Revolutionizing Camptothecin Derivative Production: Novel Intermediates for Commercial Scale-up of Complex Pharmaceutical Intermediates
Revolutionizing Camptothecin Derivative Production: Novel Intermediates for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value anticancer agents, particularly camptothecin derivatives like Irinotecan and Topotecan. Patent CN101659668A introduces a groundbreaking methodology that addresses longstanding challenges in the functionalization of the camptothecin core, specifically targeting the critical C10 hydroxylation and C7 alkylation steps. This technology represents a significant leap forward for any reliable camptothecin intermediate supplier aiming to optimize their production pipelines. By shifting away from harsh photochemical conditions towards controlled catalytic oxidation and strategic silyl protection, the disclosed process offers a pathway to higher purity intermediates with improved operational safety. The innovation lies not just in the final product but in the versatile intermediate compounds that allow for modular synthesis of various analogues, providing a flexible platform for drug development and commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a hydroxyl group at the C10 position of the camptothecin A-ring has been a bottleneck in the synthesis of potent topoisomerase I inhibitors. Traditional methods often relied on the formation of camptothecin-1-oxide followed by exposure to ultraviolet (UV) light to induce rearrangement and hydroxylation. This photochemical approach presents severe limitations for industrial application, including the requirement for specialized quartz reactors, difficulties in light penetration at larger scales, and the generation of complex impurity profiles due to non-selective radical pathways. Furthermore, the handling of N-oxide intermediates can be hazardous, and the overall yield of the UV-mediated step is frequently inconsistent, leading to batch-to-batch variability that is unacceptable for GMP manufacturing of active pharmaceutical ingredients. These factors collectively drive up the cost of goods and complicate the regulatory approval process for generic versions of these life-saving drugs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a two-step sequence involving catalytic hydrogenation followed by chemical oxidation to achieve the same structural transformation with superior control. As illustrated in the reaction scheme below, camptothecin is first hydrogenated to a tetrahydro-derivative, which is then subjected to oxidation using reagents like palladium diacetate or lead acetate in the presence of a Bronsted acid. 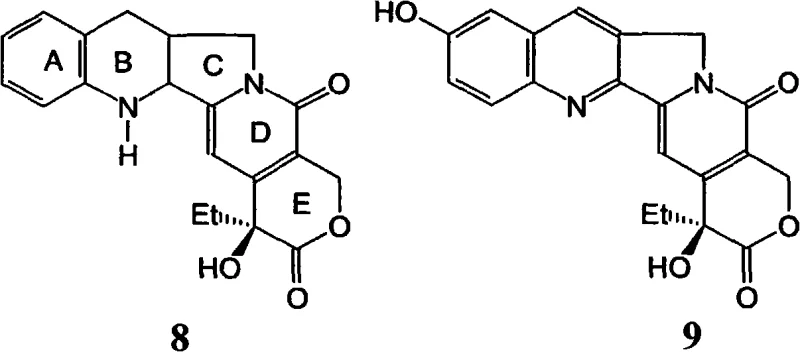 This chemical oxidation strategy completely eliminates the need for UV irradiation, allowing the reaction to proceed in standard stainless steel reactors under mild thermal conditions. The re-aromatization of the B-ring occurs concurrently with the introduction of the hydroxyl group, streamlining the synthesis and significantly reducing the number of unit operations required. This method not only enhances the safety profile of the manufacturing process but also ensures a more consistent and high-quality output of 10-hydroxycamptothecin, which is the pivotal precursor for both Irinotecan and Topotecan.
This chemical oxidation strategy completely eliminates the need for UV irradiation, allowing the reaction to proceed in standard stainless steel reactors under mild thermal conditions. The re-aromatization of the B-ring occurs concurrently with the introduction of the hydroxyl group, streamlining the synthesis and significantly reducing the number of unit operations required. This method not only enhances the safety profile of the manufacturing process but also ensures a more consistent and high-quality output of 10-hydroxycamptothecin, which is the pivotal precursor for both Irinotecan and Topotecan.
