Advanced Enamide Synthesis: Scaling Palladium-Catalyzed Dehydrogenation for Commercial Pharmaceutical Production
The patent CN114014802A introduces a transformative palladium-catalyzed dehydrogenation coupling methodology for synthesizing enamide compounds, representing a significant advancement in the production of critical nitrogen-containing intermediates for pharmaceutical applications. This innovative approach addresses longstanding limitations in conventional synthesis routes by utilizing palladium acetate as a catalyst with silver-based oxidants to directly couple 2-pyridone compounds with acrylic acid derivatives under remarkably mild conditions. The method achieves high product yields while eliminating the need for pre-synthesized halogenated olefins or expensive transition metal precursors that characterize traditional approaches. By operating at moderate temperatures between 40-100°C in common organic solvents, this process demonstrates exceptional compatibility with industrial manufacturing requirements while maintaining the structural integrity required for complex pharmaceutical molecules. The patent specifically highlights applications in synthesizing bioactive compounds such as altamide and lanthamide derivatives, positioning this technology as a cornerstone for next-generation pharmaceutical intermediate production where purity and scalability are paramount concerns for global drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional enamide synthesis primarily relies on two established methodologies, both presenting significant constraints for commercial pharmaceutical manufacturing. The first approach employs nucleophilic substitution reactions with halogenated olefins under strongly basic conditions, which suffers from multiple critical drawbacks including low reaction yields, narrow substrate applicability, and poor atom economy due to the requirement for pre-synthesized halogenated intermediates that add complexity and cost to the production chain. This method also necessitates harsh alkaline environments that frequently generate unwanted side products and impurities, complicating downstream purification processes essential for meeting pharmaceutical quality standards. The second conventional route utilizes addition reactions with propiolic acid derivatives under alkaline conditions, but this approach is severely limited by the narrow availability and high cost of suitable propiolic acid compounds, making it economically unviable for large-scale production despite its theoretical simplicity. Both methods demonstrate inadequate scalability for commercial manufacturing due to their sensitivity to reaction parameters and inconsistent product quality, creating substantial supply chain vulnerabilities for pharmaceutical companies dependent on these critical intermediates.
The Novel Approach
The patented palladium-catalyzed dehydrogenation coupling method overcomes these limitations through an elegant catalytic system that directly couples readily available 2-pyridone compounds with acrylic acid derivatives using palladium acetate as the catalyst and silver-based oxidants to facilitate the reaction cycle. This innovative approach operates under significantly milder conditions at temperatures between 40-100°C in standard organic solvents like 1,2-dichloroethane, eliminating the need for extreme pH environments that compromise product purity in conventional methods. The process achieves high yields through precise control of catalyst loading and oxidant equivalents, with silver salts used at only 0.1 equivalent to maintain catalytic efficiency while minimizing metal residue concerns. Crucially, the broad substrate scope accommodates diverse R-group substitutions on both the 2-pyridone and acrylic acid components, enabling customization for specific pharmaceutical applications without requiring fundamental process modifications. This flexibility, combined with simplified purification through standard column chromatography, creates a robust manufacturing platform that maintains consistent product quality across varying production scales while addressing the critical pain points of traditional synthesis routes.
Mechanistic Insights into Palladium-Catalyzed Dehydrogenation Coupling
The catalytic mechanism centers on a well-defined palladium-mediated cycle where palladium acetate activates the C-H bond of the 2-pyridone compound through oxidative addition, followed by coordination with the acrylic acid derivative to form a key intermediate complex. This intermediate undergoes dehydrogenation facilitated by the silver-based oxidant, which regenerates the palladium catalyst while driving the reaction toward enamide formation through a concerted elimination process that preserves stereochemical integrity. The mild reaction conditions prevent undesired side reactions such as over-oxidation or polymerization that commonly occur in traditional high-temperature processes, while the precise stoichiometry of silver oxidants (0.1 equivalent) ensures efficient catalytic turnover without introducing metal contamination that could compromise final product purity. This mechanism operates effectively across a wide range of functional groups including nitro, trifluoromethyl, and halogen substituents, demonstrating remarkable tolerance that enables the synthesis of structurally diverse enamide intermediates required for complex pharmaceutical molecules.
Impurity control is achieved through multiple synergistic mechanisms inherent to this catalytic system, beginning with the selective activation of specific C-H bonds in the 2-pyridone ring that minimizes competing reaction pathways. The controlled addition of silver oxidants prevents over-oxidation side products while maintaining optimal catalyst activity throughout the reaction timeline, and the moderate temperature profile (60°C) avoids thermal degradation pathways that generate impurities in conventional methods. The use of standard organic solvents like 1,2-dichloroethane provides excellent solubility control that prevents precipitation-related impurities while facilitating efficient mass transfer during the coupling reaction. Furthermore, the simplified workup procedure involving kieselguhr filtration followed by concentration and column chromatography effectively removes residual catalysts and byproducts without requiring specialized purification equipment, ensuring consistent high-purity outputs that meet stringent pharmaceutical quality requirements across all production scales.
