Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks efficient pathways to access nitrogen-containing heterocycles due to their prevalence in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These scaffolds are ubiquitous in medicinal chemistry, serving as core structures for agents with anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic incorporation of a trifluoromethyl group into these heterocycles is not merely a structural variation; it fundamentally alters the physicochemical profile of the molecule, enhancing metabolic stability and membrane permeability. As a leading entity in fine chemical manufacturing, we recognize that mastering such synthetic methodologies is crucial for maintaining a competitive edge in the global supply chain of active pharmaceutical ingredients.
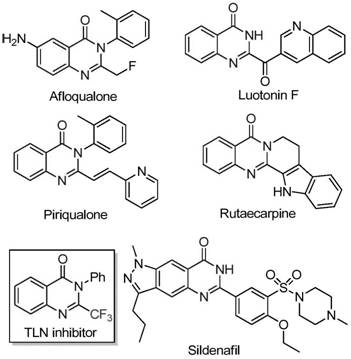
The significance of this technology extends beyond academic interest; it addresses critical bottlenecks in process chemistry. Traditional routes often rely on expensive reagents or harsh conditions that limit throughput and increase waste. By leveraging the insights from this patent, manufacturers can transition towards more sustainable and economically viable production models. The ability to synthesize these complex fused-ring systems with high regioselectivity and yield is a key differentiator for any reliable pharmaceutical intermediate supplier aiming to support the development of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has been fraught with challenges that impede large-scale manufacturing. Conventional literature methods typically employ cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While chemically feasible, these approaches suffer from severe limitations including narrow substrate scope and the requirement for stringent reaction conditions. Furthermore, the starting materials for these traditional routes are often costly and sensitive to moisture, necessitating rigorous anhydrous environments that drive up operational expenditures. The low yields frequently associated with these older protocols result in significant material loss, complicating the economic feasibility of producing high-purity intermediates for clinical trials or commercial launch.
The Novel Approach
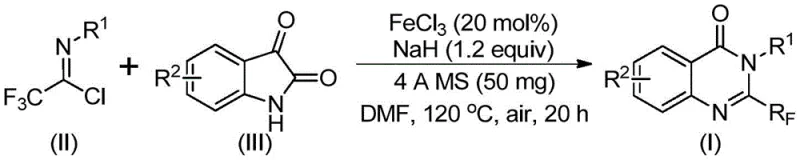
In stark contrast, the methodology outlined in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials. This shift in synthon strategy represents a paradigm change in how these valuable heterocycles are constructed. By employing a cheap metal iron catalyst, specifically ferric chloride, the reaction achieves high efficiency under relatively mild conditions compared to precious metal catalysis. The process tolerates a wide range of functional groups, allowing for the direct synthesis of diverse derivatives without extensive protecting group manipulation. This robustness translates directly into process simplification, where fewer unit operations are required to achieve the target purity, thereby streamlining the overall manufacturing workflow for complex pharmaceutical intermediates.
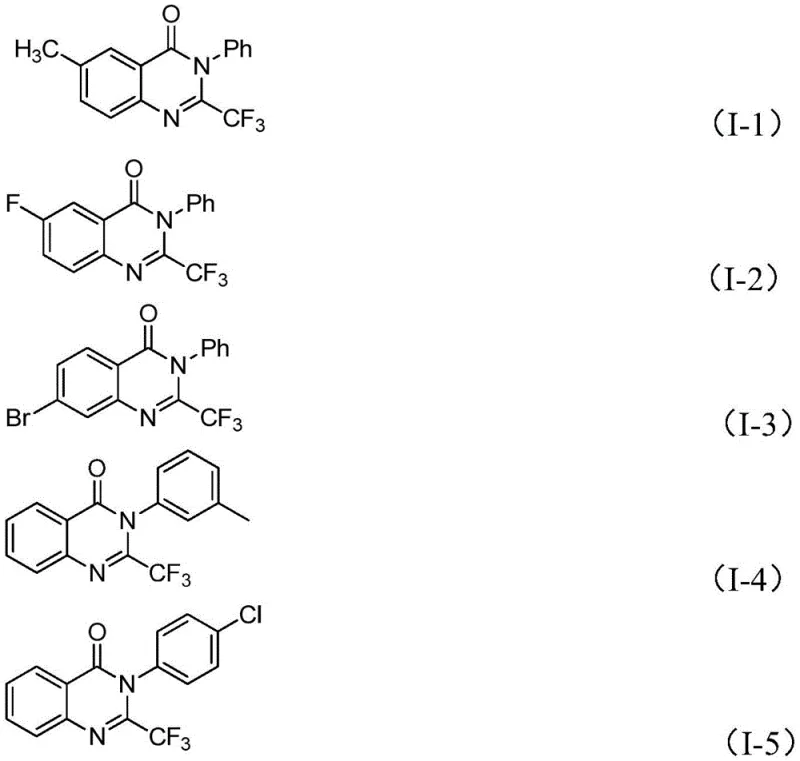
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the iron-catalyzed decarbonylation and cyclization mechanism. The reaction initiates with the formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate, likely promoted by the base sodium hydride. This intermediate then undergoes an iron-catalyzed transformation where the carbonyl group of the isatin moiety is extruded, facilitating the ring closure to form the quinazolinone core. The use of ferric chloride is particularly advantageous as it acts as a Lewis acid to activate the substrates while remaining cost-effective compared to noble metals. The presence of 4A molecular sieves in the reaction mixture plays a critical role in scavenging trace water, which ensures the stability of the reactive imidoyl chloride species and prevents hydrolysis side reactions that could degrade yield.
