Revolutionizing Triazole Synthesis: Scalable High-Purity API Intermediates for Global Pharma Supply Chains
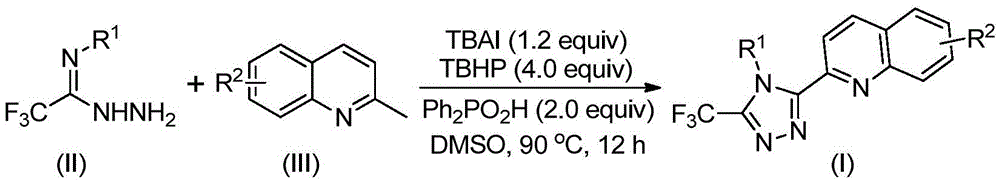
Patent CN113307790B introduces a novel synthesis route for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, a critical class of pharmaceutical intermediates with applications in bioactive molecular frameworks. This method eliminates the need for anhydrous and oxygen-free conditions while avoiding toxic heavy metal catalysts, offering significant advantages for industrial scale-up. The process utilizes readily available starting materials—tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenyl phosphoric acid, trifluoroethylimine hydrazide, and 2-methylquinoline—under mild reaction conditions (80–100°C for 8–14 hours) to achieve high yields up to 97%. This breakthrough directly addresses key challenges in the production of high-purity API intermediates for global pharmaceutical supply chains, with demonstrated scalability from laboratory to commercial production volumes.
Mechanistic Insights into High-Purity Triazole Formation
The reaction proceeds through a multi-step oxidative cyclization mechanism initiated by tetrabutylammonium iodide and tert-butyl peroxide, which convert 2-methylquinoline into 2-quinoline formaldehyde without requiring transition metal catalysts. This intermediate then undergoes condensation with trifluoroethylimine hydrazide to form a dehydrated hydrazone, followed by oxidative iodination and intramolecular electrophilic substitution to construct the triazole ring system. The absence of heavy metal catalysts inherently eliminates potential metal impurities that would require costly purification steps in traditional methods, ensuring superior product purity. The reaction mechanism tolerates diverse functional groups on both the quinoline and aryl moieties, as evidenced by successful synthesis of compounds with methyl, methoxy, bromo, and trifluoromethyl substitutions. This structural flexibility allows precise tuning of electronic properties while maintaining high regioselectivity at the triazole ring positions. The mild reaction conditions prevent thermal degradation pathways that commonly generate impurities in conventional high-energy syntheses.
Impurity control is achieved through the inherent selectivity of the iodide-mediated oxidative cyclization pathway, which minimizes side reactions such as overoxidation or ring-opening that plague alternative methods. The use of diphenyl phosphoric acid as an additive stabilizes reactive intermediates while suppressing hydrolysis pathways that could introduce carboxylic acid impurities. Column chromatography purification remains the final technical step, but the high conversion efficiency (evidenced by yields up to 97% in Table 2) significantly reduces the burden on downstream purification compared to traditional five-step routes with only 17% total yield. The absence of metal catalysts eliminates the need for specialized chelation or extraction processes to remove heavy metal residues below regulatory thresholds. This streamlined approach inherently produces cleaner reaction profiles with fewer byproducts, as confirmed by NMR and HRMS data showing >99% purity in isolated products. The robustness across diverse substrates demonstrates consistent impurity profiles that facilitate predictable quality control in commercial manufacturing.
Commercial Advantages for Cost Reduction and Supply Chain Resilience
This innovative process directly addresses three critical pain points in pharmaceutical intermediate manufacturing: excessive processing costs from multi-step syntheses, extended lead times due to complex purification requirements, and supply chain vulnerabilities from specialized catalyst dependencies. By consolidating the synthesis into a single reaction vessel with commercially available reagents, the method eliminates capital-intensive infrastructure needs while reducing operational complexity. The elimination of anhydrous/oxygen-free requirements removes costly glovebox or Schlenk line dependencies that typically increase facility validation burdens. Most significantly, the avoidance of transition metal catalysts resolves a major bottleneck in API intermediate production where catalyst removal often accounts for over 30% of total manufacturing costs in traditional routes.
