Advanced Heterogeneous Catalysis for Scalable Diphenyl Sulfide Production and Commercial Intermediates
The landscape of carbon-sulfur bond formation is undergoing a significant transformation driven by the urgent need for greener, safer, and more economically viable synthetic routes in the fine chemical industry. Patent CN116143673A, published in May 2023, introduces a groundbreaking methodology for the synthesis of diphenyl sulfide compounds utilizing a heterogeneous nickel-based composite metal oxide catalyst. This innovation addresses critical bottlenecks in traditional C-S coupling reactions by replacing hazardous thiol reagents with benign elemental sulfur and substituting expensive, difficult-to-separate homogeneous catalysts with robust, recyclable heterogeneous systems. For R&D directors and process chemists, this represents a paradigm shift towards sustainable manufacturing, offering a pathway to produce high-purity pharmaceutical intermediates with significantly reduced environmental footprints and operational complexities. The core of this technology lies in the synergistic interaction between nickel and secondary transition metals like copper or iron, which facilitates efficient oxidative addition and reductive elimination cycles under remarkably mild thermal conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of diaryl sulfide scaffolds has relied heavily on the nucleophilic substitution of aryl halides with thiols or the cross-coupling of aryl halides with thiols using palladium or copper catalysts. These conventional methodologies suffer from severe inherent drawbacks that hinder their applicability in large-scale commercial production. Primarily, thiols are notoriously volatile, possessing extremely unpleasant and persistent odors that pose significant occupational health and safety risks, requiring specialized containment infrastructure and increasing facility operating costs. Furthermore, thiols have a strong affinity for transition metal centers, often leading to catalyst poisoning and deactivation, which necessitates the use of excessive catalyst loading or expensive ligand systems to maintain activity. Additionally, many traditional protocols require harsh reaction conditions, including high temperatures and strong bases, which are incompatible with sensitive functional groups often present in complex drug molecules, thereby limiting the substrate scope and requiring additional protection-deprotection steps that reduce overall atom economy.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a heterogeneous Ni-Cu composite metal oxide catalyst to drive the coupling of aryl halides directly with elemental sulfur, completely bypassing the need for malodorous thiols. This method operates under mild conditions, typically between 60°C and 80°C, which preserves the integrity of sensitive functional groups and reduces energy consumption. The use of elemental sulfur as the sulfur source not only enhances safety but also drastically lowers raw material costs compared to specialized thiol reagents. Crucially, the heterogeneous nature of the catalyst allows for straightforward separation via simple filtration, eliminating the need for complex purification steps to remove trace metal contaminants, a critical requirement for pharmaceutical grade intermediates. This streamlined workflow significantly simplifies the downstream processing, reducing solvent usage and waste generation while ensuring the final product meets stringent purity specifications required by regulatory bodies.
The mechanistic underpinnings of this Ni-Cu composite oxide catalyzed reaction offer profound insights into how bimetallic synergy can enhance catalytic performance in C-S bond formation. The catalyst, typically formulated as NiyMzOx where M is Cu, Fe, or Co, functions through a surface-mediated mechanism where the nickel sites likely facilitate the oxidative addition of the aryl halide, while the copper or secondary metal sites assist in activating the elemental sulfur species. This cooperative effect lowers the activation energy barrier for the rate-determining steps, allowing the reaction to proceed efficiently at lower temperatures compared to monometallic systems. The basic environment provided by additives like potassium carbonate or potassium hydroxide plays a pivotal role in generating the active sulfur nucleophile in situ, which then attacks the activated aryl-metal intermediate. The robustness of the oxide lattice structure ensures that the active metal species remain anchored, preventing leaching into the product stream and maintaining catalytic activity over multiple cycles.
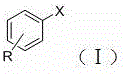
Impurity control is another critical aspect where this heterogeneous system excels, particularly for applications in the synthesis of high-purity pharmaceutical intermediates. In homogeneous catalysis, residual metal contamination is a persistent challenge that often requires scavenging resins or extensive chromatography, adding cost and time to the process. With the Ni-Cu oxide system, the solid catalyst is physically separated from the reaction mixture by filtration, inherently minimizing the risk of metal leaching. Furthermore, the mild reaction conditions suppress side reactions such as homocoupling of the aryl halide or over-sulfurization, which are common pitfalls in aggressive thermal protocols. The selectivity of the catalyst towards the desired C-S bond formation ensures a clean reaction profile, simplifying the purification process and resulting in higher isolated yields of the target diphenyl sulfide compound, often exceeding 85% even with complex substrates bearing electron-withdrawing or electron-donating groups.
