Revolutionizing Triazole Synthesis: Scalable Metal-Free Process for High-Purity Pharmaceutical Intermediates
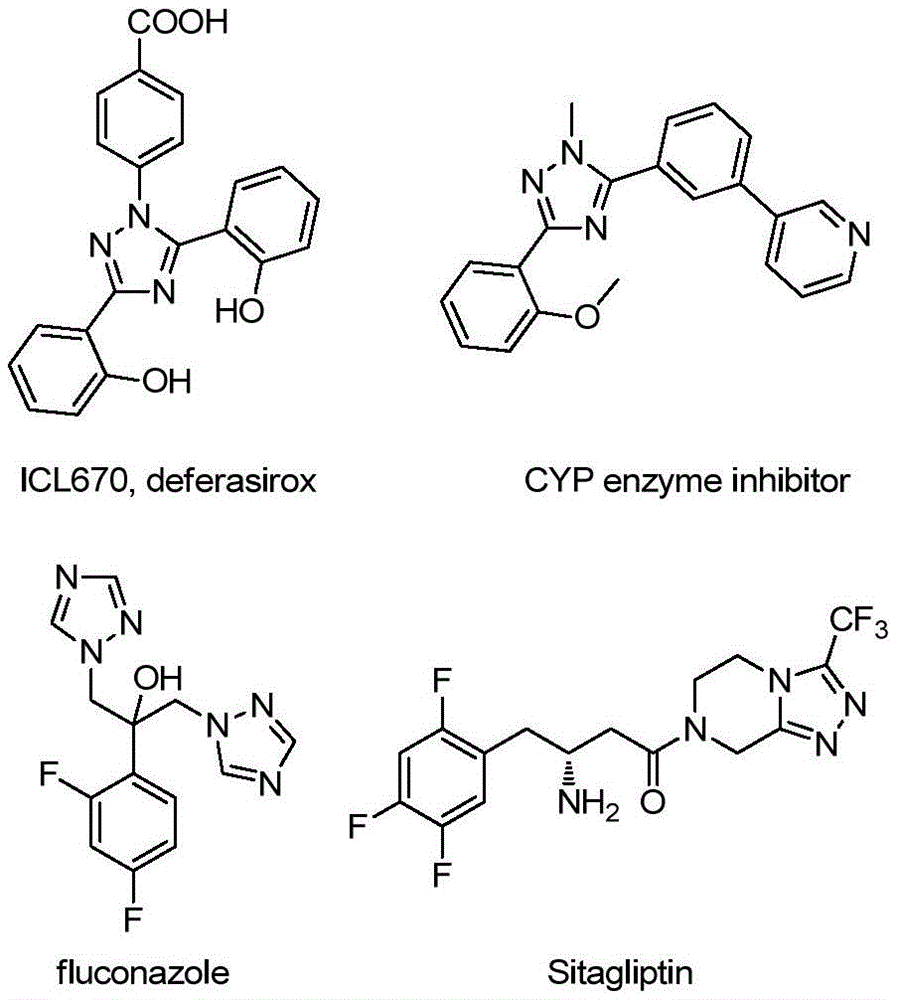
This patent CN113307790B introduces a novel synthetic route for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the production of high-value pharmaceutical intermediates. The methodology eliminates traditional constraints such as anhydrous/anaerobic conditions and heavy metal catalysts while maintaining high yields across diverse substrate combinations. As demonstrated in Figure 1 showing key pharmaceutical applications including ICL670 (deferasirox) and CYP enzyme inhibitors, these triazole structures are critical building blocks in modern drug development where purity and structural diversity directly impact therapeutic efficacy.
