Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking efficient, safe, and scalable routes to construct privileged heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, base-promoted cyclization strategy. By utilizing readily available trifluoroethylimidoyl chloride and diazo compounds as starting materials, this invention circumvents the inherent safety risks associated with explosive organic azides and the regulatory burdens of heavy metal contamination. For R&D directors and procurement managers alike, this methodology offers a compelling value proposition: it simplifies the synthetic pathway while maintaining high reaction efficiency and broad substrate scope, thereby facilitating the rapid development of next-generation therapeutic agents and crop protection chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic variants involving trifluoromethyl ketones. As illustrated in the general reaction schemes below, these conventional pathways present substantial drawbacks for industrial application. The primary concern is the reliance on organic azides, which are notoriously toxic and possess high potential for explosivity, posing severe safety hazards during scale-up operations. Furthermore, the use of copper catalysts necessitates rigorous downstream purification processes to remove trace metal residues, which is a critical requirement for Active Pharmaceutical Ingredients (APIs) but adds significant time and cost to the manufacturing process. Additionally, the requirement for specialized trifluoromethylating reagents in some variations can drive up raw material costs and complicate supply chain logistics, making these traditional methods less attractive for cost-sensitive commercial production.
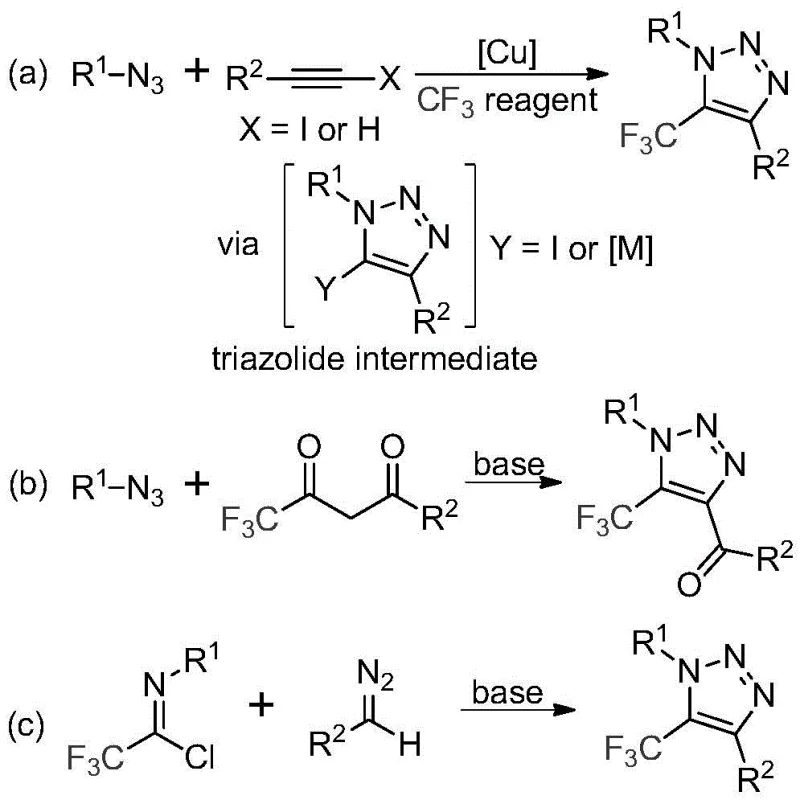
The Novel Approach
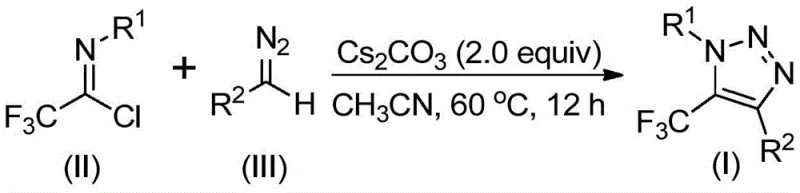
In stark contrast to the hazardous and complex legacy methods, the technology described in patent CN113121462B introduces a streamlined, metal-free protocol that fundamentally alters the synthetic landscape for these valuable intermediates. The core innovation lies in the direct coupling of trifluoroethylimidoyl chloride with diazo compounds under mild basic conditions, completely bypassing the need for unstable azide intermediates. This approach not only enhances operational safety by eliminating explosive reagents but also drastically simplifies the reaction setup. The process operates effectively in common aprotic solvents like acetonitrile at moderate temperatures, typically around 60°C, which is highly energy-efficient compared to high-temperature alternatives. By removing the dependency on precious metal catalysts, this novel route inherently reduces the environmental footprint and lowers the barrier to entry for large-scale manufacturing, positioning it as a superior choice for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
Mechanistic Insights into Base-Promoted Cyclization
From a mechanistic perspective, this transformation is a fascinating example of base-mediated cascade reactivity that achieves complex ring construction in a single pot. The reaction is initiated by the deprotonation of the diazo compound by the cesium carbonate base, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular nucleophilic addition-elimination sequence is critical for establishing the initial carbon-carbon bond connectivity. Following this addition, the intermediate undergoes a spontaneous intramolecular 5-endo-dig cyclization, driven by the nucleophilic attack of the nitrogen atom onto the activated triple bond or equivalent electrophilic center within the transient structure. This cyclization step is highly regioselective, ensuring the formation of the desired 1,4-disubstituted 1,2,3-triazole scaffold with the trifluoromethyl group precisely positioned at the 5-position. The elegance of this mechanism lies in its atom economy and the avoidance of external oxidants or reductants, relying solely on the intrinsic reactivity of the substrates activated by a simple inorganic base.
