Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking multi-component strategy for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and trifluoromethyl-imidazole motifs. This innovation addresses critical challenges in modern organic synthesis by replacing hazardous gaseous reagents with safer liquid alternatives while maintaining high reaction efficiency. The disclosed method leverages a palladium-catalyzed cascade reaction that operates under remarkably mild conditions, typically at 30°C, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. For R&D directors and process chemists, this represents a significant leap forward in accessing diversified chemical space without the logistical burdens associated with high-pressure carbonylation equipment.
Furthermore, the structural versatility of the resulting products, characterized by the general Formula (I), offers immense potential for structure-activity relationship (SAR) studies in drug discovery programs. The ability to incorporate various substituents on the aromatic rings allows medicinal chemists to fine-tune the physicochemical properties of lead compounds. By integrating a trifluoromethyl group, these molecules gain enhanced metabolic stability and lipophilicity, traits highly desirable in candidate selection. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such advanced synthetic routes is crucial for ensuring a steady supply of high-quality building blocks for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbonyl-bridged heterocyclic systems has relied heavily on direct coupling reactions or oxidative cyclizations that often suffer from poor atom economy and harsh reaction conditions. A major bottleneck in conventional carbonylation strategies is the dependence on carbon monoxide (CO) gas, which is not only highly toxic but also requires specialized high-pressure reactors and rigorous safety protocols to handle effectively. These infrastructure requirements create significant barriers to entry for smaller laboratories and increase the capital expenditure for large-scale production facilities. Additionally, many existing methods struggle with substrate compatibility, frequently failing when sensitive functional groups are present on the starting materials, thereby limiting the diversity of the chemical libraries that can be generated. The need for stoichiometric oxidants or expensive coupling reagents in alternative pathways further exacerbates cost and waste disposal issues, making these processes less attractive for green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology described in CN115353511A utilizes a transition metal palladium-catalyzed carbonylation cascade that elegantly circumvents the need for external CO gas. Instead, it employs a mixture of formic acid and acetic anhydride as an efficient, in-situ source of carbon monoxide, dramatically improving the safety profile and operational simplicity of the reaction. This approach enables a one-pot, multi-component synthesis where three distinct building blocks converge to form the complex biheterocyclic core in a single step. The reaction proceeds smoothly at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating exceptional functional group tolerance. 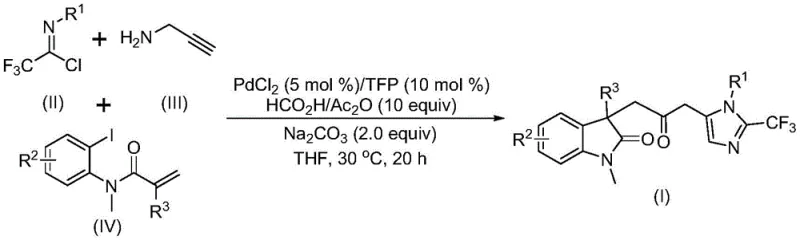
This novel route not only simplifies the synthetic workflow by eliminating multiple isolation steps but also enhances the overall yield and purity of the final products. The use of cheap and commercially available reagents, such as propargylamine and acrylamide derivatives, ensures that the process remains economically viable for industrial scale-up. By shifting from hazardous gas handling to a liquid-phase reagent system, this technology offers a pragmatic solution for cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with the industry's drive towards safer and more sustainable chemical processes.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated sequence of organometallic steps initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This generates a reactive aryl-palladium intermediate which subsequently undergoes an intramolecular Heck-type reaction to form a divalent alkyl-palladium species. Crucially, the carbon monoxide required for the carbonyl bridge is released from the formic acid and acetic anhydride mixture and inserts into the palladium-carbon bond, yielding an acyl-palladium intermediate. Concurrently, a base-promoted intermolecular reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine compound in situ. This amidine species then undergoes isomerization and subsequent nucleophilic attack on the activated acyl-palladium complex.