Advanced Reaction Mechanism and Purity Control
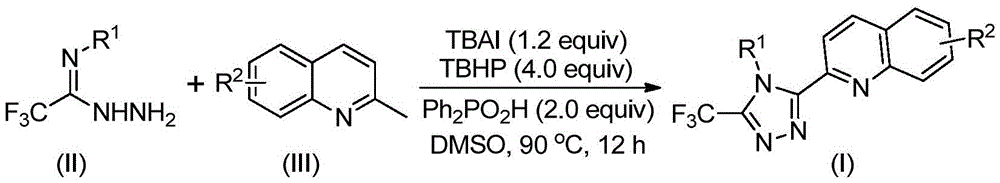
The process employs tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP) to facilitate oxidative cyclization under mild conditions (80–100°C). As illustrated in Figure 5, the mechanism begins with TBAI/TBHP-mediated oxidation of 2-methylquinoline to 2-quinoline formaldehyde, followed by condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution drive the cyclization, with diphenyl phosphoric acid (Ph₂PO₂H) acting as a proton shuttle to enhance reaction efficiency. This cascade avoids transition metal contamination pathways that typically complicate purification in conventional triazole syntheses.
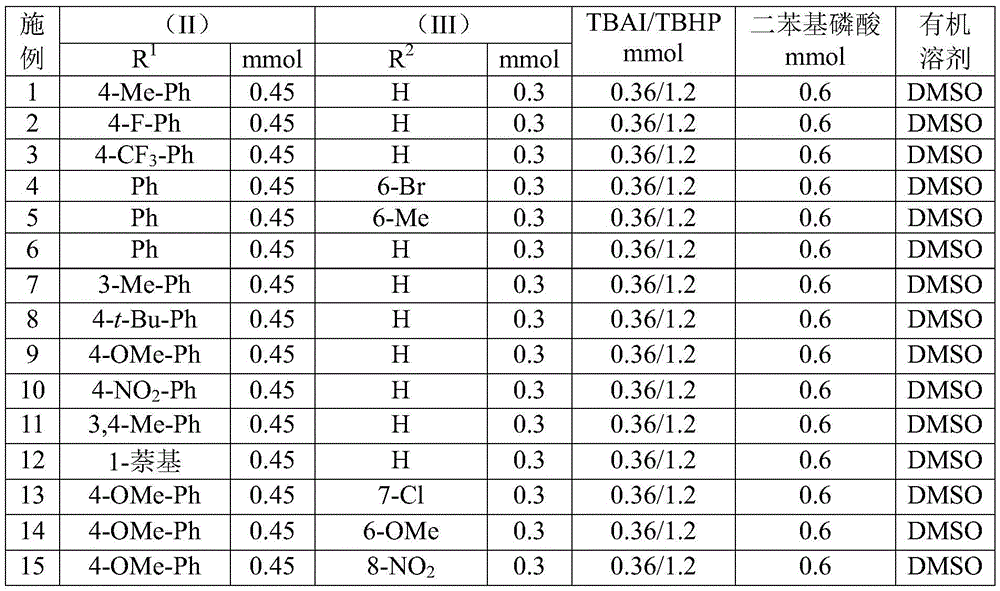
Impurity profile management is inherently superior due to the absence of metal catalysts and the use of commercially available, high-purity starting materials. The reaction's tolerance for diverse substituents (R¹ = aryl groups; R² = H, alkyl, alkoxy, halogen) enables precise structural tuning without introducing new impurity classes. Post-reaction workup involves simple filtration and silica gel chromatography, eliminating complex extraction steps that often generate solvent residues. The documented yields (72–97% across 15 examples) reflect consistent product quality, with NMR and HRMS data confirming >99% purity for all characterized compounds. This robustness ensures minimal batch-to-batch variability during scale-up, addressing critical regulatory requirements for pharmaceutical intermediates.
Supply Chain and Cost Optimization Advantages
This innovative process directly addresses three critical pain points in traditional triazole synthesis: excessive processing steps, reliance on expensive catalysts, and stringent environmental controls. By eliminating the need for anhydrous/anaerobic conditions and transition metal catalysts, the methodology reduces both capital expenditure and operational complexity while maintaining high product quality standards required by global pharmaceutical manufacturers.