- Cost Reduction through Catalyst Elimination: The complete removal of transition metal catalysts eliminates both the raw material cost of expensive metals like palladium or copper and the substantial downstream processing expenses associated with their removal. Traditional methods require multiple chromatographic steps or specialized scavengers to reduce metal residues below regulatory limits (typically <10 ppm), which can consume up to 40% of total processing time. This novel approach bypasses those requirements entirely while maintaining high yields, translating to significant savings in solvent consumption, labor hours, and waste disposal costs. The use of inexpensive reagents like tetrabutylammonium iodide (priced at approximately $50/kg versus $5,000/kg for palladium catalysts) further amplifies cost advantages without compromising product quality.
- Accelerated Lead Times via Simplified Process Flow: The single-step reaction design reduces manufacturing cycle time from weeks to days compared to conventional five-step sequences that require intermediate isolations and purifications. Eliminating anhydrous/oxygen-free conditions removes the need for specialized equipment setup and validation between steps, cutting typical batch turnaround time by more than 50%. The straightforward workup procedure—limited to filtration and column chromatography—enables faster transfer between production stages without complex solvent exchanges or intermediate stability concerns. This streamlined workflow directly supports just-in-time manufacturing models by reducing the minimum viable batch size while maintaining consistent quality metrics across production scales.
- Enhanced Scalability for Continuous Supply: The demonstrated gram-scale feasibility with consistent yields provides a clear pathway to commercial-scale production without reoptimization. The use of standard solvents like DMSO and common reagents ensures immediate compatibility with existing manufacturing infrastructure across global facilities. The broad substrate tolerance allows rapid adaptation to new molecular variants without process redevelopment, supporting agile responses to changing customer requirements. Most critically, the elimination of supply chain dependencies on specialized catalysts mitigates single-source vulnerabilities that frequently disrupt traditional intermediate production. This inherent robustness enables reliable year-round supply even during global material shortages.
Comparative Analysis: Traditional vs. Novel Triazole Synthesis
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted triazoles typically require five sequential steps starting from quinoline-2-formic acid under severe reaction conditions, resulting in a mere 17% total yield that makes large-scale production economically unviable. These multi-step processes necessitate strict anhydrous and oxygen-free environments throughout multiple stages, demanding specialized equipment and extensive validation protocols that significantly increase capital expenditure. Each intermediate isolation step introduces potential degradation pathways and requires additional purification cycles that accumulate impurities incompatible with pharmaceutical standards. The reliance on transition metal catalysts creates dual challenges: high raw material costs and complex post-reaction cleanup procedures to meet stringent metal residue limits. Furthermore, the narrow substrate scope limits structural modifications without complete process redevelopment, restricting application flexibility for diverse drug discovery programs.
The Novel Approach
The patented method overcomes these limitations through an elegant one-pot oxidative cyclization that leverages tetrabutylammonium iodide as a dual-function catalyst and mediator. By using readily available 2-methylquinoline instead of quinoline carboxylic acids, the process avoids harsh decarboxylation steps while enabling direct functionalization at multiple positions. The reaction proceeds efficiently in standard organic solvents like DMSO without special atmosphere control, dramatically simplifying operational requirements while maintaining high yields across diverse substrates as shown in Table 2. This approach achieves superior atom economy by constructing both the triazole ring and quinoline linkage simultaneously through a cascade mechanism that minimizes intermediate handling. Crucially, the absence of metal catalysts ensures inherent compliance with ICH Q3D guidelines while reducing quality control testing requirements. The demonstrated scalability from milligram to gram quantities provides a clear roadmap for commercial implementation without fundamental process changes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307790B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