Regarding impurity control and product purity, the mild nature of the cesium carbonate promoter plays a pivotal role in minimizing side reactions. Unlike strong bases that might induce decomposition of the sensitive diazo functionality or hydrolysis of the imidoyl chloride, cesium carbonate provides a buffered basic environment that favors the desired cyclization pathway. The absence of transition metals eliminates the risk of metal-complexed byproducts, which are often difficult to separate and can catalyze degradation during storage. Furthermore, the reaction conditions are compatible with a wide range of functional groups, including halogens and esters, without causing unwanted dehalogenation or transesterification. This high level of chemoselectivity ensures that the crude reaction mixture is relatively clean, simplifying the final purification via standard silica gel chromatography and resulting in high-purity 1,2,3-triazole derivatives that meet stringent quality specifications required for downstream biological testing.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory discovery teams and pilot plant engineers. The protocol involves charging a reaction vessel with the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in anhydrous acetonitrile. The mixture is then heated to a moderate temperature, typically between 50°C and 70°C, and stirred for a duration of 8 to 16 hours to ensure full conversion. The workup procedure is straightforward, involving simple filtration to remove inorganic salts followed by concentration and purification. This simplicity stands in contrast to multi-step sequences requiring cryogenic conditions or inert gas manifolds for azide handling. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in anhydrous acetonitrile within a Schlenk tube under inert atmosphere.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion of the starting materials.
- Filter the mixture, concentrate the filtrate, and purify the crude residue via silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of hazardous azides and heavy metal catalysts directly addresses two of the most significant cost drivers in fine chemical manufacturing: safety compliance and purification overhead. By shifting to a safer reagent profile, facilities can reduce insurance premiums and minimize the need for specialized blast-proof infrastructure, leading to substantial capital expenditure savings. Moreover, the removal of copper catalysts obviates the need for expensive scavenger resins or complex extraction protocols designed to lower metal content to ppm levels, thereby shortening the overall production cycle time and increasing throughput capacity.
- Cost Reduction in Manufacturing: The economic impact of this metal-free process is profound, primarily driven by the substitution of expensive transition metal catalysts with inexpensive, commodity-grade cesium carbonate. Traditional copper-catalyzed routes often require ligand optimization and rigorous metal removal steps, which add significant material and labor costs. In this new protocol, the reagents are not only cheaper but also more stable, reducing waste associated with reagent degradation and storage. The simplified workup procedure further contributes to cost efficiency by reducing solvent consumption and processing time, allowing for a leaner manufacturing operation that maximizes margin potential in competitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, commercially available starting materials. Unlike specialized trifluoromethylating reagents or unstable azides that may have limited suppliers and long lead times, trifluoroethylimidoyl chlorides and diazo compounds are part of established supply chains with multiple global sources. This diversification of supply reduces the risk of production stoppages due to raw material shortages. Furthermore, the stability of these precursors allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to hedge against market volatility and ensure continuous availability of critical intermediates for their clients.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental liabilities, but this base-promoted cyclization is inherently scalable due to its mild thermal profile and lack of exothermic hazards associated with azide decomposition. The process generates minimal hazardous waste, aligning with increasingly strict environmental regulations and corporate sustainability goals. The absence of heavy metals simplifies wastewater treatment and disposal, reducing the environmental compliance burden. This 'green' profile not only mitigates regulatory risk but also enhances the brand value of the final product, appealing to end-users who prioritize sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Why is this new synthesis method safer than traditional copper-catalyzed routes?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive, alongside heavy metal catalysts like copper that require complex removal. This novel protocol utilizes stable diazo compounds and imidoyl chlorides under metal-free conditions, significantly enhancing operational safety and reducing hazardous waste.
Q: What are the key advantages for large-scale manufacturing of these intermediates?
A: The process eliminates the need for expensive transition metal catalysts and the subsequent purification steps required to meet strict ppm limits for residual metals in APIs. Furthermore, the use of commercially available, stable starting materials ensures a robust supply chain suitable for kilogram-to-ton scale production.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and alkoxy groups on both the aromatic amine and the diazo components, allowing for the rapid generation of diverse chemical libraries for drug screening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value fluorinated heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, maintaining stringent purity specifications through our rigorous QC labs to guarantee that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs, ensuring a secure and efficient supply of these critical building blocks.
