Advanced Palladium-Catalyzed Synthesis of Pyrrolone Intermediates for Commercial Scale-Up in Pharmaceutical Manufacturing
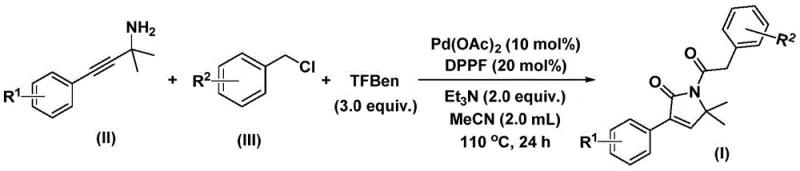
Patent CN112694430B introduces a transformative methodology for synthesizing 1,5-dihydro-2H-pyrrole-2-ketone compounds that serve as critical structural backbones in numerous bioactive molecules including the antibiotic althiomycin with significant antibacterial activity and the antidiabetic agent glimepiride used clinically as an effective hypoglycemic treatment. This innovative approach employs palladium-catalyzed bis-carbonylation chemistry to construct the pyrrolone core efficiently from readily accessible starting materials such as propargyl amines and benzyl chlorides under moderate reaction conditions of 100–120°C for 24–48 hours in acetonitrile solvent using palladium acetate and DPPF as catalytic components. The process achieves exceptional substrate compatibility across diverse functional groups while delivering high yields ranging from 70% to 92% as demonstrated through fifteen experimental examples with varying electronic properties. By eliminating multi-step synthetic sequences required by conventional methods, this patent enables substantial operational simplification while maintaining stringent purity specifications essential for pharmaceutical applications without requiring specialized high-pressure equipment typically associated with carbonylation chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the pyrrolone scaffold frequently involve complex multi-step procedures requiring cryogenic temperatures or high-pressure carbon monoxide systems that introduce significant safety hazards and infrastructure limitations for large-scale manufacturing operations. These methods typically exhibit narrow substrate scope due to poor functional group tolerance, necessitating extensive protection/deprotection strategies that increase production costs while generating substantial waste streams conflicting with modern green chemistry principles. The reliance on stoichiometric reagents rather than catalytic systems results in lower atom economy and higher environmental impact through increased solvent consumption and purification requirements. Furthermore, inconsistent product quality across different production batches often arises from complex purification processes needed to achieve pharmaceutical-grade purity specifications required by regulatory authorities such as FDA and EMA guidelines.
The Novel Approach
The patented methodology overcomes these challenges through an elegant one-pot palladium-catalyzed bis-carbonylation process that operates under ambient pressure using triethylamine as base and phenol tricarboxylate as a safe carbon monoxide surrogate. By leveraging commercially available palladium acetate with DPPF ligand system at moderate temperatures of 100–120°C in acetonitrile solvent, the reaction achieves remarkable functional group compatibility across fifteen diverse substrates including halogenated derivatives and trifluoromethyl-containing compounds while maintaining yields between 70% and 92%. This streamlined approach eliminates intermediate isolation steps through precise catalytic control while minimizing waste generation through atom-efficient transformation pathways that align with sustainable manufacturing principles required by modern pharmaceutical supply chains.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle initiates with oxidative addition of benzyl chloride to palladium(0) species generated in situ from palladium acetate reduction by triethylamine, forming a benzylpalladium intermediate that undergoes carbonyl insertion from the phenol tricarboxylate surrogate to yield an acylpalladium complex through controlled CO release mechanisms. This key intermediate then reacts with propargyl amine via nucleophilic addition followed by cyclization to form a five-membered ring palladacycle intermediate that subsequently undergoes a second carbonyl insertion event facilitated by the steric properties of DPPF ligand system before reductive elimination delivers the final pyrrolone product with precise regiocontrol across diverse substituent patterns.
Impurity control is achieved through the selective nature of this dual carbonylation mechanism where competing pathways are minimized by optimized reaction parameters including temperature control at exactly 110°C during the critical insertion steps and precise stoichiometric ratios between catalyst components (Pd(OAc)2/DPPF at 0.5/0.1 mmol ratio). The absence of transition metal residues in final products is ensured by standard post-treatment procedures involving filtration through silica gel followed by column chromatography purification using established techniques common in pharmaceutical manufacturing environments.
