Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for nitrogen-containing heterocycles, particularly quinazolinones, due to their profound biological activities ranging from anticancer to antifungal properties. A pivotal advancement in this domain is disclosed in patent CN111675662A, which details a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology addresses critical bottlenecks in traditional synthesis by utilizing an inexpensive iron-catalyzed tandem cyclization strategy. The introduction of the trifluoromethyl group is strategically significant, as it enhances the metabolic stability, lipophilicity, and bioavailability of the final drug molecules, making them superior candidates for modern therapeutic applications. By leveraging readily available starting materials such as isatin and trifluoroethylimidoyl chloride, this method offers a streamlined approach that aligns perfectly with the demands of a reliable pharmaceutical intermediate supplier seeking to optimize production workflows.
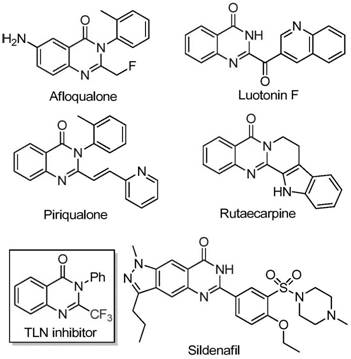
Historically, the construction of trifluoromethyl-substituted quinazolinone scaffolds has been plagued by significant technical and economic limitations that hinder widespread adoption in commercial manufacturing. Conventional literature methods predominantly rely on the cyclization of pre-functionalized synthons, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide or isatoic anhydride. These legacy processes are frequently characterized by severe reaction conditions that demand stringent safety protocols and specialized equipment, thereby inflating operational expenditures. Furthermore, the substrates required for these traditional routes are often prohibitively expensive and suffer from narrow substrate scopes, limiting the chemical diversity accessible to medicinal chemists. The low yields associated with these older methodologies not only waste valuable raw materials but also generate excessive chemical waste, creating substantial burdens for environmental compliance teams. Consequently, there has been an urgent industry-wide demand for a more sustainable and economically viable alternative that can deliver high-purity intermediates without the baggage of complex purification requirements.
In stark contrast to these cumbersome legacy techniques, the novel approach outlined in the patent data introduces a paradigm shift through the utilization of earth-abundant iron catalysis. This innovative methodology employs ferric chloride as a catalyst, which is not only drastically cheaper than noble metals but also exhibits remarkable efficiency in promoting the necessary bond formations. The reaction design cleverly bypasses the need for expensive trifluoromethyl synthons by utilizing trifluoroethylimidoyl chloride, a reagent that is both cost-effective and widely accessible in the global chemical market. The operational simplicity is another hallmark of this new route, as it proceeds under relatively mild thermal conditions compared to the extreme temperatures often required by previous art. This transition represents a major leap forward in cost reduction in API manufacturing, allowing producers to achieve higher throughput with lower input costs. The broad applicability of this system ensures that diverse analogues can be synthesized rapidly, accelerating the drug discovery timeline for R&D departments worldwide.
Mechanistic Insights into FeCl3-Catalyzed Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated sequence of events that underscores the elegance of transition metal catalysis in organic synthesis. The reaction is believed to initiate with an alkali-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a reactive trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring-closing events. The presence of the iron catalyst is crucial here, as it facilitates a decarbonylation cyclization process that would otherwise be energetically unfavorable. Following the cyclization, an isomerization step occurs to yield the thermodynamically stable 2-trifluoromethyl substituted quinazolinone core. Understanding this mechanism is vital for process chemists, as it highlights the specific role of each reagent, such as sodium hydride acting as the base to drive the initial nucleophilic attack. The precise control over these mechanistic steps ensures high selectivity, minimizing the formation of regioisomers or side products that could complicate downstream purification.
