Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
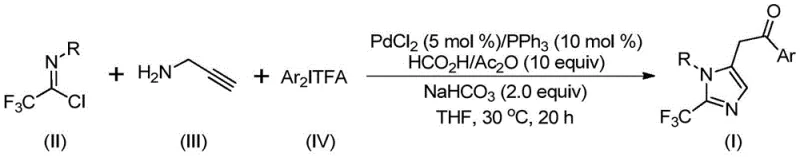
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on drug pharmacokinetics. As detailed in the groundbreaking patent CN111423381B, a novel preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed that addresses many historical challenges in heterocyclic chemistry. This technology leverages a sophisticated palladium-catalyzed carbonylation strategy to construct the imidazole core efficiently. The introduction of the trifluoromethyl moiety is known to significantly enhance electronegativity, bioavailability, metabolic stability, and lipophilicity of parent molecules, making these scaffolds highly desirable for modern drug discovery programs. By utilizing a multicomponent reaction system involving trifluoroacetimidoyl chloride, propargylamine, and diaryl iodonium salts, this process offers a streamlined pathway to complex molecular architectures that were previously difficult to access.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of synthons containing the trifluoromethyl group with suitable substrates. Common trifluoromethylating agents such as trifluorodiazoethane or specialized trifluoroethylimidoyl halides have been employed, yet these methods often suffer from significant drawbacks. These conventional routes frequently require harsh reaction conditions, expensive or hazardous reagents, and exhibit limited substrate scope, particularly when sensitive functional groups are present on the aromatic rings. Furthermore, the direct installation of the trifluoromethyl group onto a pre-formed imidazole ring can be regioselectively challenging and often results in poor yields or complex mixtures of isomers that are difficult to separate. The reliance on high-pressure carbon monoxide gas in some carbonylation protocols also poses significant safety and infrastructure barriers for standard laboratory or pilot plant operations, limiting the widespread adoption of these otherwise useful transformations in commercial settings.
The Novel Approach
The methodology disclosed in patent CN111423381B represents a paradigm shift by employing a transition metal palladium-catalyzed series reaction that operates under remarkably mild conditions. Instead of relying on dangerous gases or unstable diazo compounds, this innovative approach utilizes a safe and efficient carbon monoxide surrogate system generated in situ from formic acid and acetic anhydride. The reaction proceeds at a gentle temperature of 30°C, which is exceptionally low for such complex bond-forming events, thereby minimizing thermal degradation of sensitive intermediates. The use of cheap and easily obtainable starting materials, specifically trifluoroacetimidoyl chloride and propargylamine, drastically lowers the raw material costs compared to specialized fluorinated building blocks. Moreover, the process demonstrates exceptional compatibility with a wide range of functional groups, allowing for the synthesis of diversified 2-trifluoromethyl imidazole derivatives simply by varying the aryl substituents on the starting materials, thus broadening the utility of this method for generating diverse chemical libraries.
Mechanistic Insights into Palladium-Catalyzed Carbonylation and Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of bond formations orchestrated by the palladium center. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the base, likely sodium bicarbonate, to generate a trifluoroacetamidine intermediate. This species then undergoes isomerization to align the reactive centers for the subsequent metal-mediated steps. The palladium catalyst, activated by triphenylphosphine ligands, facilitates the palladation of the alkyne moiety within the propargylamine derivative, forming a crucial alkenyl palladium intermediate. This intermediate subsequently isomerizes to an alkyl palladium species, positioning the metal center for the critical carbonylation event. The carbon monoxide, released slowly and steadily from the decomposition of the formic acid/acetic anhydride mixture, inserts into the palladium-carbon bond to create an acyl palladium intermediate. Finally, the diaryl iodonium salt participates through an oxidative addition process to generate a high-valent tetravalent palladium species, which undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalytic species.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high purity in the final API intermediate. The use of a mild base like sodium bicarbonate helps to neutralize acidic byproducts without promoting unwanted side reactions such as hydrolysis of the sensitive imidoyl chloride or the final ketone product. The stepwise nature of the catalytic cycle, particularly the controlled release of carbon monoxide, prevents the accumulation of reactive acyl species that could lead to polymerization or oligomerization byproducts. Furthermore, the specificity of the oxidative addition step with the diaryl iodonium salt ensures that the aryl group is installed selectively at the desired position, minimizing the formation of regioisomers that often plague electrophilic aromatic substitution methods. The ability to tune the electronic properties of the aryl groups on both the imidoyl chloride and the iodonium salt allows chemists to fine-tune the reaction kinetics, further suppressing the formation of trace impurities and simplifying the downstream purification process.