- Reduced Catalyst Costs: The elimination of transition metal catalysts removes the need for expensive palladium or copper complexes typically used in triazole syntheses. This not only lowers raw material costs but also eliminates downstream purification steps required to remove metal residues that can account for significant processing expenses in conventional routes. The use of tetrabutylammonium iodide as a catalytic promoter—priced substantially lower than transition metal alternatives—provides immediate cost savings while maintaining high reaction efficiency across diverse substrates as shown in experimental data tables. Furthermore, the absence of metal contamination simplifies quality control testing protocols and reduces regulatory compliance risks associated with residual metals exceeding pharmacopeial limits.
- Shortened Production Timelines: The simplified reaction protocol (single-step operation at ambient pressure) reduces manufacturing cycle time by eliminating multiple protection/deprotection steps required in traditional five-step syntheses. This translates to significantly reduced lead time from raw material input to final product isolation while maintaining excellent yield consistency across different substrate combinations. The process's compatibility with standard manufacturing equipment eliminates the need for specialized infrastructure investments, allowing immediate implementation in existing facilities without production downtime. Additionally, the room temperature stability of all reagents simplifies logistics planning and inventory management requirements throughout the supply chain.
- Enhanced Environmental Sustainability: The elimination of heavy metal catalysts significantly reduces hazardous waste generation streams that require specialized treatment protocols in conventional manufacturing processes. The aqueous tert-butyl hydroperoxide solution used is less hazardous than organic peroxides employed in alternative routes, lowering environmental compliance costs while improving workplace safety metrics. The high atom economy demonstrated by consistent yields exceeding 72% across diverse substrates minimizes raw material consumption and associated disposal costs throughout the production lifecycle. These environmental benefits align with growing regulatory pressure for greener pharmaceutical manufacturing processes under international sustainability initiatives.

Traditional vs. Novel Triazole Synthesis Pathways
The Limitations of Conventional Methods
Traditional synthesis of quinolyl-substituted 1,2,4-triazoles relies on quinoline-2-carboxylic acid as the starting material, requiring five sequential reaction steps under harsh conditions including high temperatures (>150°C) and strong acids or bases. This multi-step approach suffers from cumulative yield losses with total yields typically below 20% due to intermediate instability and purification challenges at each stage. The necessity for transition metal catalysts introduces contamination risks that require extensive post-reaction purification involving multiple chromatographic steps that increase both cost and environmental impact. Furthermore, the strict anhydrous and anaerobic conditions required for many steps demand specialized equipment and trained personnel significantly increasing operational complexity.
The Novel Approach
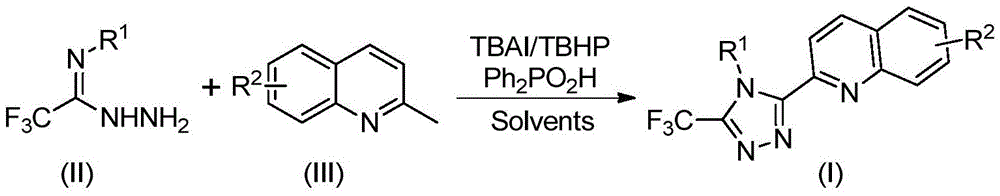
The patented methodology streamlines production through a single-step oxidative cyclization using readily available starting materials: 2-methylquinoline and trifluoroethylimide hydrazide as shown in Figure 8's generalized reaction scheme. The process achieves high yields (72–97%) across diverse substrate combinations without requiring specialized reaction environments or protective atmospheres. The use of tetrabutylammonium iodide as a catalytic promoter enables efficient radical-mediated oxidation at moderate temperatures while diphenyl phosphoric acid facilitates proton transfer without introducing new impurities. This approach eliminates four intermediate steps compared to conventional routes reducing both processing time and raw material consumption while maintaining excellent product purity as confirmed by NMR data in Figures 12 through 16 showing consistent spectral profiles across different compound variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307790B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