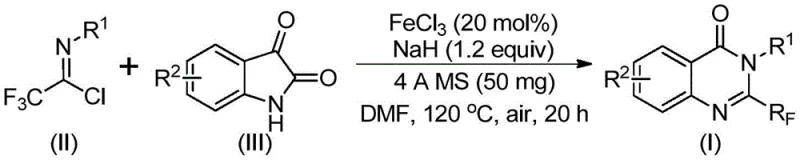
From a quality control perspective, the impurity profile generated by this iron-catalyzed system is exceptionally clean, which is a paramount concern for any R&D Director overseeing API development. The high functional group tolerance of the reaction means that sensitive moieties on the aromatic rings, such as halogens or alkoxy groups, remain intact throughout the rigorous thermal cycling. This chemoselectivity prevents the generation of complex impurity spectra that often arise from non-selective radical processes or harsh acidic conditions found in alternative methods. Moreover, the use of 4A molecular sieves in the reaction mixture plays a critical role in sequestering moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material and lead to yield loss. The result is a crude reaction mixture that is amenable to standard purification techniques like column chromatography or recrystallization, ensuring that the final product meets the stringent purity specifications required for clinical trials. This level of control over the chemical outcome provides a solid foundation for scaling the process while maintaining consistent quality batch after batch.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The protocol involves a two-stage heating process that is critical for driving the reaction to completion while managing the exothermic nature of the initial bond formation. Operators must ensure that the reaction environment is strictly anhydrous, as the presence of water can deactivate the sodium hydride and degrade the acid chloride reagent. The following guide outlines the standardized steps derived from the patent examples to ensure reproducibility and optimal performance of the catalytic system.
- Combine ferric chloride catalyst, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an aprotic organic solvent such as DMF.
- Initiate the reaction at a moderate temperature of 40°C for 8 to 10 hours to facilitate initial bond formation.
- Increase the temperature to 120°C and maintain for 18 to 20 hours to complete the tandem cyclization and decarbonylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic benefits that extend far beyond simple chemistry. The primary advantage lies in the drastic simplification of the raw material supply chain, as the key starting materials—isatin and trifluoroethylimidoyl chloride—are commodity chemicals available from multiple global vendors. This multi-sourcing capability significantly reduces the risk of supply disruptions that often plague proprietary or exotic reagents, ensuring enhanced supply chain reliability for long-term production contracts. Furthermore, the elimination of precious metal catalysts removes a major cost driver and a regulatory hurdle, as residual heavy metals in APIs require expensive removal steps to meet ICH guidelines. By switching to iron, manufacturers can achieve substantial cost savings in both material acquisition and waste treatment, directly improving the gross margin of the final active ingredient.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with ferric chloride results in a direct decrease in catalyst expenditure, which is a significant portion of the COGS for complex heterocycles. Additionally, the high atom economy of this tandem reaction minimizes the volume of solvent and reagents required per kilogram of product, further driving down operational costs. The simplified workup procedure reduces labor hours and consumable usage during purification, contributing to overall process efficiency. These cumulative effects create a leaner manufacturing model that is highly competitive in the global generic and specialty chemical markets.
- Enhanced Supply Chain Reliability: Since the starting materials are structurally simple and widely produced, lead times for raw material procurement are significantly shorter compared to specialized fluorinated building blocks. This agility allows production planners to respond more quickly to fluctuations in market demand without the fear of long backlog periods for key inputs. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, reducing the rate of batch failures and ensuring consistent output. Such stability is crucial for maintaining just-in-time inventory levels and meeting tight delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The patent explicitly notes the scalability of this method from gram to industrial scales, indicating that heat transfer and mixing issues have been considered in the reaction design. The use of DMF, a common industrial solvent, facilitates easy integration into existing manufacturing infrastructure without the need for costly reactor modifications. From an environmental standpoint, the avoidance of toxic heavy metals simplifies wastewater treatment and reduces the ecological footprint of the manufacturing site. This alignment with green chemistry principles not only lowers disposal costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of using iron catalysis over precious metals for quinazolinone synthesis?
A: Iron catalysis offers significant cost reductions by eliminating expensive palladium or ruthenium catalysts. It also simplifies downstream processing by removing the need for rigorous heavy metal scavenging steps required for pharmaceutical compliance.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is scalable from gram levels to industrial applications. The use of commercially available raw materials and standard solvents like DMF supports robust commercial scale-up of complex pharmaceutical intermediates.
Q: What is the functional group tolerance of this trifluoromethylation method?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on the aryl ring including halogens, alkyl groups, and methoxy groups without compromising reaction efficiency or yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology in advancing the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest international standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our dedicated technical team is committed to maintaining the integrity of your product throughout the entire lifecycle.
We invite you to collaborate with us to leverage this cost-effective and scalable synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline. Let us be your trusted partner in delivering high-quality chemical solutions that drive innovation and efficiency in your drug development pipeline.
