Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
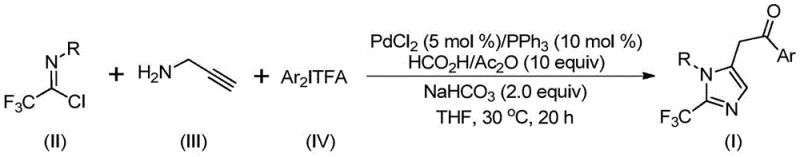
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology represents a paradigm shift from traditional, often hazardous trifluoromethylation protocols to a sophisticated palladium-catalyzed carbonylation strategy. By leveraging a multicomponent reaction system involving trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, this method achieves high conversion rates under remarkably mild conditions. For R&D directors and procurement specialists alike, the implications are profound: a pathway to high-purity heterocyclic building blocks that are critical for modern drug discovery, particularly for enhancing metabolic stability and lipophilicity in lead compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into nitrogen-containing heterocycles has been a formidable challenge in organic synthesis. Conventional methods often rely on the use of specialized trifluoromethyl synthons such as trifluorodiazoethane or require harsh reaction conditions that limit functional group tolerance. These traditional approaches frequently suffer from poor atom economy, the generation of toxic byproducts, and the necessity for expensive, moisture-sensitive reagents that complicate supply chain logistics. Furthermore, many existing protocols demand high temperatures or strong bases, which can lead to decomposition of sensitive substrates and the formation of difficult-to-remove impurities. For a reliable pharmaceutical intermediate supplier, these factors translate into higher production costs, longer lead times, and increased safety risks during scale-up, ultimately hindering the rapid development of new therapeutic agents.
The Novel Approach
In stark contrast, the methodology described in patent CN111423381B offers a streamlined and robust alternative. This novel approach utilizes a transition metal palladium catalyst to facilitate a cascade reaction that efficiently constructs the imidazole ring while simultaneously installing the trifluoromethyl motif. The reaction proceeds at a mild temperature of 30°C, significantly reducing energy consumption and thermal stress on the reactants. By employing cheap and easily obtainable starting materials like trifluoroethylimidoyl chloride and propargylamine, the process drastically simplifies the raw material sourcing strategy. The use of diaryl iodonium salts as coupling partners further enhances the versatility of the method, allowing for the introduction of diverse aryl groups. This flexibility is crucial for medicinal chemists seeking to optimize structure-activity relationships (SAR) without being constrained by synthetic limitations.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for ensuring reproducibility and quality control in a manufacturing setting. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the base, yielding a trifluoroacetamidine intermediate. This species then undergoes isomerization, setting the stage for the palladium cycle. The palladium catalyst, typically generated in situ from PdCl2 and PPh3, coordinates with the alkyne moiety of the propargylamine derivative. Subsequent palladation leads to an alkenyl palladium intermediate, which isomerizes to a more stable alkyl palladium species. A critical step follows: the carbonylation reaction. Here, carbon monoxide is released in situ from the decomposition of formic acid and acetic anhydride, inserting into the palladium-carbon bond to form an acyl palladium intermediate. This elegant use of a CO surrogate avoids the need for handling hazardous carbon monoxide gas directly, a major safety advantage for industrial operations.
The final stages of the catalytic cycle involve the oxidative addition of the diaryl iodonium salt to the acyl palladium species, generating a high-valent tetravalent palladium intermediate. Reductive elimination from this complex releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst. From an impurity control perspective, the mild reaction temperature of 30°C is instrumental. It suppresses side reactions such as polymerization of the alkyne or decomposition of the iodonium salt, which are common at elevated temperatures. The high selectivity of the palladium center ensures that the desired cyclization pathway is favored, resulting in a clean crude reaction profile that simplifies downstream purification. This mechanistic clarity allows process chemists to fine-tune parameters such as ligand loading and base equivalents to maximize yield and minimize waste.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant requires careful attention to reagent quality and reaction monitoring. The protocol outlined in the patent provides a clear roadmap for achieving consistent results. The process begins with the preparation of the catalytic system in an aprotic solvent, preferably tetrahydrofuran (THF), which effectively dissolves all organic components. The sequential addition of reagents ensures proper mixing and initiation of the catalytic cycle. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-efficiency route.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The shift towards this palladium-catalyzed method addresses several critical pain points in the sourcing of complex heterocyclic intermediates. By utilizing commodity chemicals as starting materials, the dependency on niche, high-cost fluorinating agents is eliminated. This diversification of the supply base enhances resilience against market volatility and raw material shortages. Furthermore, the operational simplicity of the process—requiring only standard stirring and mild heating—means that it can be executed in existing multipurpose reactors without the need for specialized high-pressure or cryogenic equipment. This compatibility with standard infrastructure accelerates technology transfer and reduces capital expenditure requirements for scaling up production.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the utilization of inexpensive and widely available feedstocks. Trifluoroethylimidoyl chloride and propargylamine are commodity chemicals with established global supply chains, ensuring stable pricing. Additionally, the catalytic nature of the palladium system means that only low loadings of the precious metal are required, minimizing the cost associated with noble metals. The mild reaction conditions also translate to significant energy savings, as there is no need for prolonged heating or cooling cycles. The simplified workup procedure, involving basic filtration and chromatography, reduces solvent consumption and labor hours compared to multi-step traditional syntheses. These factors collectively contribute to a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical manufacturers, and this synthesis route bolsters reliability through its robust substrate scope. The method tolerates a wide range of functional groups on both the imidoyl chloride and the iodonium salt components, including electron-donating and electron-withdrawing substituents. This flexibility allows manufacturers to source alternative derivatives if a specific starting material becomes unavailable, without needing to revalidate an entirely new synthetic route. Moreover, the reaction's high efficiency and short reaction time of approximately 20 hours enable faster batch turnover. This increased throughput capacity allows suppliers to respond more agilely to fluctuating demand from downstream API manufacturers, reducing lead times and preventing stockouts.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety, but this method is inherently designed for scalability. The exothermicity of the reaction is manageable at 30°C, reducing the risk of thermal runaway incidents during large-scale production. From an environmental standpoint, the use of formic acid as a carbon monoxide source is a greener alternative to using pressurized CO gas cylinders, significantly lowering the safety footprint of the facility. The high atom economy of the multicomponent reaction minimizes waste generation, aligning with increasingly stringent environmental regulations. The ability to produce high-purity products with minimal byproducts also reduces the burden on waste treatment facilities, supporting sustainable manufacturing practices and long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride and operates under mild conditions (30°C), avoiding the harsh reagents often required in direct trifluoromethylation strategies.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process demonstrates excellent substrate compatibility and high reaction efficiency with yields up to 97%, making it highly scalable for commercial manufacturing of complex pharmaceutical intermediates.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride act as a carbon monoxide surrogate, generating CO in situ which is essential for the carbonylation step that constructs the imidazole core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has thoroughly analyzed the potential of the palladium-catalyzed route described in CN111423381B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole intermediate meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for these valuable heterocyclic building blocks. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