Mechanistic Insights into Selective N-Silylation and Grignard Alkylation
A critical component of this patented technology is the strategic use of N-silyl protection to direct subsequent alkylation reactions with high regioselectivity. The process involves treating the 10-hydroxycamptothecin intermediate with a silylating agent, preferably t-butyldimethylsilyl triflate (TBDMSOTf), to form an N-silyl camptothecin derivative. This silyl group serves a dual purpose: it protects the nitrogen atom from unwanted nucleophilic attacks and electronically activates the adjacent C7 position for electrophilic substitution. When this N-silyl intermediate is exposed to a Grignard reagent, such as ethyl magnesium bromide, the alkylation occurs exclusively at the C7 position of the quinoline ring system. The mechanism relies on the coordination of the magnesium species with the electron-deficient centers of the heterocyclic system, facilitated by the steric and electronic environment created by the bulky silyl group at N1. This level of control is essential for preventing the formation of N-alkylated byproducts, which are difficult to separate and can compromise the biological activity of the final drug substance.
Following the alkylation step, the process incorporates an elegant oxidative workup to remove the silyl protecting group and restore the aromaticity of the system while retaining the newly installed ethyl group. The reaction mixture is treated with an oxidant, such as oxygen or specific metal salts, which cleaves the silicon-nitrogen bond and simultaneously ensures the stability of the lactone ring. 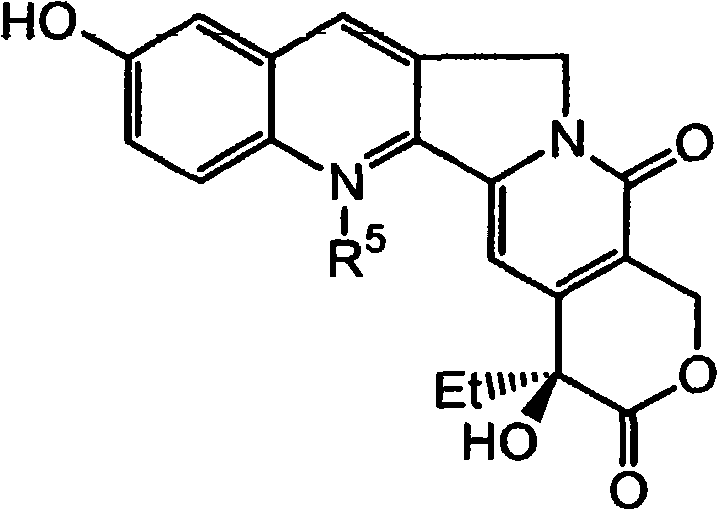 This sequence demonstrates a sophisticated understanding of heterocyclic chemistry, where the interplay between protection, activation, and deprotection is managed within a cohesive synthetic flow. For R&D teams, this mechanistic clarity offers a robust framework for troubleshooting and optimizing reaction parameters, such as temperature and stoichiometry, to maximize yield. The ability to perform these transformations without affecting the sensitive C20 stereocenter is particularly noteworthy, as the (S)-configuration at C20 is crucial for the antitumor activity of camptothecin derivatives. This precision in stereochemical retention underscores the suitability of the method for producing high-purity pharmaceutical intermediates that meet stringent regulatory specifications.
This sequence demonstrates a sophisticated understanding of heterocyclic chemistry, where the interplay between protection, activation, and deprotection is managed within a cohesive synthetic flow. For R&D teams, this mechanistic clarity offers a robust framework for troubleshooting and optimizing reaction parameters, such as temperature and stoichiometry, to maximize yield. The ability to perform these transformations without affecting the sensitive C20 stereocenter is particularly noteworthy, as the (S)-configuration at C20 is crucial for the antitumor activity of camptothecin derivatives. This precision in stereochemical retention underscores the suitability of the method for producing high-purity pharmaceutical intermediates that meet stringent regulatory specifications.
How to Synthesize 7-Ethyl-10-hydroxycamptothecine Efficiently
The synthesis of 7-ethyl-10-hydroxycamptothecine (SN-38), the active metabolite of Irinotecan, is achieved through a streamlined sequence that leverages the novel intermediates described above. The process begins with the preparation of the N-silyl protected 10-hydroxycamptothecin, which serves as a stable and reactive platform for carbon-carbon bond formation. By utilizing ether solvents like tetrahydrofuran (THF) and maintaining low temperatures during the addition of the Grignard reagent, the reaction minimizes side reactions and ensures high conversion rates. The subsequent oxidative removal of the silyl group is performed under controlled conditions to prevent degradation of the sensitive lactone moiety. This integrated approach reduces the total number of isolation steps, thereby minimizing material loss and solvent consumption. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide provided below.
