Scalable Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for fluorinated heterocycles, driven by the unique metabolic stability and lipophilicity that trifluoromethyl groups impart to bioactive molecules. Patent CN113121462A introduces a significant advancement in this domain by disclosing a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology addresses critical bottlenecks in traditional heterocycle synthesis by utilizing a base-promoted cyclization strategy that bypasses the need for hazardous azides or expensive transition metal catalysts. For R&D directors and procurement specialists alike, this represents a pivotal shift towards safer, more cost-efficient manufacturing of key pharmaceutical intermediates. The method leverages readily available starting materials, specifically trifluoroacetimidoyl chlorides and diazo compounds, reacting them under mild thermal conditions to achieve high efficiency. This report analyzes the technical merits and commercial implications of this innovation, highlighting its potential to streamline supply chains for complex nitrogen-containing scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
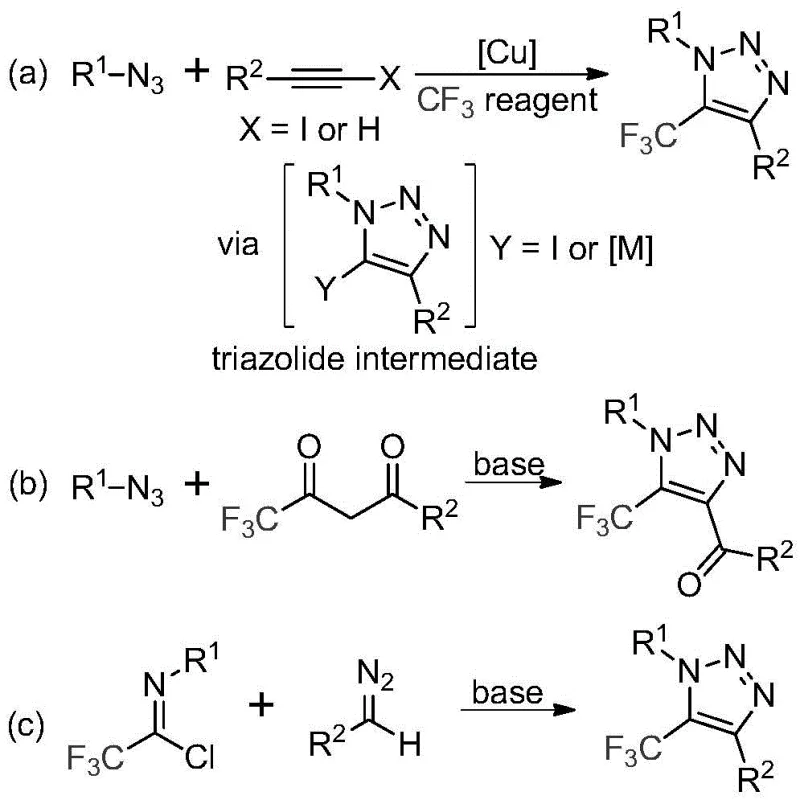
Historically, the construction of trifluoromethyl-substituted triazoles has relied heavily on copper-catalyzed azide-alkyne cycloadditions (CuAAC) followed by subsequent trifluoromethylation steps, or 1,3-dipolar cycloadditions involving organic azides. As illustrated in the reaction schemes below, these conventional pathways often necessitate the handling of toxic and potentially explosive organic azides, posing severe safety risks during scale-up. Furthermore, the reliance on copper catalysts introduces significant downstream processing challenges, particularly the rigorous requirement to remove trace heavy metals to meet stringent pharmaceutical purity standards. These legacy methods frequently suffer from narrow substrate scope, harsh reaction conditions, and the generation of complex impurity profiles that complicate purification. The economic burden of ligand systems and metal scavengers further erodes the cost-effectiveness of these traditional routes, making them less attractive for high-volume commercial production of API intermediates.
The Novel Approach
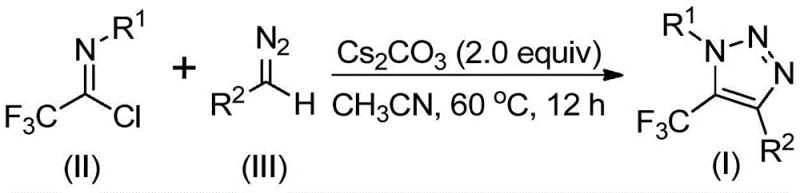
In stark contrast to the hazardous and metal-dependent legacy protocols, the methodology described in CN113121462A offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic logic. The core innovation lies in the direct coupling of trifluoroacetimidoyl chloride with diazo compounds in the presence of a mild inorganic base, specifically cesium carbonate. This approach completely eliminates the need for explosive azide reagents and toxic transition metals, thereby drastically reducing safety hazards and environmental impact. The reaction proceeds efficiently in common aprotic solvents like acetonitrile at moderate temperatures ranging from 50°C to 70°C. By avoiding heavy metal catalysts, the process inherently produces a cleaner crude reaction mixture, which translates to simplified workup procedures and higher overall yields. This novel pathway not only enhances operational safety but also aligns perfectly with green chemistry principles, offering a sustainable solution for the manufacture of high-purity pharmaceutical intermediates.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this transformation relies on a sequential intermolecular nucleophilic addition-elimination followed by an intramolecular cyclization event. Initially, the base activates the diazo compound, facilitating a nucleophilic attack on the electrophilic carbon of the trifluoroacetimidoyl chloride. This step forms a key intermediate through the displacement of the chloride leaving group. Subsequently, the system undergoes a 5-endo-dig cyclization, where the internal nitrogen nucleophile attacks the activated nitrile or imine functionality to close the triazole ring. This cascade occurs smoothly under the promoted basic conditions without requiring external activation by Lewis acids or transition metals. The tolerance of this mechanism towards various functional groups on both the aryl ring of the imidoyl chloride and the substituent of the diazo compound is remarkable, allowing for the synthesis of diverse derivatives including esters, ketones, and phosphonates. Such mechanistic robustness ensures that the process remains reliable even when scaling to larger batches, providing consistent results across different substrate classes.