From an impurity control perspective, this mechanism offers distinct advantages. The high functional group tolerance means that substituents such as halogens, alkyl groups, and methoxy groups on the aromatic rings remain intact throughout the vigorous heating phases. The reaction conditions, involving an initial phase at 40°C followed by heating to 120°C in DMF, are optimized to drive the equilibrium towards the cyclized product while minimizing the formation of polymeric byproducts. Understanding this mechanistic pathway allows process chemists to fine-tune parameters such as catalyst loading and reaction time to maximize the purity profile, ensuring that the final API intermediate meets the stringent specifications required by regulatory bodies.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure reproducibility and safety. The protocol involves a sequential addition of reagents where the timing and temperature ramps are critical for success. The use of anhydrous DMF as the solvent is preferred due to its ability to dissolve both the organic substrates and the inorganic catalyst effectively. Operators must adhere to strict safety guidelines when handling sodium hydride, as it is a potent base that reacts violently with water. The following guide outlines the standardized procedure derived from the patent data to assist R&D teams in replicating these results.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), and 4A molecular sieves in anhydrous DMF under air atmosphere.
- Add trifluoroethylimidoyl chloride and isatin derivative to the reaction mixture and stir at 40°C for 10 hours.
- Heat the reaction to 120°C and continue stirring for 20 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond simple chemistry. The shift from expensive precious metal catalysts to abundant iron salts drastically reduces the raw material cost base. Additionally, the elimination of complex purification steps associated with removing heavy metal residues simplifies the downstream processing, leading to faster batch turnover times. This efficiency is crucial for maintaining lean inventory levels and responding rapidly to market demands for key pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The replacement of costly reagents like trifluoroacetic anhydride with trifluoroethylimidoyl chloride, combined with the use of inexpensive ferric chloride, significantly lowers the bill of materials. Furthermore, the high atom economy of the cyclization reaction minimizes waste generation, reducing disposal costs and environmental levies associated with chemical manufacturing.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted isatins and aromatic amines, are commodity chemicals available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions caused by single-source dependencies, ensuring continuous production capability even during market volatility.
- Scalability and Environmental Compliance: The reaction operates under air atmosphere rather than requiring inert gas protection for the entire duration, which simplifies reactor setup and reduces utility consumption. The robustness of the method allows for seamless scale-up from gram to kilogram quantities, facilitating the transition from process development to commercial production without the need for extensive re-optimization.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries regarding the synthesis and application of these compounds. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost advantages over precious metal catalysts like palladium or rhodium. It is inexpensive, readily available, and demonstrates high tolerance for various functional groups, reducing the need for complex protection-deprotection strategies.
Q: How does this method improve the metabolic stability of the final drug molecule?
A: The introduction of the trifluoromethyl group at the 2-position significantly enhances the electronegativity and lipophilicity of the quinazolinone scaffold. This modification improves bioavailability and metabolic stability, which are critical parameters for oral drug candidates.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides possibilities for industrial scale application. The use of common solvents like DMF and robust reaction conditions supports scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN111675662B to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your drug development programs and accelerate your time to market.