- Perform catalytic hydrogenation of camptothecin using palladium hydroxide in glacial acetic acid to obtain the tetrahydro-camptothecin intermediate.
- Oxidize the tetrahydro-intermediate using palladium diacetate or lead acetate in the presence of a Bronsted acid to introduce the C10 hydroxyl group and re-aromatize the B-ring.
- Protect the N1 position with a silylating agent like t-butyldimethylsilyl triflate, followed by Grignard alkylation with ethyl magnesium bromide to install the C7 ethyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of photochemical steps removes a significant capital expenditure barrier, as manufacturers no longer need to invest in specialized UV reactors or manage the associated maintenance and safety protocols. This simplification of the equipment footprint directly translates to lower overhead costs and facilitates easier technology transfer between different manufacturing sites globally. Furthermore, the reliance on commodity chemicals such as palladium catalysts, acetic acid, and Grignard reagents ensures a stable and predictable supply of raw materials, mitigating the risk of production delays caused by the scarcity of exotic reagents. The robustness of the chemical oxidation step also implies a wider operating window, reducing the likelihood of batch failures and ensuring a consistent supply of critical intermediates to downstream formulation partners.
- Cost Reduction in Manufacturing: The transition from UV-mediated oxidation to chemical oxidation drastically simplifies the process infrastructure, removing the need for expensive quartz ware and high-energy light sources. By consolidating the re-aromatization and hydroxylation into a single chemical step, the overall processing time is significantly reduced, leading to lower utility costs and increased reactor throughput. Additionally, the high regioselectivity of the N-silyl directed alkylation minimizes the formation of hard-to-remove impurities, which reduces the burden on purification processes such as chromatography and crystallization. This efficiency gain results in substantial cost savings per kilogram of produced API intermediate, enhancing the overall competitiveness of the supply chain.
- Enhanced Supply Chain Reliability: The use of widely available reagents like ethyl magnesium bromide and palladium diacetate ensures that the supply chain is not vulnerable to the bottlenecks often associated with custom-synthesized specialty chemicals. The process conditions, involving standard temperatures and pressures, are compatible with existing multipurpose manufacturing facilities, allowing for rapid scale-up without the need for extensive retrofitting. This flexibility enables suppliers to respond quickly to fluctuations in market demand for camptothecin-based therapeutics, ensuring continuity of supply for patients relying on these critical cancer treatments. The stability of the N-silyl intermediates also allows for potential storage and transport, decoupling the production of key building blocks from the final assembly steps.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are manageable in large-scale waste streams. The avoidance of UV light reduces energy consumption, aligning with green chemistry principles and corporate sustainability goals. Moreover, the high selectivity of the reactions leads to cleaner reaction profiles, which simplifies effluent treatment and reduces the environmental footprint of the manufacturing process. The ability to run these reactions in standard glass-lined or stainless steel reactors ensures that the process can be scaled from pilot plant to commercial production with minimal technical risk, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel camptothecin synthesis technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers highlight the balance between chemical innovation and practical industrial application.
Q: What are the advantages of the N-silyl protection strategy in camptothecin synthesis?
A: The N-silyl protection strategy allows for highly regioselective alkylation at the C7 position while preventing unwanted side reactions at the nitrogen atom, significantly improving the purity profile of the final intermediate.
Q: How does the new oxidation method differ from traditional UV-based methods?
A: Unlike traditional methods that rely on harsh UV irradiation of N-oxides, this patent utilizes chemical oxidants like palladium diacetate under mild acidic conditions, eliminating the need for specialized photochemical equipment and reducing safety risks.
Q: Is this process suitable for large-scale manufacturing of Irinotecan precursors?
A: Yes, the process utilizes standard reagents such as Grignard reagents and common metal catalysts, avoiding exotic conditions, which makes it highly amenable to commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the methodologies described in CN101659668A and possesses the technical capability to implement these advanced processes immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. Our state-of-the-art facilities are equipped to handle the specific catalytic hydrogenation and oxidation steps required for these camptothecin derivatives, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates for your drug development programs.
We invite you to collaborate with us to leverage these technological advancements for your next-generation oncology portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our camptothecin intermediate library and to discuss route feasibility assessments for your proprietary analogues. Together, we can accelerate the delivery of life-saving medications to patients worldwide while optimizing the economic efficiency of your supply chain.