From an impurity control perspective, the absence of transition metals eliminates a major source of contamination that typically plagues heterocyclic synthesis. In traditional copper-catalyzed routes, metal-ligand complexes can persist through workup, requiring specialized scavenging resins that add cost and waste. Here, the primary byproducts are inorganic salts derived from the base and the leaving group, which are easily removed during the aqueous workup or filtration steps. The mild thermal conditions (50-70°C) further suppress thermal decomposition pathways and polymerization side reactions that are common with highly reactive diazo species at elevated temperatures. This results in a superior impurity profile, facilitating easier purification via standard column chromatography or crystallization. For quality control teams, this means a more predictable and manageable impurity landscape, ensuring that the final API intermediate meets rigorous regulatory specifications with minimal effort.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this patented method makes it highly accessible for laboratory and pilot plant settings. The procedure involves combining the trifluoroacetimidoyl chloride, the specific diazo compound, and cesium carbonate in an organic solvent such as acetonitrile. Molecular sieves are optionally added to maintain anhydrous conditions, which can be critical for the stability of the diazo reagent. The mixture is then heated to a controlled temperature between 50°C and 70°C for a duration of 8 to 16 hours. Upon completion, the reaction mixture is filtered to remove solids, and the filtrate is concentrated. The crude product is typically purified using standard silica gel column chromatography to afford the target triazole in high purity. This straightforward protocol minimizes the need for specialized equipment or hazardous handling procedures, making it an ideal candidate for rapid process development and scale-up activities.
- Combine cesium carbonate, molecular sieves, trifluoroacetimidoyl chloride, and the diazo compound in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 50°C and 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 5-trifluoromethyl substituted triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive transition metal catalysts and their associated ligands represents a direct reduction in raw material costs. Furthermore, the removal of heavy metal scavenging steps from the downstream processing workflow significantly reduces the consumption of auxiliary materials and waste disposal costs. The use of commodity chemicals like cesium carbonate and acetonitrile ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or regulated reagents. This process stability allows for more accurate forecasting and inventory management, crucial for maintaining continuous production schedules in a volatile market environment.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the exclusion of precious metal catalysts, which are subject to significant price volatility and supply constraints. By utilizing a base-promoted organocatalytic approach, manufacturers can avoid the substantial capital expenditure associated with metal recovery systems and the recurring cost of high-purity metal salts. Additionally, the simplified purification train reduces solvent consumption and energy usage during the isolation phase. These factors collectively contribute to a lower cost of goods sold (COGS), enabling more competitive pricing for the final pharmaceutical intermediates while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroacetimidoyl chlorides and diazo compounds, are derived from widely available industrial feedstocks. This broad availability reduces dependency on single-source suppliers for exotic reagents, thereby de-risking the supply chain against geopolitical disruptions or logistical bottlenecks. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor fluctuations in temperature or mixing efficiency. This reliability ensures consistent delivery timelines to downstream customers, fostering stronger long-term partnerships and trust within the global pharmaceutical supply network.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a distinct advantage by avoiding the generation of heavy metal waste streams. The absence of copper or other transition metals simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne-scale production. This scalability, combined with a greener chemical profile, positions manufacturers to meet increasingly stringent environmental regulations without compromising on production volume or efficiency, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require toxic azide reagents?
A: No, unlike conventional copper-catalyzed methods that often rely on explosive organic azides, this patented process utilizes stable diazo compounds and trifluoroacetimidoyl chlorides, significantly enhancing operational safety.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions and provides the possibility for industrial scale production due to its mild conditions and simple post-treatment procedures.
Q: What represents the primary cost advantage of this route?
A: The primary cost advantage stems from the elimination of expensive transition metal catalysts and ligands, as well as the avoidance of complex purification steps required to remove heavy metal residues from the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN113121462A can be seamlessly transferred to industrial manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the most stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to explore the full potential of this metal-free triazole synthesis for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how this route can enhance your bottom line. Please contact us today to request specific COA data for our available triazole derivatives and to discuss detailed route feasibility assessments for your custom synthesis needs. Let us help you accelerate your path to market with reliable, high-quality chemical solutions.