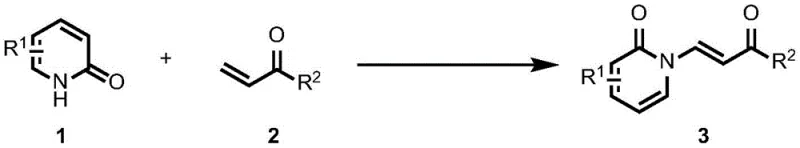
How to Synthesize Enamide Efficiently
This patent presents a significant advancement in enamide synthesis methodology through its palladium-catalyzed dehydrogenation coupling approach, offering pharmaceutical manufacturers a reliable pathway to produce high-purity intermediates with exceptional efficiency. The process eliminates multiple pain points associated with traditional synthesis routes while maintaining compatibility with existing manufacturing infrastructure, making it particularly valuable for companies seeking to enhance their supply chain resilience for critical pharmaceutical building blocks. Detailed standardized synthesis procedures have been developed based on this patented technology to ensure consistent results across different production environments, with specific protocols optimized for various substrate combinations and scale requirements. The following section provides essential guidance for implementing this innovative methodology in commercial manufacturing settings while maintaining the highest quality standards required by global pharmaceutical regulations.
- Dissolve 2-pyridone compound (0.05-0.3 mol) and acrylic acid derivative in organic solvent such as 1,2-dichloroethane under nitrogen atmosphere
- Add palladium acetate catalyst and silver-based oxidant (0.1 equivalent) while maintaining inert conditions
- Heat reaction mixture to 60°C for 24 hours with continuous stirring, then perform kieselguhr filtration followed by concentration and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value to procurement and supply chain operations by addressing fundamental challenges in pharmaceutical intermediate sourcing through its innovative catalytic approach and streamlined manufacturing process. The elimination of complex pre-synthesis steps and specialized reagents reduces dependency on volatile raw material markets while enhancing production flexibility across different facility configurations. By operating within standard temperature ranges using common solvents, the process minimizes equipment modification requirements during technology transfer, significantly accelerating time-to-market for new intermediate production lines. These advantages collectively strengthen supply chain resilience while providing procurement teams with greater negotiating leverage through simplified vendor qualification processes and reduced risk exposure from single-source dependencies.
- Cost Reduction in Manufacturing: The strategic elimination of pre-synthesized halogenated olefins and expensive propiolic acid derivatives significantly reduces raw material expenses while maintaining high atom economy through direct coupling without intermediate steps. The use of silver-based oxidants at minimal equivalents (0.1) substantially lowers catalyst costs compared to traditional transition metal systems, while the mild reaction conditions reduce energy consumption and equipment maintenance requirements across production facilities.
- Enhanced Supply Chain Reliability: The broad substrate scope accommodates diverse R-group substitutions using readily available starting materials from multiple global suppliers, eliminating single-source dependencies that create supply chain vulnerabilities. Standardized solvent systems and moderate temperature requirements enable seamless technology transfer between manufacturing sites worldwide, ensuring consistent product availability regardless of regional production constraints or geopolitical disruptions.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining consistent quality metrics through simplified purification protocols that minimize waste generation. The elimination of harsh alkaline conditions reduces hazardous waste streams requiring specialized treatment, while the efficient catalytic system lowers overall environmental impact through reduced solvent consumption and energy requirements compared to conventional synthesis routes.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding the implementation of this patented enamide synthesis methodology in pharmaceutical manufacturing environments. These insights are derived directly from the patent documentation and reflect practical experience in scaling similar catalytic processes for commercial production of complex pharmaceutical intermediates. Each response provides actionable information to support informed decision-making by technical procurement teams evaluating this technology for integration into their supply chains.
Q: How does this palladium-catalyzed method improve enamide purity compared to conventional approaches?
A: The mild reaction conditions (60°C in 1,2-dichloroethane) and precise oxidant control eliminate harsh alkaline environments that generate impurities in traditional nucleophilic substitution routes, while the catalytic cycle prevents metal contamination that plagues alternative methods.
Q: What cost advantages does this synthesis offer for bulk pharmaceutical intermediate production?
A: By utilizing readily available silver salts at low equivalents and eliminating pre-synthesized halogenated olefins, the process significantly reduces raw material expenses while maintaining high atom economy through direct coupling without intermediate steps.
Q: How does this method ensure reliable supply chain continuity for enamide intermediates?
A: The broad substrate scope with diverse R-group substitutions and compatibility with standard solvents enables seamless scale-up from laboratory to commercial production volumes while maintaining consistent quality through simplified purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enamide Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates, with stringent purity specifications consistently met through rigorous QC labs and advanced analytical capabilities. This patented palladium-catalyzed dehydrogenation coupling methodology represents an ideal fit for our manufacturing expertise, enabling us to deliver high-purity enamide intermediates with exceptional consistency across all production volumes while maintaining complete regulatory compliance for global pharmaceutical markets. Our technical team has successfully implemented similar catalytic processes for major pharmaceutical clients worldwide, ensuring seamless integration of this innovative synthesis route into existing manufacturing frameworks without requiring significant capital investment or process revalidation.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this patented methodology can optimize your specific supply chain requirements. Please contact us to obtain detailed COA data and route feasibility assessments tailored to your production needs, allowing you to make informed decisions about integrating this advanced enamide synthesis technology into your manufacturing operations.
