Advanced Synthetic Routes for Camptothecin Derivatives and Mappicine Intermediates
The pharmaceutical landscape for anticancer agents continues to evolve, with camptothecin derivatives like irinotecan (CPT-11) remaining critical components in oncology treatment regimens. Patent CN100537538C discloses a comprehensive suite of novel intermediates and procedures designed to streamline the synthesis of these complex molecules. This intellectual property outlines a robust chemical pathway starting from readily available citrazinic acid, navigating through a series of protected intermediates labeled as 2G through 14G, and ultimately yielding high-purity camptothecin cores. For R&D directors and procurement specialists, understanding the nuances of this synthetic route is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The disclosed methods address longstanding challenges in stereochemical control and functional group tolerance, offering a viable alternative to traditional extraction or semi-synthetic routes that often suffer from supply volatility.
The significance of this technology extends beyond mere academic interest; it represents a tangible opportunity for cost reduction in pharmaceutical intermediates manufacturing. By utilizing a convergent synthesis strategy that incorporates efficient protection and deprotection sequences, the process minimizes the formation of difficult-to-remove impurities. The patent explicitly details the preparation of key precursors such as compound 10CPT and its resolved enantiomers 10CPT(R) and 10CPT(S), which serve as pivotal junctions in the assembly of the pentacyclic ring system. For supply chain heads, the ability to source these advanced intermediates from a partner with deep technical expertise ensures continuity of supply for downstream API production, mitigating risks associated with raw material shortages or regulatory compliance issues in natural product sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of camptothecin analogues has been plagued by inefficiencies inherent in isolating the natural product from plant sources or relying on lengthy total syntheses with poor overall yields. Conventional semi-synthetic approaches often struggle with the regioselective functionalization of the pyridine ring, leading to complex mixtures that require extensive and costly purification protocols. Furthermore, achieving high enantiomeric excess in the side chains without resorting to expensive chiral pool starting materials has been a persistent bottleneck. Traditional oxidation methods used to install carboxylic acid or ester functionalities frequently involve hazardous reagents or generate significant waste streams, complicating environmental compliance and increasing the cost of goods sold. These limitations create vulnerabilities in the supply chain, where batch-to-batch variability can jeopardize clinical trial timelines or commercial launch schedules for life-saving medications.
The Novel Approach
In contrast, the methodology presented in CN100537538C introduces a highly controlled synthetic architecture that leverages modern organometallic catalysis and biocatalysis. The route begins with the conversion of citrazinic acid to dichloro-isonicotinic acid derivatives, followed by precise Grignard additions to establish the carbon framework. A standout feature is the use of ketal protection groups (Step 3) to mask reactive ketones during subsequent transformations, preventing unwanted side reactions and enhancing the stability of intermediates like 3CPT. The integration of palladium-catalyzed carbonylation (Step 7GG) allows for the mild and efficient installation of ester groups, a transformation that is notoriously difficult to achieve with high selectivity using classical methods. This novel approach not only improves the chemical yield at each stage but also simplifies the purification workflow, making it an attractive option for commercial scale-up of complex pharmaceutical intermediates.
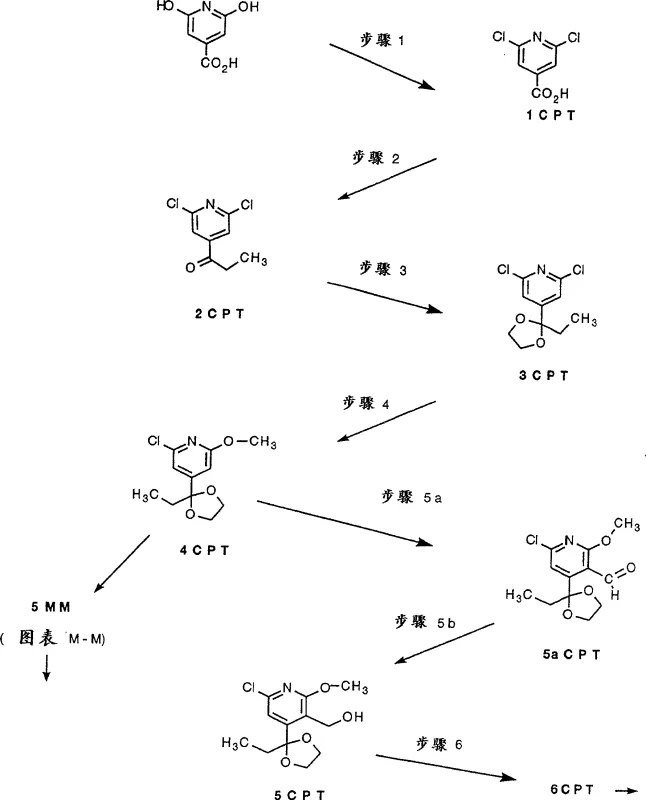
Mechanistically, the success of this synthetic pathway relies on the careful orchestration of electronic and steric effects throughout the molecular scaffold. The palladium-catalyzed carbonylation step, for instance, exploits the reactivity of the 2-chloropyridine moiety, which undergoes oxidative addition with Pd(0) species more readily than simple aryl chlorides. This facilitates the insertion of carbon monoxide and subsequent nucleophilic attack by alcohols to form esters under relatively mild thermal conditions (50°C to 80°C). Furthermore, the patent details a sophisticated enzymatic resolution strategy (Step 10 split) utilizing lipases such as Pseudomonas cepacia. This biocatalytic step selectively acetylates one enantiomer of the racemic diol intermediate 10G, allowing for the physical separation of the desired chiral alcohol from the acetate byproduct. Such kinetic resolution is critical for ensuring the biological activity of the final drug substance, as the therapeutic properties of camptothecins are highly dependent on their stereochemistry. By combining transition metal catalysis with enzymatic specificity, the process achieves a level of purity and control that is difficult to replicate with purely chemical methods.
