Revolutionizing Triazole Compound Production: Scalable Metal-Free Synthesis for Pharmaceutical Intermediates
The patent CN113105402B introduces a groundbreaking methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds through an iodine-catalyzed cascade reaction. This innovation addresses critical limitations in traditional triazole synthesis by eliminating the need for transition metal catalysts and specialized reaction environments. The process leverages commercially available aryl ethyl ketones and trifluoroethylimide hydrazides as starting materials, operating under standard atmospheric conditions without requiring anhydrous or oxygen-free setups. This represents a significant advancement for pharmaceutical manufacturers seeking sustainable routes to complex heterocyclic intermediates essential in drug development pipelines. The methodology's compatibility with diverse functional groups expands the structural diversity achievable in triazole-based pharmacophores while maintaining operational simplicity across production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of polysubstituted triazoles frequently require transition metal catalysts such as copper or palladium systems that necessitate expensive purification steps to remove toxic metal residues from final products. These processes often operate under strictly controlled anhydrous and oxygen-free conditions that demand specialized equipment and increase operational complexity. The narrow substrate scope of many existing methods limits structural diversity in the resulting triazole compounds, restricting their applicability in drug discovery programs. Furthermore, the high cost of metal catalysts and the need for rigorous environmental controls significantly elevate production expenses while introducing potential supply chain vulnerabilities through reliance on scarce metal resources. The multi-step nature of conventional approaches also generates substantial waste streams that complicate regulatory compliance and increase environmental impact.
The Novel Approach
The patented methodology overcomes these limitations through a streamlined two-stage process utilizing elemental iodine as a non-toxic catalyst in dimethyl sulfoxide solvent. This system eliminates the need for transition metals entirely while maintaining high functional group tolerance across diverse aryl substrates. The reaction proceeds efficiently under standard atmospheric conditions without requiring specialized moisture or oxygen controls, significantly simplifying facility requirements. The process demonstrates remarkable substrate flexibility with R1 and R2 groups accommodating various substituted aryl and heteroaryl moieties including halogenated and trifluoromethyl variants. Crucially, the methodology achieves consistent yields across multiple substrate combinations while operating within practical temperature ranges (90-130°C) that align with standard industrial reactor capabilities. This approach represents a paradigm shift toward sustainable triazole synthesis that maintains pharmaceutical-grade purity standards without compromising operational efficiency.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with iodine-mediated oxidation of aryl ethyl ketones to form aryl diketone intermediates through a Kornblum-type process in DMSO solvent. This initial transformation occurs at 90-110°C over 4-6 hours without requiring additional oxidants or catalysts. The resulting diketone then undergoes condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate in the presence of sodium dihydrogen phosphate and pyridine additives. The phosphate buffer maintains optimal pH conditions while pyridine facilitates imine formation through proton transfer catalysis. This dual-additive system creates a synergistic environment that promotes efficient hydrazone formation without side reactions.
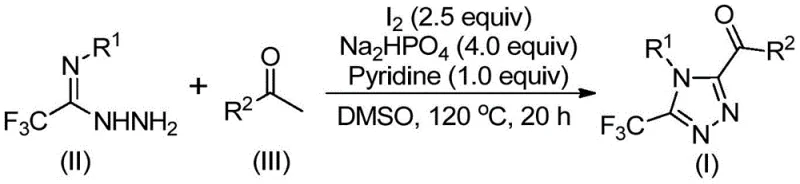
The final cyclization step proceeds through an intramolecular nucleophilic attack where the hydrazone nitrogen attacks the carbonyl carbon, forming the triazole ring structure. Iodine plays a dual role in this stage by activating the carbonyl group while simultaneously facilitating dehydration through its oxidizing properties. The phosphate buffer prevents acid-catalyzed decomposition pathways that commonly generate impurities in traditional triazole syntheses. This carefully balanced catalytic system ensures high regioselectivity at the 3,4,5 positions while minimizing common side products such as open-chain hydrazones or over-oxidized byproducts. The resulting triazole compounds consistently demonstrate high purity profiles suitable for pharmaceutical applications without requiring extensive post-synthesis purification beyond standard column chromatography.