How to Synthesize Diphenyl Sulfide Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for process development teams aiming to transition from benchtop discovery to pilot plant production. The protocol involves a straightforward one-pot procedure where the aryl halide, elemental sulfur, catalyst, and base are combined in a polar aprotic solvent such as DMSO. The reaction mixture is then heated to the optimal temperature range and stirred until conversion is complete, monitored typically by HPLC or TLC. Upon completion, the workup is exceptionally simple, involving cooling, filtration of the catalyst, and standard aqueous extraction, avoiding the need for quenching hazardous reagents or handling pyrophoric materials. This operational simplicity translates directly into reduced labor costs and improved batch-to-batch consistency, key metrics for any reliable pharmaceutical intermediate supplier.
- Prepare the reaction mixture by adding phenyl halide, elemental sulfur source, Ni-Cu composite oxide catalyst, and inorganic base into a polar organic solvent such as DMSO.
- Heat the reaction mixture to a temperature between 60°C and 80°C under magnetic stirring for a duration of 16 to 24 hours to ensure complete conversion.
- Filter the solid heterogeneous catalyst for recycling, extract the product with ethyl acetate, and purify via column chromatography to obtain high-purity diphenyl sulfide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this heterogeneous catalytic technology offers substantial strategic advantages that extend beyond mere technical feasibility. The shift away from thiols and homogeneous precious metal catalysts fundamentally alters the cost structure and risk profile of the supply chain, providing a more resilient and economical foundation for long-term production contracts. By utilizing abundant and inexpensive base metals like nickel and copper instead of scarce precious metals, manufacturers can insulate themselves from the volatility of commodity prices associated with palladium or rhodium. Moreover, the ability to recycle the catalyst multiple times without significant loss of activity dramatically reduces the effective cost per kilogram of the catalyst, contributing to significant overall cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive thiol reagents and the reduction in catalyst loading due to high turnover numbers. Since the catalyst is heterogeneous and reusable, the recurring cost of catalyst purchase is amortized over many batches, leading to a lower variable cost per unit of production. Additionally, the simplified workup procedure reduces the consumption of solvents and purification media, further driving down the operational expenditure associated with waste disposal and material handling. These factors combine to create a highly competitive cost structure that allows for more aggressive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Relying on elemental sulfur and base metal oxides mitigates supply chain risks associated with the sourcing of specialized organosulfur reagents which may have limited suppliers or long lead times. Elemental sulfur is a commodity chemical with a stable and abundant global supply, ensuring continuity of production even during market disruptions. The robustness of the catalyst also means that production schedules are less likely to be impacted by catalyst degradation or the need for frequent replenishment, allowing for more predictable planning and inventory management. This reliability is crucial for maintaining just-in-time delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of volatile, toxic thiols make this process inherently safer and easier to scale up to multi-ton production volumes. The reduced environmental impact, characterized by lower E-factors and the absence of noxious emissions, aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. This compliance advantage reduces the regulatory burden and potential liability associated with hazardous chemical handling, facilitating smoother permitting processes for new manufacturing lines and enhancing the company's reputation as a responsible green chemistry partner.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis method, we have compiled a set of frequently asked questions based on the specific data and findings presented in the patent literature. These answers provide clarity on the operational parameters, substrate scope, and sustainability metrics that are critical for decision-makers evaluating this technology for integration into their existing supply chains. Understanding these nuances is essential for assessing the fit of this methodology within specific project timelines and quality requirements.
Q: What are the primary advantages of using elemental sulfur over thiols in this synthesis?
A: Using elemental sulfur eliminates the safety hazards and unpleasant odors associated with volatile thiols, while also preventing catalyst deactivation often caused by thiol coordination to transition metals.
Q: Can the Ni-Cu composite oxide catalyst be reused effectively?
A: Yes, the heterogeneous nature of the catalyst allows for simple filtration and washing, enabling it to be recycled and reused for more than 10 cycles while maintaining yields above 85%.
Q: What types of substituents are tolerated on the phenyl halide substrate?
A: The method demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like chloro and cyano substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced heterogeneous catalysis in reshaping the production of critical pharmaceutical intermediates like diphenyl sulfide. Our team of expert process chemists is dedicated to leveraging innovations such as the Ni-Cu oxide catalyzed route to deliver superior value to our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of laboratory patents are fully realized in industrial practice. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications protocols to guarantee that every batch of high-purity diphenyl sulfide meets the exacting standards required for drug substance manufacturing.
We invite procurement leaders and R&D directors to collaborate with us to explore how this cost-effective and sustainable synthesis route can be tailored to your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this heterogeneous protocol for your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