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemistry teams looking to implement new routes quickly. The procedure involves charging a reaction vessel with the palladium catalyst system, the carbon monoxide source, and the three key organic components in a suitable aprotic solvent such as tetrahydrofuran. The reaction is then allowed to stir at ambient or slightly elevated temperatures for a defined period, typically around 20 hours, to ensure full conversion. Post-reaction processing is straightforward, involving basic filtration to remove inorganic salts and palladium black, followed by standard chromatographic purification. This eliminates the need for complex quenching procedures or specialized high-pressure equipment, making the technology accessible even to facilities with limited infrastructure. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup parameters, please refer to the standardized protocol below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroacetimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from exotic fluorinating reagents towards commodity chemicals like propargylamine and simple aromatic amines significantly de-risks the supply chain. Since the starting materials are widely available from multiple global vendors, the risk of supply disruption due to single-source dependency is drastically minimized. Additionally, the mild reaction conditions translate to lower energy consumption during the manufacturing process, as there is no need for cryogenic cooling or high-temperature heating, contributing to substantial cost savings in utility expenditures. The high atom economy and efficient conversion rates mean that less raw material is wasted, further enhancing the overall cost-effectiveness of the production campaign.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as trifluorodiazoethane or high-pressure carbon monoxide cylinders leads to a significant reduction in operational expenditures. By utilizing palladium chloride, a relatively inexpensive palladium source compared to other precious metal complexes, and common ligands like triphenylphosphine, the catalyst cost is kept to a minimum. The simplified workup procedure, which avoids complex extractions or distillations, reduces labor hours and solvent usage, resulting in a leaner and more cost-efficient manufacturing process that enhances profit margins for high-volume production.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials ensures consistent quality and availability throughout the year. Trifluoroacetimidoyl chlorides and diaryl iodonium salts can be stockpiled without significant degradation, allowing for better inventory management and just-in-time manufacturing strategies. The robustness of the reaction against moisture and air, facilitated by the choice of reagents and conditions, reduces the likelihood of batch failures due to environmental factors, thereby guaranteeing reliable delivery schedules to downstream customers and preventing costly production delays.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable from milligram to gram levels with consistent yields, indicating a smooth path to kilogram and ton-scale production. The use of tetrahydrofuran as a solvent, which is easily recoverable and recyclable, aligns with green chemistry principles and simplifies waste management. Furthermore, the avoidance of toxic heavy metal waste streams associated with stoichiometric fluorination reagents simplifies environmental compliance and reduces the costs associated with hazardous waste disposal, making this process sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
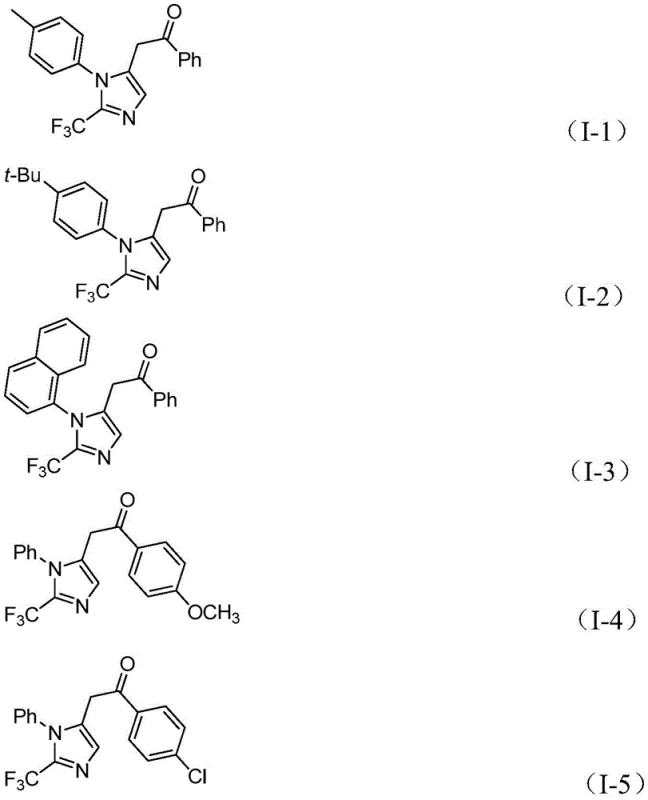
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic methodology. These insights are derived directly from the experimental data and scope studies presented in the patent documentation, providing clarity on reaction parameters and substrate limitations. Understanding these nuances is critical for R&D teams planning to integrate this chemistry into their existing pipelines for API intermediate synthesis.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method utilizes cheap and readily available starting materials like propargylamine and trifluoroacetimidoyl chloride under mild conditions (30°C), avoiding the harsh reagents often required for direct trifluoromethylation. It also offers excellent functional group tolerance and high reaction efficiency.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride act as a carbon monoxide surrogate system. They generate CO in situ, which is essential for the carbonylation step that constructs the imidazole ring backbone without requiring high-pressure CO gas cylinders.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the patent explicitly mentions that the method is operable on a gram level and provides possibilities for industrial large-scale production due to its simple operation, inexpensive catalysts, and straightforward post-treatment procedures involving filtration and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic building blocks in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the potential of this palladium-catalyzed carbonylation route and is fully equipped to leverage its advantages for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee the quality of every batch.
We invite you to collaborate with us to optimize this synthesis for your target molecules. Our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for process intensification and waste reduction. Please contact our technical procurement team today to request specific COA data for related imidazole intermediates and to discuss route feasibility assessments for your proprietary compounds. Let us be your partner in transforming innovative chemistry into commercial reality.
