Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
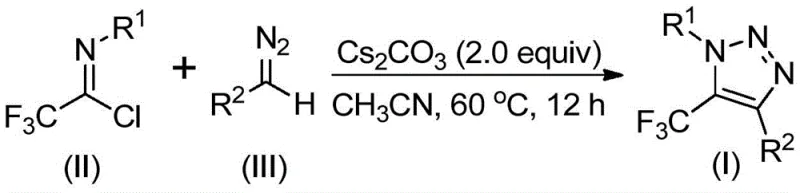
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding safety profiles and synthetic efficiency. A pivotal advancement in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a significant departure from legacy methodologies by eliminating the reliance on hazardous organic azides and transition metal catalysts. For R&D directors and procurement specialists alike, this innovation offers a pathway to high-purity pharmaceutical intermediates with a drastically improved safety footprint. The core of this invention lies in a base-promoted cyclization strategy that couples trifluoroethylimidoyl chloride with diazo compounds, yielding complex triazole scaffolds under mild thermal conditions. This report analyzes the technical merits and commercial implications of adopting this metal-free route for the production of critical fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,3-triazole ring, a privileged scaffold in medicinal chemistry, has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions. While effective, these traditional pathways present substantial logistical and safety hurdles for large-scale manufacturing. The primary concern is the utilization of organic azides, which are inherently unstable, toxic, and possess shock-sensitive properties that pose severe explosion risks during storage and handling. Furthermore, the requirement for copper catalysts introduces the challenge of residual metal contamination, necessitating expensive downstream purification processes to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). Alternative organocatalytic methods involving trifluoromethyl ketones also suffer from the azide dependency, limiting their viability for green chemistry initiatives. These factors collectively inflate the cost of goods sold (COGS) and extend lead times due to rigorous safety protocols and waste management requirements.
The Novel Approach
In stark contrast, the methodology described in patent CN113121462B circumvents these bottlenecks by employing a completely different disconnection strategy. By utilizing readily available trifluoroethylimidoyl chloride and diazo compounds as starting materials, the process achieves the desired 5-trifluoromethyl substitution without ever generating free azide species. The reaction is driven by cesium carbonate, a mild and inexpensive inorganic base, rather than precious or toxic transition metals. This shift not only enhances the intrinsic safety of the operation by removing explosive hazards but also simplifies the workup procedure. The absence of heavy metals means that the final product requires less aggressive purification to remove metal traces, directly translating to higher overall yields and reduced processing time. This approach aligns perfectly with the industry's move towards sustainable and economically viable synthetic routes for complex heterocycles.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction likely proceeds through a sophisticated cascade initiated by the deprotonation or activation of the diazo compound by the cesium carbonate base. This activation facilitates a nucleophilic attack on the electrophilic carbon of the trifluoroethylimidoyl chloride. Unlike traditional cycloadditions that form the triazole ring in a single concerted step, this pathway involves an intermolecular nucleophilic addition-elimination sequence that first establishes the critical carbon-carbon bond. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is energetically favorable in this system due to the electronic influence of the trifluoromethyl group and the stabilization provided by the nitrogen lone pairs. The result is the formation of the aromatic 1,2,3-triazole core with the trifluoromethyl group precisely positioned at the 5-position, a regioselectivity that is often difficult to control in other synthetic manifolds.
From an impurity control perspective, this mechanism offers distinct advantages. The stepwise nature of the bond formation allows for better monitoring of reaction progress and intermediate stability. Since the reaction does not involve radical intermediates typical of some metal-catalyzed processes, the formation of homocoupling byproducts or polymerization side-reactions is minimized. The use of acetonitrile as the preferred solvent further supports this mechanism by providing a polar aprotic environment that stabilizes the ionic intermediates without participating in side reactions. This high degree of chemoselectivity ensures that sensitive functional groups on the R1 and R2 substituents—such as esters, phosphonates, or halogens—remain intact, thereby expanding the scope of accessible derivatives for structure-activity relationship (SAR) studies.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this patented method makes it highly attractive for process chemists looking to implement robust manufacturing protocols. The procedure typically involves charging a reactor with cesium carbonate, the specific trifluoroethylimidoyl chloride derivative, and the corresponding diazo compound in anhydrous acetonitrile. The mixture is then heated to a moderate temperature range of 50°C to 70°C, with 60°C being the optimal setpoint for balancing reaction rate and energy consumption. Reaction times generally span between 8 to 16 hours, allowing for complete conversion even with sterically hindered substrates. Upon completion, the workup is straightforward, involving simple filtration to remove inorganic salts followed by standard silica gel chromatography. For detailed standard operating procedures and specific stoichiometric ratios optimized for different substrate classes, please refer to the technical guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in anhydrous acetonitrile under inert atmosphere.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The most significant advantage lies in the drastic simplification of the supply chain risk profile. By eliminating the need for organic azides, facilities can avoid the high insurance premiums and specialized storage infrastructure required for explosive materials. This directly contributes to substantial cost savings in facility operations and compliance. Furthermore, the starting materials—trifluoroethylimidoyl chlorides and diazo compounds—are commercially available or easily synthesized from commodity chemicals, ensuring a stable and continuous supply stream that is less susceptible to geopolitical disruptions affecting rare earth metals or specialized catalysts.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling. The replacement of expensive transition metal catalysts (like copper or ruthenium) with inexpensive cesium carbonate significantly lowers the raw material cost per kilogram. Moreover, the removal of heavy metals from the process eliminates the need for specialized scavenger resins or complex extraction protocols designed to reduce metal content to ppm levels. This streamlining of the downstream processing reduces solvent consumption and labor hours, leading to a leaner manufacturing cost structure. The high atom economy of the reaction also minimizes waste generation, further reducing disposal costs associated with hazardous chemical waste.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain. This method utilizes robust reagents that have long shelf lives and do not require cryogenic storage or inert atmosphere handling to the same extent as sensitive organometallic reagents. The tolerance of the reaction to various functional groups means that a single standardized protocol can be applied to a wide library of intermediates, reducing the need for custom process development for each new SKU. This flexibility allows suppliers to respond more rapidly to changing demand forecasts and reduces the lead time for delivering high-purity pharmaceutical intermediates to clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen thermal and safety challenges, but this base-promoted cyclization is inherently scalable. The reaction exotherm is manageable, and the use of acetonitrile, a common industrial solvent, simplifies solvent recovery and recycling systems. From an environmental standpoint, the absence of toxic heavy metals in the effluent simplifies wastewater treatment and helps manufacturers meet increasingly strict environmental, social, and governance (ESG) targets. The ability to scale from gram to multi-kilogram batches without re-optimizing the core chemistry provides a clear path for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,3-triazole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, eliminating the handling risks associated with high-energy azide intermediates.
Q: What represents the primary cost driver in this manufacturing process?
A: The process utilizes cesium carbonate as a promoter, which is relatively inexpensive compared to transition metal catalysts like copper or rhodium. Additionally, the absence of heavy metals removes the need for costly purification steps to meet residual metal specifications.
Q: Can this reaction be scaled for industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with mild reaction conditions (60°C) and standard solvents like acetonitrile, making it highly suitable for kilogram-to-ton scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with a technically proficient CDMO is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and trace metal testing, guaranteeing that every batch meets the highest international standards. We understand the critical nature of supply continuity and have established robust sourcing channels for key precursors like diazo compounds and imidoyl chlorides.
We invite you to leverage our technical expertise to optimize your supply chain for these valuable intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a reliable, cost-effective, and safe supply of 5-trifluoromethyl-1,2,3-triazoles for your next generation of therapeutics.