How to Synthesize Triazole Intermediates Efficiently
This innovative synthesis pathway provides pharmaceutical manufacturers with a practical route to high-value triazole intermediates through a reproducible three-stage process that maintains exceptional operational simplicity. The methodology eliminates complex catalyst handling procedures while delivering consistent product quality across diverse substrate combinations. Detailed standardized synthesis steps for manufacturing scale-up are provided below to ensure seamless technology transfer from laboratory to production environments.
- Prepare aryl diketone intermediate by reacting aryl ethyl ketone with iodine in DMSO at 90-110°C for 4-6 hours under standard atmospheric conditions
- Form hydrazone intermediate by adding trifluoroethylimide hydrazide, sodium dihydrogen phosphate, and pyridine to the reaction mixture
- Complete cyclization by heating to 110-130°C for 12-20 hours followed by silica gel chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in specialty chemical manufacturing. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement, handling, and removal processes while enhancing product safety profiles for pharmaceutical applications. The simplified reaction setup reduces capital expenditure requirements for specialized equipment while improving production flexibility across existing manufacturing facilities.
- Cost Reduction in Manufacturing: The complete avoidance of expensive transition metal catalysts eliminates both procurement costs and downstream purification expenses required to meet stringent metal residue specifications in pharmaceutical intermediates. The use of commodity chemicals as starting materials combined with standard solvent systems creates significant raw material cost advantages over conventional approaches requiring specialized reagents. Process simplification through elimination of anhydrous/oxygen-free requirements reduces utility consumption and operational complexity while maintaining consistent product quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including aryl ethyl ketones and iodine ensures robust supply chain resilience against market fluctuations that commonly affect specialized chemical reagents. The atmospheric reaction conditions eliminate dependencies on specialized gas supplies or moisture-sensitive handling protocols that frequently cause production delays. This methodology's demonstrated scalability from laboratory to commercial production provides procurement teams with confidence in consistent supply availability regardless of volume requirements.
- Scalability and Environmental Compliance: The process demonstrates excellent linear scalability from gram-scale laboratory demonstrations to industrial production volumes without requiring reoptimization of reaction parameters. The elimination of toxic heavy metals significantly reduces hazardous waste generation while simplifying waste treatment procedures to meet environmental regulations. The use of standard solvents and reagents facilitates straightforward regulatory documentation for global manufacturing sites while supporting sustainability initiatives through reduced environmental impact.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations for implementing this innovative triazole synthesis methodology in pharmaceutical manufacturing operations. These insights are derived directly from the patent's technical specifications and experimental validation data.
Q: How does this method eliminate heavy metal catalysts while maintaining high yield?
A: The iodine/DMSO system facilitates Kornblum oxidation and subsequent cyclization without transition metals. Iodine acts as both oxidant and catalyst in a synergistic cycle with phosphate buffer and pyridine, enabling metal-free synthesis while achieving comparable yields to traditional methods.
Q: What scalability advantages does this iodine-catalyzed process offer?
A: The reaction operates under ambient conditions without specialized equipment requirements. Patent data demonstrates successful gram-scale production with consistent yields across diverse substrates, indicating straightforward linear scale-up potential to industrial volumes without reoptimization.
Q: How does eliminating anhydrous/oxygen-free conditions improve manufacturing reliability?
A: The robust reaction system tolerates standard atmospheric conditions, removing complex moisture/oxygen control requirements. This simplifies facility setup and reduces batch failure risks associated with stringent environmental controls in conventional triazole syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Intermediate Supplier
Our patented methodology represents a significant advancement in triazole compound manufacturing that delivers both technical excellence and commercial viability for pharmaceutical applications. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our integrated manufacturing platform combines deep expertise in heterocyclic chemistry with robust quality systems that ensure consistent product performance meeting global regulatory requirements for pharmaceutical intermediates.
We invite your technical procurement team to request a Customized Cost-Saving Analysis demonstrating how this innovative synthesis can optimize your specific supply chain requirements. Contact us today to obtain detailed COA data and route feasibility assessments tailored to your production needs.
