Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding safety, efficiency, and scalability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from traditional, hazard-prone methodologies towards a more sustainable and operationally simple base-promoted cyclization. For R&D directors and procurement specialists alike, this patent offers a compelling alternative for constructing critical pharmacophores found in beta-3 adrenergic receptor agonists and various agrochemical agents. By leveraging cheap and readily available diazo compounds and trifluoroethylimidoyl chlorides, this process eliminates the reliance on toxic azides and expensive transition metal catalysts, thereby streamlining the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by two primary synthetic strategies, both of which carry substantial drawbacks for large-scale manufacturing. The first common approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides to form triazole metal complexes, which subsequently react with trifluoromethyl reagents. The second strategy employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. While chemically effective on a small scale, both methods share a critical vulnerability: the mandatory use of organic azides. Azides are notoriously toxic and possess high explosive potential, creating severe safety hazards that complicate regulatory compliance and increase insurance costs for chemical plants. Furthermore, the reliance on copper catalysts introduces the risk of heavy metal contamination in the final API, necessitating costly and time-consuming purification steps to meet stringent residual metal specifications required by global health authorities.
The Novel Approach
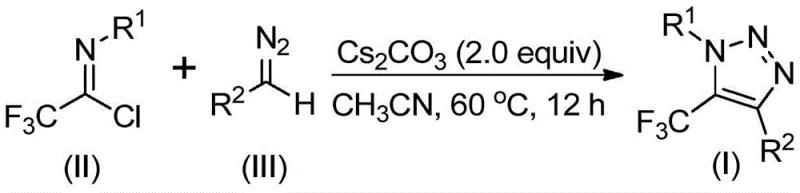
In stark contrast to these legacy methods, the technology described in patent CN113121462B introduces a metal-free, azide-free pathway that fundamentally alters the risk profile of triazole synthesis. As illustrated in the reaction scheme below, this innovative route utilizes a base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds. This approach not only bypasses the safety liabilities associated with azide handling but also removes the need for transition metals entirely. The result is a cleaner reaction profile that significantly simplifies downstream processing. By avoiding heavy metal catalysts, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through the elimination of specialized scavenging resins and extensive metal testing protocols. Moreover, the reaction conditions are remarkably mild, typically proceeding at temperatures between 50°C and 70°C, which enhances energy efficiency and allows for the tolerance of sensitive functional groups that might degrade under harsher catalytic conditions.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for diverse substrate scopes. The reaction is hypothesized to proceed through a sophisticated cascade initiated by the base, typically cesium carbonate. Initially, the base promotes an intermolecular nucleophilic addition-elimination process where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is critical as it establishes the new carbon-carbon bond necessary for ring closure while expelling the chloride leaving group. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is energetically favorable in this system due to the electronic activation provided by the trifluoromethyl group and the adjacent nitrogen atoms. The final aromatization step yields the stable 5-trifluoromethyl-1,2,3-triazole core. This mechanism highlights the elegance of the design, as it achieves complex heterocyclic construction through simple acid-base chemistry rather than complex organometallic cycles.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional copper-catalyzed routes often suffer from the formation of regioisomers (1,4- vs 1,5-disubstituted triazoles) and metal-complexed byproducts that are difficult to separate. In this base-promoted system, the regioselectivity is inherently controlled by the structure of the starting imidoyl chloride and diazo compound, leading to a highly defined substitution pattern at the 1 and 4 positions of the triazole ring. The absence of metal coordination complexes means that the impurity profile is largely composed of organic byproducts that can be effectively removed via standard silica gel chromatography or crystallization. This predictability in the impurity spectrum is invaluable for quality control teams, as it reduces the analytical burden and ensures consistent batch-to-batch purity, a key requirement for reliable pharmaceutical intermediate suppliers.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The protocol requires minimal specialized equipment, relying on standard Schlenk techniques or even simple flask setups under inert atmosphere if necessary, though the robustness of the reagents often allows for less stringent conditions. The key to success lies in the precise stoichiometric balance of the base and the selection of the appropriate aprotic solvent to maximize solubility and reaction kinetics. Detailed standardized synthetic steps for replicating this high-efficiency transformation are provided in the guide below.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Filter the reaction mixture, concentrate, and purify the crude product via silica gel column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational benefits and risk mitigation. The shift away from hazardous reagents and expensive catalysts creates a more resilient supply chain that is less susceptible to regulatory bottlenecks and raw material price volatility. By focusing on commodity chemicals like cesium carbonate and readily synthesized imidoyl chlorides, companies can secure a more stable sourcing strategy.
- Cost Reduction in Manufacturing: The economic implications of switching to this metal-free protocol are profound. By eliminating the need for copper catalysts, manufacturers remove the entire cost center associated with metal removal technologies, such as activated carbon treatments or specialized scavenger resins. Additionally, the avoidance of explosive azides reduces the need for specialized blast-proof infrastructure and lowers insurance premiums associated with hazardous chemical storage. The use of cesium carbonate, while a premium base, is offset by its high efficiency and the ability to drive reactions to completion with minimal excess, reducing waste disposal costs. Furthermore, the simplified workup procedure, often requiring only filtration and chromatography, reduces labor hours and solvent consumption per kilogram of product produced.
- Enhanced Supply Chain Reliability: Supply chain continuity is heavily dependent on the availability of starting materials. The precursors for this reaction, specifically the trifluoroethylimidoyl chlorides and diazo compounds, are derived from abundant aromatic amines and acid chlorides, which are produced on a massive global scale. This contrasts sharply with specialized trifluoromethylating reagents or unstable azides that may have limited suppliers and long lead times. By utilizing a chemistry based on bulk commodities, procurement teams can negotiate better pricing and ensure consistent delivery schedules. The robustness of the reaction also means that slight variations in raw material quality are less likely to cause batch failures, further stabilizing the supply of critical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this methodology is inherently scalable. The reaction exotherm is manageable, and the conditions (50-70°C) are easily maintained in large reactors without requiring cryogenic cooling or extreme heating. From an environmental standpoint, the absence of heavy metals significantly reduces the toxicity of the waste stream, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The atom economy is favorable, and the ability to recycle solvents like acetonitrile further enhances the green chemistry profile of the process, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this synthetic route.
Q: Why is this new synthesis method safer than traditional copper-catalyzed routes?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive, posing significant safety risks during scale-up. This patented method utilizes stable diazo compounds and imidoyl chlorides, eliminating the need for hazardous azide reagents and heavy metal catalysts.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent data indicates that using cesium carbonate (2.0 equivalents) in acetonitrile at 60°C for 12 hours provides optimal results, with yields reaching up to 91% for specific substrates like ethyl esters.
Q: Can this process be scaled for industrial production?
A: Yes, the methodology is designed for scalability. The use of inexpensive, commercially available starting materials and mild reaction conditions facilitates easy expansion from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN113121462B for the production of advanced heterocyclic building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest international standards for pharmaceutical applications. We understand that consistency is key, and our process engineering team is dedicated to optimizing these metal-free routes for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your overall manufacturing costs.