The final stage involves an intramolecular cyclization catalyzed by the acyl-palladium intermediate, which closes the ring to form the stable carbonyl-bridged biheterocyclic structure shown in Formula (I). 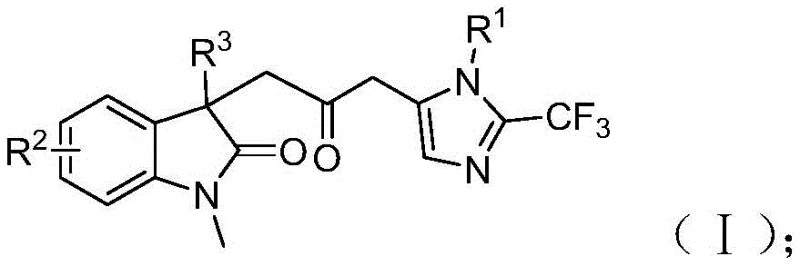 This intricate cascade highlights the precise control exerted by the palladium catalyst and the TFP ligand system, ensuring high regioselectivity and minimizing the formation of side products. For quality control teams, understanding this mechanism is vital for impurity profiling, as it identifies potential byproducts arising from incomplete cyclization or alternative insertion pathways. The robustness of this catalytic cycle allows for the accommodation of diverse electronic and steric environments on the substrates, making it a versatile tool for synthesizing a wide array of analogues required for comprehensive biological screening.
This intricate cascade highlights the precise control exerted by the palladium catalyst and the TFP ligand system, ensuring high regioselectivity and minimizing the formation of side products. For quality control teams, understanding this mechanism is vital for impurity profiling, as it identifies potential byproducts arising from incomplete cyclization or alternative insertion pathways. The robustness of this catalytic cycle allows for the accommodation of diverse electronic and steric environments on the substrates, making it a versatile tool for synthesizing a wide array of analogues required for comprehensive biological screening.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory glassware and avoiding exotic conditions. The protocol involves mixing the palladium catalyst, ligand, base, and the CO-source mixture in a solvent, followed by the addition of the three key organic substrates. The reaction is allowed to stir at ambient or slightly elevated temperatures for a defined period, after which simple filtration and chromatographic purification yield the target molecule. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yields across different batches.
- Combine palladium chloride, TFP ligand, sodium carbonate, and the CO source mixture (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, then filter and purify the crude mixture via column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented method offers substantial strategic advantages by decoupling production from volatile supply chains associated with specialized gases and high-pressure equipment. The reliance on commodity chemicals like formic acid, acetic anhydride, and simple amines ensures a stable and predictable raw material supply, mitigating risks of production stoppages due to reagent shortages. Furthermore, the elimination of toxic carbon monoxide gas removes the need for expensive safety infrastructure and specialized training for operators, leading to significant overhead cost reductions. The mild reaction conditions also translate to lower energy consumption, as there is no need for extensive heating or cooling systems, contributing to a smaller carbon footprint and reduced utility costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The substitution of hazardous carbon monoxide gas with a liquid mixture of formic acid and acetic anhydride eliminates the capital expenditure required for high-pressure autoclaves and gas handling systems. This shift significantly lowers the barrier to entry for production and reduces the ongoing maintenance and inspection costs associated with pressure vessels. Additionally, the use of inexpensive and widely available starting materials like propargylamine ensures that the cost of goods sold (COGS) remains competitive, allowing for better margin management in the final API pricing.
- Enhanced Supply Chain Reliability: By utilizing a multi-component reaction that tolerates a wide range of functional groups, manufacturers can source substrates from multiple global suppliers without stringent purity specifications that often bottleneck supply chains. The robustness of the reaction means that minor variations in raw material quality do not necessarily compromise the final yield, providing a buffer against supply chain disruptions. This flexibility ensures consistent delivery schedules for downstream clients, fostering stronger long-term partnerships and reliability in the global pharmaceutical market.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable to gram-level and beyond without loss of efficiency, indicating strong potential for ton-scale commercial production. The absence of toxic gas emissions simplifies environmental compliance and waste treatment procedures, as the primary byproducts are manageable liquids rather than hazardous gases. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's capabilities and limitations for potential adopters.
Q: What are the safety advantages of this carbonylation method compared to traditional techniques?
A: Unlike conventional carbonylation reactions that require toxic and high-pressure carbon monoxide gas, this patented method utilizes a formic acid and acetic anhydride mixture as a safe, in-situ CO source, significantly reducing operational hazards and equipment costs.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating R1 groups such as alkyl, substituted phenyl (with methyl, methoxy, halogen, nitro, or trifluoromethyl), and R3 groups including alkyl, phenyl, or benzyl, allowing for diverse library generation.
Q: Why are carbonyl-bridged biheterocycles valuable in drug discovery?
A: These scaffolds combine indolinone and imidazole motifs, both of which are privileged structures in medicinal chemistry known for broad-spectrum biological activities, making them highly attractive intermediates for developing new therapeutic agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN115353511A for accelerating drug discovery pipelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We understand that time-to-market is critical, and our optimized processes are designed to deliver high-purity intermediates with minimal lead times.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
