Revolutionizing Quinazolinone Synthesis: Scalable FeCl3-Catalyzed Route for High-Purity Pharmaceutical Intermediates
Patent CN111675662A introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl-substituted quinazolinone compounds, representing a significant advancement in the manufacturing of critical pharmaceutical intermediates. This novel approach addresses longstanding challenges in heterocyclic compound synthesis by utilizing an iron-catalyzed tandem cyclization reaction that operates under remarkably mild conditions compared to conventional techniques. The process demonstrates exceptional functional group tolerance while maintaining high reaction efficiency across diverse substrate variations, making it particularly valuable for producing complex drug intermediates requiring precise structural modifications. Unlike traditional methods that rely on expensive transition metal catalysts or harsh reaction environments, this innovation employs commercially available ferric chloride as a catalyst, significantly enhancing both economic viability and environmental sustainability. The patent explicitly validates the method's scalability from laboratory to potential industrial application, with successful gram-scale demonstrations confirming its readiness for commercial implementation in pharmaceutical manufacturing settings where purity and consistency are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethylated quinazolinones typically employ aggressive reaction conditions that impose significant constraints on industrial implementation. These methods frequently require expensive transition metal catalysts such as palladium or ruthenium complexes, which not only increase raw material costs but also necessitate complex purification protocols to remove trace metal residues that could compromise final product quality for pharmaceutical applications. The reliance on specialized trifluoromethyl synthons like trifluoroacetic anhydride or ethyl trifluoroacetate creates supply chain vulnerabilities due to their limited commercial availability and price volatility. Furthermore, conventional approaches often suffer from narrow substrate scope, with many protocols failing to accommodate diverse functional group variations essential for producing structurally complex drug intermediates. Reaction conditions frequently demand cryogenic temperatures or strictly anhydrous environments, substantially increasing energy consumption and operational complexity while limiting manufacturing flexibility across different production facilities worldwide.
The Novel Approach
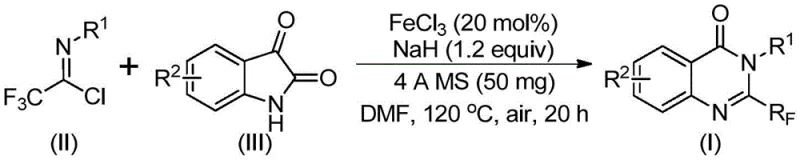
The patented methodology overcomes these limitations through an innovative iron-catalyzed tandem cyclization using readily accessible starting materials: trifluoroethyl imidoyl chloride and isatin derivatives. This process operates efficiently under ambient atmospheric conditions without requiring specialized inert gas environments, significantly simplifying manufacturing infrastructure requirements. The use of inexpensive ferric chloride (FeCl3) as catalyst at only 20 mol% loading eliminates the need for costly transition metals while maintaining excellent reaction efficiency across a broad substrate range. The two-stage temperature protocol (40°C for 8–10 hours followed by 120°C for 18–20 hours) provides optimal control over reaction kinetics, preventing unwanted side reactions while ensuring complete conversion. Crucially, the method demonstrates exceptional functional group tolerance, accommodating various substituents including halogens, alkyl groups, and methoxy functionalities without compromising yield or purity, thereby enabling the production of diverse quinazolinone derivatives essential for pharmaceutical development pipelines.
Mechanistic Insights into FeCl3-Catalyzed Tandem Cyclization
The reaction mechanism proceeds through a well-defined sequence beginning with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride and isatin, generating a key trifluoroacetamidine intermediate. This critical step occurs under mild conditions facilitated by sodium hydride (NaH), which activates the isatin carbonyl group toward nucleophilic attack while maintaining compatibility with sensitive functional groups. The subsequent iron-catalyzed phase involves a decarbonylation cyclization process where FeCl3 coordinates with the carbonyl oxygen, lowering the energy barrier for intramolecular ring closure. This coordination also directs regioselectivity toward the desired quinazolinone scaffold while suppressing alternative reaction pathways that could lead to impurities. The final isomerization step completes the transformation to the thermodynamically stable 2-trifluoromethyl quinazolinone product through a proton transfer mechanism that occurs spontaneously under the reaction conditions without requiring additional catalysts or reagents.