Impurity control is another cornerstone of this technological advancement. The use of protecting groups like ketals and benzyl ethers serves to isolate reactive centers during harsh reaction conditions, thereby suppressing the formation of polymeric byproducts or degradation species. For example, the removal of the benzyl group via hydrogenation (Step 12, Approach B) is performed under controlled pressure and temperature to prevent over-reduction of the sensitive lactone ring. Additionally, the patent describes multiple pathways (Approach A and Approach B) for converting hydroxy-aldehydes to the final lactone structures, providing process chemists with flexibility to optimize based on available equipment and reagent costs. This modularity in the synthetic design allows manufacturers to adapt the process to meet stringent regulatory specifications for residual solvents and heavy metals, ensuring that the high-purity pharmaceutical intermediates produced are suitable for GMP manufacturing environments.
How to Synthesize Camptothecin Intermediates Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize yield and optical purity. The process initiates with the chlorination of citrazinic acid using phosphoryl chloride, followed by nucleophilic substitution to introduce the methoxy group. Subsequent steps involve the formation of the ethyl side chain via Grignard reaction and protection as a ketal. The core of the synthesis involves the palladium-catalyzed carbonylation to install the ester functionality, followed by ozonolysis or osmylation to generate the diol precursor. Finally, enzymatic resolution separates the enantiomers, which are then oxidized and cyclized to form the camptothecin core. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Initiate the synthesis by converting citrazinic acid to 2,6-dichloro-isonicotinic acid derivatives using phosphoryl chloride and tetramethylammonium chloride at elevated temperatures.
- Perform Grignard addition or lithium alkylide reaction to introduce the ethyl ketone side chain, followed by ketal protection using ethylene glycol and trimethylsilyl chloride.
- Execute palladium-catalyzed carbonylation to introduce ester functionalities, followed by enzymatic kinetic resolution using lipase to separate enantiomers for high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers distinct strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the decoupling of production from agricultural constraints; unlike natural extraction which is subject to crop yields and seasonal variations, this chemical synthesis ensures a consistent and predictable supply of critical intermediates. The use of commodity chemicals such as citrazinic acid, ethylene glycol, and common solvents like toluene and methanol reduces dependency on exotic or single-source raw materials. This diversification of the supply base significantly enhances supply chain reliability, allowing manufacturers to buffer against market fluctuations and geopolitical disruptions that often impact the availability of natural product-derived starting materials.
- Cost Reduction in Manufacturing: The streamlined nature of the synthetic route eliminates several purification bottlenecks associated with traditional methods. By employing protecting group strategies that are easily installed and removed, the process reduces the number of chromatographic separations required, which are often the most expensive and time-consuming operations in fine chemical manufacturing. Furthermore, the catalytic nature of the carbonylation and enzymatic resolution steps means that expensive reagents are used in sub-stoichiometric amounts, driving down the raw material cost per kilogram of finished intermediate. The ability to recycle solvents and recover catalysts further contributes to substantial cost savings without compromising the quality of the final product.
- Enhanced Supply Chain Reliability: The modular design of the synthesis allows for the stocking of stable intermediates at various stages of the production line. Compounds like 6CPT and 10CPT are sufficiently stable to be stored and transported, enabling a just-in-time manufacturing model that reduces inventory holding costs. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, means that production can be scaled across different facilities without extensive re-validation. This flexibility ensures that supply continuity is maintained even if one production site faces operational challenges, providing a resilient backbone for the global distribution of anticancer therapies.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers significant advantages over older technologies. The avoidance of stoichiometric heavy metal oxidants in favor of catalytic osmium or enzymatic systems reduces the generation of hazardous waste streams. The processes described utilize standard unit operations such as crystallization and filtration, which are well-understood and easily scalable from pilot plant to commercial tonnage. This ease of scale-up accelerates the timeline from process development to commercial production, allowing companies to respond rapidly to increased market demand. Additionally, the reduced waste profile aligns with increasingly stringent environmental regulations, minimizing the risk of compliance-related shutdowns and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical procurement teams evaluating potential partners for long-term supply agreements.
Q: How does this patent address chiral purity in camptothecin synthesis?
A: The patent describes a specific enzymatic resolution step (Step 10 split) using lipase catalysts like Pseudomonas cepacia to separate racemic diols into single enantiomers with high optical purity (>99% ee), ensuring the final API meets strict stereochemical requirements.
Q: What are the key advantages of the palladium-catalyzed carbonylation step?
A: This method allows for the efficient introduction of ester groups onto the pyridine ring under mild conditions using carbon monoxide and alcohols, avoiding harsher traditional oxidation methods and improving overall atom economy and yield.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent details scalable unit operations such as crystallization, standard solvent extractions, and catalytic reactions that can be adapted from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial manufacturing without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of oncology drugs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and potency of every batch. Our facility is equipped to handle the complex chemistry required for camptothecin derivatives, including moisture-sensitive reactions and chiral resolutions, guaranteeing a supply chain that is both robust and compliant with international standards.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to accelerate your drug development timeline and secure a sustainable supply of these vital pharmaceutical building blocks.
