal removal steps and associated waste treatment processes that are required in conventional methodologies.
Q: What are the key advantages of this synthesis method for pharmaceutical manufacturing scale-up?
A: The process operates under standard atmospheric conditions without requiring anhydrous or oxygen-free environments. It uses readily available starting materials and demonstrates excellent scalability from laboratory to commercial production volumes while maintaining stringent purity specifications.
Q: How does this method address the challenge of incorporating both trifluoromethyl and acyl groups into triazole structures?
A: The reaction pathway specifically enables simultaneous introduction of trifluoromethyl and acyl functionalities through a tandem cyclization process. This strategic approach overcomes limitations in existing methodologies that struggle with dual functionalization of triazole molecular frameworks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Supplier
Our patented iodine-catalyzed methodology represents a significant advancement in triazole synthesis technology that directly addresses critical challenges faced by global pharmaceutical manufacturers seeking more sustainable and economically viable production pathways for complex heterocyclic intermediates. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications through our state-of-the-art QC labs and rigorous analytical protocols. Our commitment to process excellence ensures consistent product quality across all manufacturing scales while providing flexible supply chain solutions tailored to meet specific customer requirements in dynamic global markets.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your unique application requirements through our Customized Cost-Saving Analysis service—designed to help you identify optimal manufacturing pathways that balance quality requirements with economic considerations for your specific product portfolio.