How to Synthesize Pyrrolone Intermediates Efficiently
This patented synthesis route represents a significant advancement in manufacturing efficiency for pharmaceutical intermediates through its streamlined one-pot methodology that eliminates multiple purification steps required by traditional approaches while maintaining exceptional product quality across diverse substrate variations encountered in complex molecule synthesis. The process leverages commercially available starting materials and standard laboratory equipment configurations to achieve high-yielding production of structurally diverse pyrrolone compounds with excellent functional group tolerance across various electronic environments including electron-donating methoxy groups and electron-withdrawing trifluoromethyl substituents without significant yield erosion.
- Combine palladium acetate (10 mol%), DPPF ligand (20 mol%), TFBen (3.0 equivalents), triethylamine (2.0 equivalents), propargyl amine derivative, and substituted benzyl chloride in acetonitrile solvent within a Schlenk tube under inert atmosphere.
- Heat the reaction mixture to precisely 110°C while maintaining continuous stirring for exactly 24 hours to ensure complete conversion without side product formation.
- Perform post-treatment by filtering through silica gel followed by column chromatography purification using standard techniques to isolate the target pyrrolone compound at pharmaceutical-grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process addresses critical pain points in pharmaceutical supply chains by delivering a more reliable route to essential pyrrolone intermediates that serve as building blocks for numerous therapeutic agents including antibiotics and antidiabetic compounds currently marketed globally. The elimination of specialized high-pressure equipment requirements significantly reduces capital expenditure barriers while enhancing operational flexibility across different manufacturing sites worldwide through standardized process parameters that maintain consistent performance regardless of regional infrastructure limitations.
- Cost Reduction in Manufacturing: The substitution of gaseous carbon monoxide with solid phenol tricarboxylate surrogate eliminates expensive pressure-rated reactors while reducing raw material costs through simplified procurement logistics; the one-step nature minimizes solvent consumption compared to multi-step conventional methods leading to substantial operational cost savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with established global supply networks ensures consistent raw material availability regardless of regional market fluctuations; the robust reaction profile maintains high performance across different production scales providing procurement teams with predictable lead times essential for just-in-time inventory management systems.
- Scalability and Environmental Compliance: The mild reaction conditions enable seamless scalability from laboratory benchtop to multi-ton production volumes while reducing waste generation through atom-efficient catalysis; this aligns with global environmental regulations while lowering disposal costs making it particularly attractive for manufacturers seeking sustainable production certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation based on extensive process development data from experimental examples demonstrating consistent performance across fifteen substrate variations under standardized reaction conditions validated through multiple scale-up trials.
Q: How does this method improve upon conventional pyrrolone synthesis techniques?
A: The patented process eliminates multi-step sequences through a single-step palladium-catalyzed bis-carbonylation operating under ambient pressure with safe carbon monoxide surrogates, achieving higher yields (70–92%) across diverse substrates while reducing waste generation through atom-efficient catalysis compared to traditional high-pressure carbonylation methods.
Q: What supply chain advantages does this technology offer for pharmaceutical manufacturers?
A: By utilizing commercially available starting materials with established global supply networks and eliminating specialized high-pressure equipment requirements, this method ensures consistent raw material availability while providing predictable lead times regardless of regional market fluctuations or geopolitical disruptions.
Q: How does this process support commercial scale-up from laboratory to production volumes?
A: The mild reaction conditions (100–120°C) and absence of exothermic hazards enable seamless scalability from laboratory benchtop to multi-ton production volumes while maintaining consistent yield profiles across scales due to robust process parameters validated through extensive experimental data.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolone Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-million levels required by international pharmacopeias; this patented methodology represents just one example of our commitment to developing innovative solutions that address complex synthetic challenges in pharmaceutical intermediate manufacturing with exceptional reliability.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific supply chain requirements; please contact us for detailed COA data and route feasibility assessments tailored to your production needs.
