Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
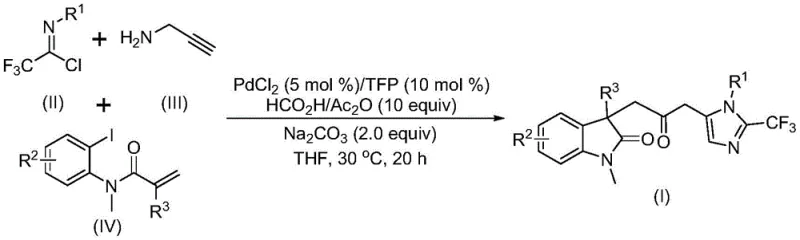
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the synthesis of indolinone-imidazole hybrids which are prevalent in medicinal chemistry. This innovation addresses the long-standing challenge of efficiently constructing carbonyl-bridged systems without relying on hazardous gaseous reagents. By leveraging a transition metal palladium-catalyzed carbonylation cascade reaction, the disclosed technology enables the one-pot assembly of diverse substituted double heterocyclic compounds containing trifluoromethyl and carbonyl functionalities. The significance of this development lies not only in its chemical elegance but also in its potential for industrial application, as the process has been successfully expanded to gram-scale reactions, demonstrating robust scalability for commercial production needs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biheterocyclic compounds has relied on strategies that often suffer from significant operational and economic drawbacks. Traditional approaches typically involve the direct coupling of two pre-formed heterocycle substrates, which requires the separate synthesis of each ring system, leading to increased step counts and lower overall yields. Alternatively, oxidative cyclization reactions using activated methyl-substituted heterocycles often necessitate stoichiometric amounts of expensive or toxic oxidants, generating substantial waste streams that complicate downstream processing. Furthermore, conventional carbonylation reactions frequently depend on the use of carbon monoxide gas, which poses severe safety risks due to its toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that drive up capital expenditure. These limitations collectively hinder the rapid exploration of chemical space and increase the cost of goods for potential drug candidates containing these privileged structures.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a multicomponent reaction strategy that merges simplicity with high efficiency. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the process bypasses the need for pre-functionalized heterocycles. The reaction proceeds under remarkably mild conditions, specifically at a temperature of 30°C, which minimizes energy consumption and reduces the risk of thermal decomposition of sensitive intermediates. Crucially, the method replaces toxic carbon monoxide gas with a safer liquid mixture of formic acid and acetic anhydride, which acts as an effective carbon monoxide surrogate. This shift not only enhances laboratory safety but also simplifies the engineering requirements for scaling the process to manufacturing levels, making it an attractive option for reliable pharmaceutical intermediate suppliers looking to optimize their production pipelines.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated sequence of organometallic steps that highlights the versatility of palladium catalysis in modern organic synthesis. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction that constructs the indolinone core, forming a divalent alkyl palladium species. Subsequently, the carbon monoxide released in situ from the formic acid and acetic anhydride mixture inserts into the palladium-carbon bond, yielding an acyl palladium intermediate. This acyl species is then poised for the final cyclization event, where it activates the trifluoroacetamidine compound—formed earlier via base-promoted coupling of the imidoyl chloride and propargylamine—to close the imidazole ring. This intricate cascade allows for the simultaneous formation of multiple carbon-carbon and carbon-heteroatom bonds in a single operational step.
From an impurity control perspective, the use of specific ligands such as tri-furyl phosphine (TFP) plays a critical role in stabilizing the active catalytic species and suppressing side reactions. The mild reaction temperature of 30°C further contributes to a cleaner reaction profile by minimizing thermal degradation pathways that often lead to complex impurity profiles in high-temperature processes. The compatibility with various functional groups, including halogens and electron-withdrawing substituents, suggests that the catalytic cycle is robust against electronic variations in the substrate. This mechanistic understanding provides a strong foundation for process optimization, ensuring that the synthesis of high-purity OLED material precursors or pharmaceutical intermediates can be achieved with consistent quality and minimal byproduct formation, thereby reducing the burden on purification units.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The protocol involves mixing the palladium catalyst, ligand, base, and carbon monoxide surrogate in an aprotic organic solvent such as tetrahydrofuran (THF), which has been identified as the optimal medium for solubilizing all components and promoting high conversion rates. The substrates are then introduced, and the mixture is stirred for a duration of 12 to 20 hours. Detailed standardized synthesis steps follow below to guide the practical implementation of this technology.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and a carbon monoxide surrogate mixture (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel.
- Stir the mixture at 30°C for 12 to 20 hours, followed by filtration and silica gel column chromatography to isolate the pure biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for raw materials. Since the method utilizes commercially available and inexpensive starting materials like propargylamine and acrylamide, there is no reliance on exotic or custom-synthesized building blocks that often carry long lead times and high price volatility. This accessibility ensures a stable supply of inputs, which is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream clients. Furthermore, the elimination of toxic gas handling reduces the regulatory burden and insurance costs associated with the facility, contributing to overall operational efficiency.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of hazardous carbon monoxide gas with liquid reagents, which removes the need for specialized high-pressure reactors and gas scrubbing systems. This capital expenditure saving is complemented by the reduced energy demand, as the reaction operates effectively at near-ambient temperatures rather than requiring intense heating. Additionally, the one-pot nature of the reaction consolidates multiple synthetic steps into a single vessel, drastically reducing solvent usage, labor hours, and waste disposal costs. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing without compromising on the quality or complexity of the final product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. The tolerance for diverse functional groups means that a single platform technology can be adapted to produce a wide library of analogues by simply swapping out commercially available substrates. This flexibility allows manufacturers to respond rapidly to changing market demands or specific client requests for structural variants. Moreover, the use of stable, non-gaseous reagents simplifies logistics and storage, reducing the risk of supply disruptions caused by transportation restrictions on hazardous materials. This reliability makes the process an ideal candidate for establishing long-term partnerships with global buyers seeking a dependable source of complex heterocyclic scaffolds.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this methodology has already demonstrated feasibility at the gram scale, indicating a clear path toward kilogram and tonne-level production. The avoidance of toxic gases and the use of standard organic solvents align well with modern environmental, health, and safety (EHS) standards, facilitating easier permitting and compliance. The simplified workup procedure, involving filtration and standard column chromatography, further supports scalability by minimizing the complexity of downstream processing. This combination of scalability and environmental friendliness positions the technology as a sustainable solution for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction parameters and substrate scope. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing development pipelines.
Q: What are the advantages of using formic acid and acetic anhydride over carbon monoxide gas?
A: Using a formic acid and acetic anhydride mixture serves as a safe and convenient in situ source of carbon monoxide. This eliminates the need for handling toxic, high-pressure CO gas cylinders, significantly improving operational safety and reducing infrastructure costs for industrial scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed cascade reaction?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on the aryl rings, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, trifluoromethyl, and nitro groups, allowing for diverse library synthesis.
Q: Why is this method considered superior for synthesizing indolinone-imidazole hybrids?
A: Traditional methods often require harsh conditions or multiple steps. This one-pot multicomponent approach constructs multiple chemical bonds simultaneously at a mild temperature of 30°C, offering higher atom economy, simpler operation, and easier purification compared to stepwise coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable commercial supplies. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our clients receive materials that meet the highest industry standards.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this technology can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market through superior chemical manufacturing solutions.