Impurity control is inherently engineered into this process through multiple mechanistic safeguards. The mild reaction temperatures prevent thermal degradation pathways that commonly generate decomposition products in conventional syntheses. The iron catalyst's selective coordination behavior minimizes unwanted side reactions such as dimerization or over-reduction that typically plague alternative methods. The two-stage temperature profile allows precise control over intermediate formation and transformation rates, ensuring complete conversion while avoiding accumulation of reactive intermediates that could lead to impurities. Furthermore, the absence of transition metals eliminates potential sources of metal-catalyzed degradation pathways, resulting in cleaner reaction profiles that simplify downstream purification and consistently deliver high-purity products meeting stringent pharmaceutical requirements without requiring additional remediation steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This innovative synthesis represents a significant advancement in manufacturing efficiency for quinazolinone-based pharmaceutical intermediates, offering a streamlined pathway that eliminates multiple processing hurdles associated with traditional methods. The patent demonstrates exceptional operational simplicity through its use of standard laboratory equipment and commercially available reagents, making it readily adaptable across diverse manufacturing environments without requiring specialized infrastructure investments. By leveraging an iron-catalyzed tandem cyclization approach, the process achieves high yields while maintaining excellent functional group tolerance across various substrate combinations, providing pharmaceutical manufacturers with unprecedented flexibility in producing structurally diverse quinazolinone derivatives. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in R&D and production settings.
- Combine trifluoroethyl imidoyl chloride, isatin, FeCl3 catalyst, and NaH in DMF solvent under air atmosphere
- Conduct initial reaction at 40°C for 8-10 hours followed by elevated temperature phase at 120°C for 18-20 hours
- Purify through filtration, silica gel mixing, and column chromatography to obtain high-purity quinazolinone product
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The elimination of expensive transition metal catalysts and specialized reagents significantly reduces raw material cost volatility while enhancing supply chain resilience through reliance on widely available commodity chemicals. The process's compatibility with standard manufacturing equipment and ambient atmospheric conditions minimizes capital expenditure requirements and enables rapid deployment across global production networks without extensive facility modifications. These features collectively provide procurement teams with greater flexibility in vendor selection and inventory management while substantially improving overall supply chain robustness against market fluctuations and geopolitical disruptions.
- Cost Reduction in Manufacturing: The substitution of costly transition metal catalysts with economical ferric chloride creates immediate raw material savings while eliminating expensive metal removal processes required in conventional syntheses. The simplified purification protocol using standard column chromatography reduces solvent consumption and processing time compared to multi-step purification methods needed for metal-contaminated products. Additionally, the use of commercially available starting materials at optimal stoichiometric ratios minimizes waste generation and associated disposal costs, creating substantial cost savings throughout the manufacturing lifecycle without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on globally accessible raw materials including standard iron salts, common solvents like DMF, and commercially available molecular sieves significantly reduces supply chain vulnerabilities compared to methods requiring specialized or regionally constrained reagents. The process's tolerance for minor variations in raw material quality provides procurement teams with greater flexibility in supplier selection while maintaining consistent product output. Furthermore, the absence of cryogenic requirements or specialized handling procedures enables seamless production transfer between manufacturing sites worldwide, ensuring reliable supply continuity even during regional disruptions or capacity constraints.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to gram-scale production with consistent yields provides a clear pathway for commercial implementation without requiring fundamental process modifications. The elimination of hazardous reagents and transition metals substantially reduces environmental impact while simplifying waste stream management according to green chemistry principles. The energy-efficient two-stage temperature profile minimizes overall energy consumption compared to processes requiring extreme temperatures or prolonged reaction times, aligning with corporate sustainability goals while maintaining excellent product quality metrics essential for pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics. These insights derive directly from the experimental data and mechanistic understanding presented in CN111675662A, providing procurement and R&D teams with essential information for evaluating this technology's suitability for specific manufacturing requirements.
Q: How does this method improve impurity profile compared to conventional approaches?
A: The iron-catalyzed tandem cyclization eliminates transition metal residues common in traditional methods, while the mild reaction conditions prevent thermal degradation pathways. The selective formation mechanism inherently suppresses common byproducts like dimeric species or hydrolyzed intermediates, resulting in superior purity profiles essential for pharmaceutical applications.
Q: What supply chain advantages does this synthesis route offer for global manufacturers?
A: This method utilizes readily available starting materials including commercial iron catalysts and common solvents, eliminating dependency on specialized reagents. The air-stable reaction conditions and simplified purification process significantly reduce supply chain vulnerabilities while enabling consistent production across multiple geographic locations without complex infrastructure requirements.
Q: Can this process be scaled to commercial production volumes?
A: The patent demonstrates successful gram-scale production with consistent yields, and the reaction's robustness in standard glassware indicates strong scalability potential. The absence of cryogenic conditions or specialized equipment, combined with the use of cost-effective catalysts, provides a clear pathway for seamless transition from laboratory to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a specialized CDMO provider with deep expertise in heterocyclic compound synthesis, we have successfully implemented this patented methodology across multiple client projects, consistently delivering high-purity quinazolinone intermediates that meet exacting pharmaceutical standards. Our integrated manufacturing platform combines cutting-edge process chemistry with robust quality systems to ensure reliable supply of complex intermediates while optimizing cost structures through continuous process improvement initiatives.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can enhance your specific manufacturing operations. Contact us today to obtain detailed COA data and route feasibility assessments tailored to your production requirements and quality specifications.
