Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds, particularly those containing nitrogen-rich motifs like 1,2,4-triazoles. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy. By leveraging a metal-free catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), this method achieves high yields under mild conditions, addressing critical pain points regarding cost, safety, and environmental compliance in modern API manufacturing. For R&D directors and procurement specialists, this innovation offers a reliable pathway to access high-value intermediates that are pivotal in the development of bioactive molecules and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale commercial application. Traditional methodologies often rely on quinoline-2-carboxylic acid as a primary starting material, necessitating a cumbersome five-step reaction sequence to construct the desired heterocyclic core. This linear approach not only suffers from a dismal overall yield of approximately 17%, leading to significant material waste and inflated production costs, but it also typically demands severe reaction conditions that pose safety risks and operational challenges. Furthermore, many conventional routes depend on the use of transition metal catalysts, which introduce the risk of heavy metal contamination in the final product—a critical quality attribute that requires expensive and time-consuming purification steps to meet stringent regulatory standards for pharmaceutical intermediates. These cumulative drawbacks make legacy methods economically unviable for the cost-sensitive and high-volume demands of the global supply chain.
The Novel Approach
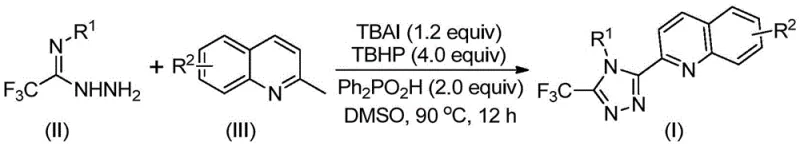
In stark contrast, the novel methodology outlined in the patent utilizes readily available and inexpensive 2-methylquinoline and trifluoroacetimidohydrazide as starting materials, bypassing the need for pre-functionalized carboxylic acid derivatives. The core of this innovation lies in the use of TBAI and TBHP to promote an oxidative cyclization reaction directly, effectively merging oxidation and ring-closure into a single operational unit. This approach eliminates the need for strict anhydrous or oxygen-free environments, significantly simplifying reactor setup and operational protocols. The reaction proceeds smoothly in polar aprotic solvents like DMSO at temperatures between 80°C and 100°C, delivering the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compounds with impressive efficiency. By avoiding toxic heavy metals entirely, this route inherently produces a cleaner crude product profile, reducing the burden on downstream purification processes and aligning perfectly with green chemistry principles.
Mechanistic Insights into Organocatalytic Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated example of iodine-mediated radical chemistry that ensures high selectivity and conversion. Initially, the tetrabutylammonium iodide acts as a catalyst precursor which, in the presence of the oxidant tert-butyl hydroperoxide, generates reactive iodine species in situ. These species facilitate the selective oxidation of the methyl group on the 2-methylquinoline substrate to an aldehyde intermediate, specifically 2-quinolinecarbaldehyde. This aldehyde then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Subsequently, the system promotes an oxidative iodination followed by an intramolecular electrophilic substitution and aromatization sequence. This cascade effectively constructs the 1,2,4-triazole ring while simultaneously installing the trifluoromethyl group at the 5-position and the quinolyl moiety at the 3-position. The presence of diphenylphosphoric acid as an additive further enhances the reaction efficiency, likely by stabilizing intermediates or facilitating proton transfer steps crucial for the cyclization event.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the formation of metal-complexed byproducts that are notoriously difficult to remove. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups on the aromatic rings, such as methoxy or halogen substituents, ensuring a clean impurity profile. The use of TBHP as a terminal oxidant results in tert-butanol and water as the primary byproducts, which are easily removed during workup. This mechanistic elegance translates directly to process robustness, allowing for the synthesis of diverse derivatives with consistent quality, a factor that is paramount for maintaining batch-to-batch consistency in commercial pharmaceutical production.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly attractive for process chemists aiming to scale up from laboratory discovery to pilot plant production. The protocol involves simply charging the reactor with the requisite molar ratios of tetrabutylammonium iodide, aqueous tert-butyl hydroperoxide, diphenylphosphoric acid, trifluoroacetimidohydrazide, and the specific 2-methylquinoline derivative in a solvent like DMSO. The mixture is then heated to the optimal temperature range of 80-100°C and stirred for a duration of 8 to 14 hours until conversion is complete. Post-reaction processing is straightforward, typically involving filtration to remove insoluble salts followed by standard silica gel column chromatography to isolate the pure product. This simplicity reduces the technical barrier for adoption and minimizes the requirement for specialized equipment.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements. Moreover, the use of commodity chemicals like 2-methylquinoline and TBAI ensures a stable and diversified supply base, reducing the risk of raw material shortages that can disrupt production schedules. The operational simplicity of the process, which does not require inert atmosphere techniques or cryogenic conditions, allows for utilization of standard glass-lined or stainless steel reactors, thereby maximizing asset utilization and minimizing capital expenditure requirements for new capacity.
- Cost Reduction in Manufacturing: The economic impact of this metal-free protocol is profound, primarily driven by the drastic simplification of the purification train. In traditional heavy-metal catalyzed processes, a substantial portion of the manufacturing budget is allocated to scavenging resins and extensive washing protocols to reduce metal residues to ppm levels required by pharmacopeial standards. By completely obviating the need for metal catalysts, this method inherently delivers a product with negligible metal content, thereby slashing downstream processing costs and shortening the overall cycle time. Additionally, the high atom economy and superior yields observed across a broad substrate scope mean that less raw material is wasted, directly improving the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, bulk commodity starting materials rather than specialized, custom-synthesized building blocks. 2-Methylquinolines and trifluoroacetimidohydrazides are produced by multiple global suppliers, creating a competitive market that drives down prices and ensures continuity of supply. The robustness of the reaction conditions, which tolerate moisture and oxygen to a significant degree, reduces the likelihood of batch failures due to minor deviations in operating parameters. This reliability translates into more predictable lead times and the ability to commit to longer-term supply agreements with confidence, a critical factor for pharmaceutical companies managing complex drug development timelines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern sustainability goals. The avoidance of toxic heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing facility. The reaction generates benign byproducts, and the use of DMSO, a solvent with a well-understood recovery profile, facilitates efficient recycling programs. The scalability of the process has been validated through successful gram-scale demonstrations, indicating a clear path to kilogram and ton-scale production without the need for fundamental process re-engineering. This ease of scale-up ensures that the technology can meet the surging demand for these intermediates as they progress through clinical trials to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier selection.
Q: Does this synthesis method require toxic heavy metal catalysts?
A: No, the method described in patent CN113307790B utilizes an organocatalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for expensive and toxic transition metal catalysts.
Q: What is the substrate scope for the quinoline component in this reaction?
A: The process demonstrates excellent tolerance for various substituents on the quinoline ring, including hydrogen, methyl, methoxy, halogens (Cl, Br), and nitro groups at different positions, allowing for the synthesis of diverse derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under relatively mild conditions (80-100°C) without strict anhydrous or oxygen-free requirements, uses cheap and commercially available starting materials, and has been demonstrated to be easily expandable to gram-scale reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113307790B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this organocatalytic process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering high-purity pharmaceutical intermediates that enable our partners to focus on what they do best: innovating life-saving therapies.
We invite you to engage with our technical team to discuss how this cost-effective and scalable synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
